Clinical and Radiological Factors Affecting Thoracolumbar Fractures Outcome: WFNS Spine Committee Recommendations
Article information
Abstract
To obtain a list of recommendations about clinical and radiological factors affecting outcome in thoraco-lumbar fractures with the aim of helping spine surgeons in daily practice. A systematic literature search in PubMed and Google Scholar database was done from 2010 to 2020 on the topic “thoracolumbar fracture AND radiology AND surgical outcomes” and “thoracolumbar fracture AND radiology AND surgical outcomes.” A total of 58 papers were analyzed and WFNS (World Federation of Neurosurgical Societies) Spine Committee organized 2 consensus meetings to formulate the specific recommendations the first in Peshawar in December 2019 and in a subsequent virtual meeting in June 2020 to reach an agreement. Both meetings utilized the Delphi method to analyze preliminary literature review statements based on the current evidence levels to generate recommendations through a comprehensive voting session. Eight statements were presented and reached the consensus about this topic. A variety of clinical factors is known to influence outcome of patients with thoracolumbar fractures. Some of these are well-known established factors such as blood pressure augmentation and patient age, while some are not well studied. Overall, the quality of evidence is low and we need more randomized controlled studies to validate our results. Similarly, radiological factors that can predict outcome are well stated and there is a high accordance worldwide. In reverse, still under debate is the application to choose which surgical treatment is advisable based on them.
INTRODUCTION
Thoracolumbar region represents one of the most common fracture sites in spinal injuries. Management is still a big challenge in spine surgery. Despite tremendous improvements in spinal imaging and management techniques over the last 2 decades, there is still a lack of consensus in several areas and no standard guidelines for surgical approach, fusion levels, amount of kyphosis correction and timing of treatment and their influence on clinical outcome are present [1].
As these fractures are usually treated by surgical intervention, posterior pedicle screw placement serves as the most common technique to address these fractures worldwide [2]. The instrumentation serves as a tool to provide mechanical stability until the bony union takes place. Based on these principles, postoperative recollapse of a well-reduced vertebral body or progressive kyphosis could be a lethal complication. These could increase neurological deficits, implant failure, and revision surgery [3,4]. Unfortunately, it is quite difficult to predict recollapse or implant failure due to a magnitude of clinical and radiological factors coming into play [5]. In 1994, McCormack et al. [6] suggested a load-sharing classification to predict implant failure; however, the scoring system had a major limitation of not including patient-related clinical factors.
Moreover, the outcome can be influenced by many other different factors that can be divided into 2 main groups: clinical factors (such as American Spinal Injury Association [ASIA] grade at admission, preoperative neurological status, time and type of surgery, age, osteoporosis, body mass index [BMI], and major comorbidities) and radiological ones (such as fracture type - AOSpine classification, anatomic location of the injury, khyphotic deformity, etc.) [7].
To deal with these issues and try to reach some globally accepted recommendations, the World Federation of Neurosurgical Societies (WFNS) Spine Committee organized a Consensus Conference on thoracolumbar fracture management. In this paper, we present the recognized factors to influence outcome with the aim of assisting physicians in their daily practice, in line with previous recommendations published by WFNS regarding some different topics in spinal surgery.
MATERIALS AND METHODS
A systematic literature search in PubMed and Google Scholar database was done from 2010 to 2020. The quality assessment and clinical relevance criteria utilized were in accordance with the Agency for Healthcare Research and Quality criteria for diagnostic studies and observational studies. Level of evidence was defined as level I to IV based on the quality of evidence developed by the U.S. Preventive Services Task Force for therapeutic interventions. Strength of evidence rate was ranked as mild, moderate, or high.
The literature research was performed using the following keywords: (1) clinical factors thoracolumbar fracture outcome, (2) thoracolumbar fracture AND radiology AND surgical outcomes.
According to the review and the specific topic, we assessed the following questions to be answered: (1) How many factors can affect outcome?; (2) Which clinical factors influence outcome?; (3) Which radiological factors can affect outcome?
This data was reviewed in a consensus meeting in Peshawar in December 2019 and then in a virtual meeting (due to limitations correlated with the coronavirus disease 2019 emergency) in June 2020 to reach an agreement. Both meetings utilized the Delphi method to administer the questionnaire to preserve a high degree of validity. To generate a consensus, the levels of agreement or disagreement on each item were voted independently in a blind fashion through a Likert-type scale from 1 to 5. The consensus was achieved when the sum for disagreement or agreement was ≥ 66%. Each consensus point was clearly defined with evidence strength, recommendation grade, and consensus level provided. The important factors are discussed below.
Based on the most significant literature, 8 statements were defined, presented, and voted.
RESULTS
With the keywords “clinical factors thoracolumbar fracture outcome” the search yielded 175 studies in Google scholar and 152 in PubMed, respectively. Some of the results were duplicated between the 2 databases. The search was further refined by excluding the duplicate articles, those not in English, or when based on animal or cadaveric subjects. The results included case reports, case series, prospective and retrospective studies, randomized studies, systematic reviews, and meta-analysis, shortlisting the count to 71. After reading the abstract, only 40 articles were selected for a full review in this consensus meeting (Fig. 1).
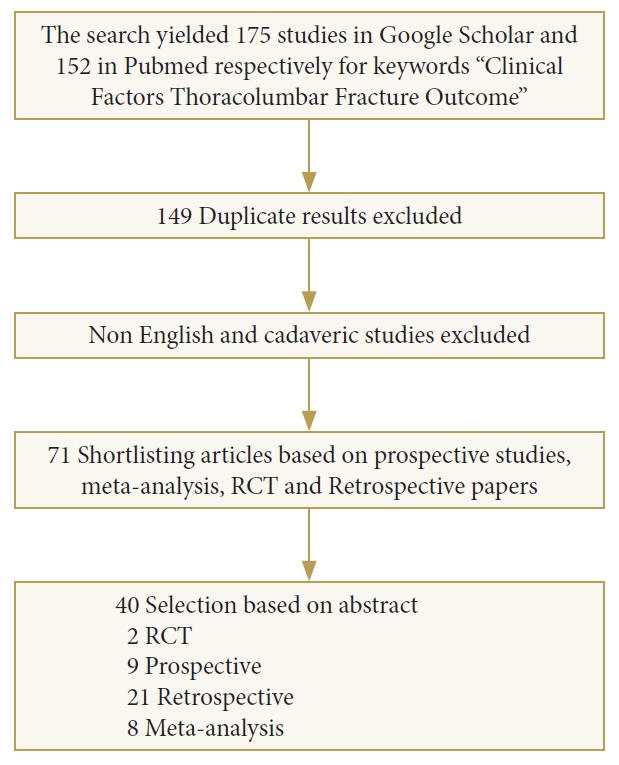
Flow chart of a literature search of thoracolumbar fracture outcome and clinical factors. RCT, randomized controlled trials.
With the keywords “thoracolumbar fracture AND radiology AND surgical outcomes” there were 526 results in PubMed and MEDLINE. A manual search resulted in 2 further articles. We removed non-English language papers, case reports, and low-quality case series. Then, we analyzed 18 papers for this review. A flowchart of the literature search is shown in Fig. 2.
Analyzing the literature, we subdivided the factors affecting outcome into 2 main groups: clinical and radiological factors.
The clinical factors recognized to predict surgical outcome are: pre-existing neurological status, timing of surgery, blood pressure, age, bone strength, BMI, smoking, and comorbidities.
While the most relevant radiological factors usually used to choose the surgical treatment and predict outcome are: vertebral body height (VBH), interspinous distance; anatomical integrity of structures such as posterior ligamentous complex (PLC), facet joints/pedicles and disc, spinal canal stenosis, displacement/dislocation of the vertebral bodies, spinal lordosis/kyphosis, and consequent deformity correction and proper reduction by stabilization.
Based on the most significant literature, 8 statements were defined and are summarized in Table 1. All the selected statements on this topic reached a positive consensus in both Consensus Meetings of the WFNS Spine Committee, as detailed in the following part.
DISCUSSION
Management of thoracolumbar is still a topic under debate. Not only do clinical and surgical managements present some open queries, but the definition of outcome can also be challenging. In fact, as reported by Rojas-Tomba et al. [8] there is often a discrepancy between radiological findings/outcome and clinical outcome.
1. Clinical Factors Affecting Outcome
1) Pre-existing neurology
ASIA scoring methodology was first published in 1982 and is widely used in evaluating, treatment, and outcomes of spinal cord injuries [9]. Most of the current studies indicate the ASIA grade at presentation is the most important factor determining the outcome. Dimar et al. [10] in a landmark study, showed that the severity of the neurologic injury was one of the most important factors influencing the outcome, including postoperative complications and longer hospital stay. Kaminski et al. [11] assessed the degree of functional improvement 1 year after spinal injury in 76 patients. Among factors such as age, sex, level, type of injury, comorbidities, and ASIA grade, ASIA grade was the dominant factor influencing the outcome. Over the course of years, multiple grades to assess neurology have been introduced. Irrespective of the scoring system used, the basic concept remains the same. However, ASIA grading continues to be the most commonly employed score due to easy reproducibility.
2) Timing of surgical intervention
The ideal time for a patient with a thoracolumbar fracture to undergo surgery has been a topic of long-standing debate. However, most of the current literature now suggests early surgery that is less than 72 hours may reduce hospital stay and morbidity and mortality. Bellabarba et al. [12] performed a systematic review of literature from 1990 to 2008 and indicated that early surgery led to better outcomes with less morbidity and mortality. Similarly, Bourassa-Moreau et al. [13] also recommended early surgery within 24 hours for spinal injuries. In a setting of associated systemic injuries, up to 72 hours of delay may be acceptable. Another important paper by Kato et al. [14] involving many spinal surgeons like Vaccaro and Fehlings suggests early surgery in patients with thoracolumbar fractures. Although the evidence is weak, their recommendation is largely based on the results extrapolated from patients with cervical spinal injuries. Early surgical intervention has been proven to be beneficial.
3) Blood pressure
Spinal cord ischemia is believed to play a central role in the secondary injury processes that cause delayed and progressive injury to the spinal cord [15]. The secondary injury can be aggravated by hypotension, which develops due to loss of sympathetic outflow in the thoracic region. The first guidelines regarding the management of blood pressure came in 2002. Those guidelines recommended that a mean arterial blood pressure (MAP) of 85–90 mmHg should be targeted in the first 7 days after spinal cord injury [16]. Those guidelines are supported by a few weak level 3 studies performed in patients with cervical spine injuries [17]. Similarly, Walters et al. [18] also suggested that mean arterial pressure below 70 was associated with poor neurological recovery. Cohen et al. [19] further complemented these findings. They reported a retrospective study of the relationship between episodes of hypotension noted in the medical record and outcomes using step-wise regression. They concluded that there might be a threshold of around 70 mmHg below which worse outcomes are seen. One of the major limitations to all these studies is that blood pressure augmentation was supplemented by other aggressive strategies to improve neurological recovery. These included different forms of physiotherapy and rehabilitation. Because no baseline control exists for all these patients, it is difficult to pinpoint the isolated role of blood pressure in improving overall outcomes. Such limitations were overcome by a detailed retrospective analysis conducted by Hawryluk et al. [20] Their results showed that the beneficial effect of high blood pressure was applicable only during the first 3 days of spinal cord injury.
The majority of the patients require some vasopressor therapy to increase blood pressure, particularly when a patient is in spinal shock. The Consortium guidelines published in 2008 declared different inotropes according to the level of injury [21]. Their recommendation was to use dopamine or norepinephrine for injuries above T6 level. However, one study demonstrated that many patients received phenylephrine at or above T6 and dopamine below T6 in conflict with the guidelines, most likely due to the preguideline patients and physician comfort with dopamine [22]. The study also showed that dopamine was significantly associated with complications even when used according to MAP goal guidelines. De Backer et al. [23] performed a meta-analysis of 2,768 patients in septic shock. His results showed that dopamine use was associated with high mortality. As a result, the current trend is to recommend norepinephrine as the first-line vasopressor for blood pressure augmentation.
4) Age and bone strength
Over the past decade, multiple clinical factors have been studied to identify their correlation with thoracolumbar fractures outcome. Age is the most extensively evaluated, with increasing age correlating with the poor overall outcome. A retrospective analysis of 208 patients was performed by Jang et al. [24] in 2019. He compared various clinical variables such as BMI, smoking, age, height, and comorbid conditions. He concluded that age > 43 years was the most important clinical factor predicting implant failure or postoperative recollapse of the vertebra. Similarly, a nationwide survey was conducted by Purvis et al. [25]. The results showed that in patients aged > 75 years, hospital stay and complications rates were much higher in operative patients compared to the nonoperative group. Although the survey had many limitations, it was still able to highlight the importance of age while planning surgical intervention. Another study by Cankaya et al. [26] highlighted the importance of age when managing elderly patients with thoracolumbar fractures. Although none of his 21 patients underwent surgery, he concluded that age > 71 years was a strong predictor of complications. Patrick conducted a retrospective review of 73 patients who underwent anterolateral fixation for thoracolumbar fractures [27]. His results showed that supplemental posterior fixation was needed in patients aged more than 59 years.
Although the association of increasing age with postoperative complications has been well established, we can still not demarcate the exact limit at which complications are more likely to occur. Based on the available literature, patients aged over 45 years are at a higher risk of implant failure or revision surgery.
5) Body mass index
Increased BMI or body weight has been associated with increased risk of postsurgical infections and an overall increase in morbidity and mortality. Soroceanu et al. [28] performed a retrospective review of 241 patients, of whom 66 were obese undergoing spinal deformity correction. His patients were followed up to 2 years postoperatively with periodic radiological assessment. His results showed that obese patients (BMI> 30 kg/m2) were at considerably high risk of complications and wound infections than nonobese patients. Although all patients improved after surgery, the degree of improvement was also less in the obese group than the nonobese group. These results are consistent with the results of others, including a meta-analysis by Jiang et al. [29]. Their study demonstrated that obesity is an independent risk factor for surgical-site infections. In patients undergoing lumbar fusion surgery, Mehta et al. [30] found that the thickness of subcutaneous fat and the skin-to-lamina distance are risk factors for surgical-site infections. This is probably because thicker subcutaneous fat may require further retraction, leading to increased dead space postoperatively, which in turn increases the risk of infection. Similarly, Formica et al. [31] performed short-segment posterior fixation with an intermediate screw in 43 patients with thoracolumbar fractures. His patients were followed for 1 year with periodic x-rays to assess loss of kyphosis. His results showed that loss of kyphosis correction was mostly seen in obese patients while other factors like sex and smoking did not influence the outcome.
6) Comorbid conditions/high-dose steroids
As stated previously, higher-aged patients are at greater risk of complications irrespective of the treatment modality chosen, surgical or nonsurgical. One of the major contributing factors is the presence of underlying comorbidities such as diabetes, hypertension, and ischemic heart disease, which complicates the management of a trauma patient. A prospective multicentric study was conducted by Dimar et al. [10] in 2010 involving 230 patients. Factors such as neurological status at presentation, age, smoking, steroid use, and comorbidities through Charlson Comorbidity Index (CCI) were evaluated while following the patients for 6 months. An important parameter to note was that about 35% of patients were smokers. Multivariate logistic regression analysis showed that high CCI and steroid use were the most important clinically significant factors leading to untoward complications amongst all the stated factors. The CCI was first described in 1987 and is commonly used to estimate the risk of death from comorbid conditions [32]. Each specific comorbidity is scored according to a weighted index to predict the cumulative effect of medical illnesses on mortality. In addition to CCI, multiple other scores have been developed to assess comorbidities, including the modified friability index and American Society of Anesthesiologists (ASA) physical status scoring system. Irrespective of the system used, all the scoring systems work similarly. A comparative analysis of all these comorbid scoring systems was done by Ondeck et al. [33]. He performed a retrospective survey of 16,495 patients who underwent elective lumbar fusion. Although none of the selected patients were of trauma, his results showed that ASA physical status scoring was the most sensitive predictor of poor outcome with higher ASA physical status grades correlating with higher complications.
After a spinal cord injury, the use of steroids has been a controversial issue since the completion of the NASCIS (National Acute Spinal Cord Injury Study) studies, which demonstrated a small but significant improvement in neurologic outcomes when high doses were given intravenously immediately after spinal cord injury [34]. However, historical data on the use of methylprednisolone in spinal injuries have been challenged, with more recent studies stating no benefit of steroids [35]. Similarly, Chiu et al. [36] used steroids in 6 of his 8 patients presenting with L1 burst fracture with conus medullaris syndrome. His results showed that steroids played no role in patient recovery, and their use was largely dictated by faith rather than objective evidence. A survey published in 2006 revealed that most respondents continue to administer methylprednisolone, but they are motivated predominantly by fear of litigation [37].
7) Smoking
The injurious effects of smoking on health are well known. A recent review summarized the results from multiple studies documenting the negative impact of smoking on the overall hospital care of surgical patients, including increased risk of perioperative complications, morbidity, mortality, and admission to the intensive care unit in patients undergoing general surgery [38]. The complications associated with smoking include wound infections, delayed wound healing, pneumonia, and myocardial infarction [39]. Smoking has similar adverse effects in patients with spinal injuries and is known to impair the fusion process significantly in patients undergoing spinal fixations. A cross-sectional analysis of 136,511 patients with spinal diseases was performed by Bisson et al. [40] over 5 years. Ten percent of his patients were smokers. His results showed that smoking patients admitted for spinal disease in the sample had worse outcomes, increased complications, and higher costs than their nonsmoking counterparts. Similarly, Berman et al. [41] showed that smoking significantly increases the risk of pseudoarthrosis for patients undergoing both lumbar and cervical fusions. In addition to nonunion, smoking also increases the risk of other perioperative complications such as infection and adjacent-segment pathology.
However, Dimar et al. [10] in their prospective study, were unable to document any deleterious effects of smoking in the management of patients with thoracolumbar fractures. Similarly, Kuo et al. [42] performed spinal surgery including fixations in 306 patients. Thirty-four patients were smokers, and all patients were clinically and radiologically followed for 2 years. His results did not show any significant increase in complications or pseudoarthrosis among smokers compared to nonsmokers. On the contrary, Antoni et al. [43] performed anterior cage placement in 106 patients with thoracolumbar fractures. His results showed a higher rate of pseudoarthrosis among smokers compared to nonsmokers. The effects of smoking in delaying fusion and pseudoarthrosis are well known. However, such pseudoarthrosis does not always translate into clinical symptomatology. Since the current literature shows no benefit of fusion versus nonfusion surgery in thoracolumbar fractures [44,45]. it might explain the paucity of data correlating smoking with adverse outcomes in patients with thoracolumbar fractures (Table 2).
2. Radiological Factors Affecting Outcome
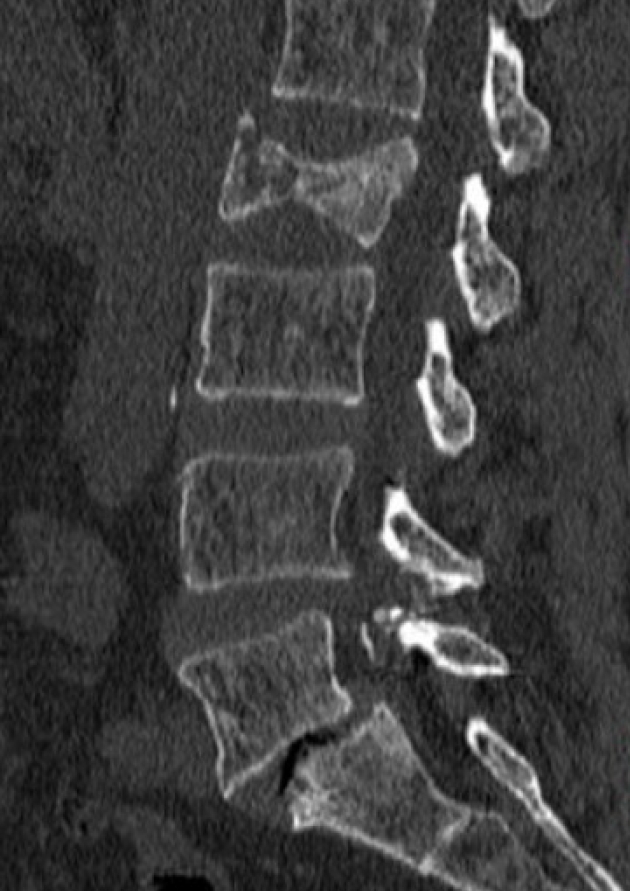
1) Vertebral body height
This parameter can be studied by x-ray and detailed with a computed tomography (CT) scan. It is an important parameter because a VBH loss > 50% with kyphotic deformity influence both type of surgical treatment and outcome in terms of postoperative disability and pain [46] (Fig. 3).
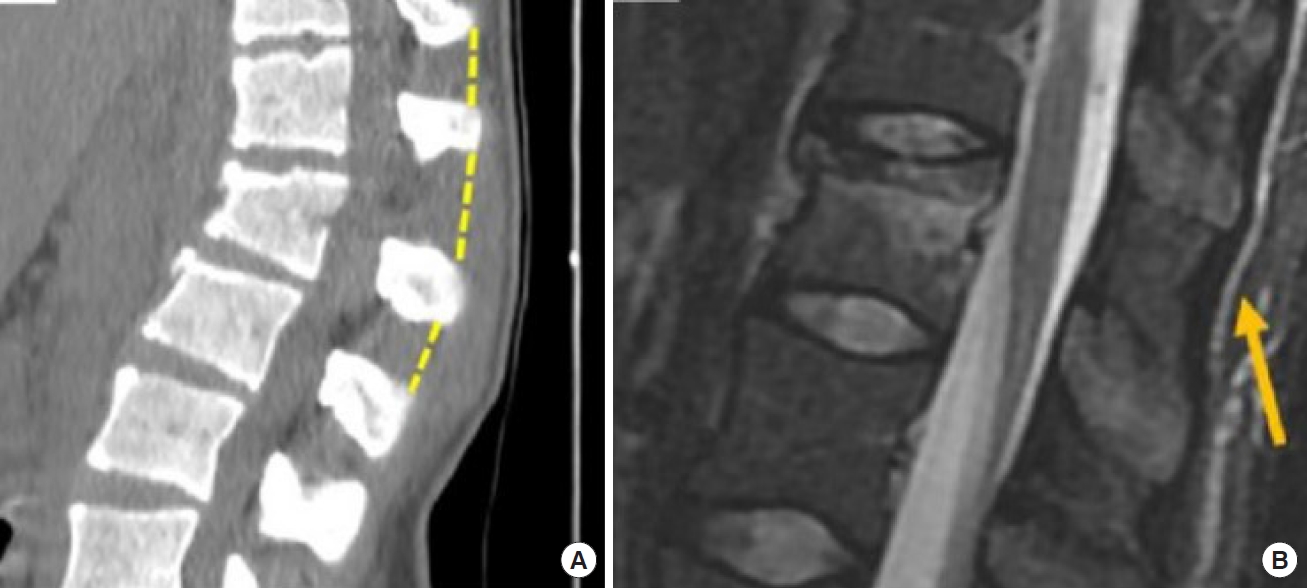
2) PLC/interspinous distance variation
The failure of PLC significantly influences the severity of the fracture and is related to neurological damage in patients with acute thoracic and lumbar burst fractures, furthermore it correlates with treatment that can shift from conservative to surgical [2].
It can be suspected on plain x-ray but should be characterized by CT scan. Significant interspinous distance increase can relate with pure osseous failure of the posterior tension band [47].
Radiological signs such as vertebral translation > 3.5 mm or interspinous distance variations > 20% of widening (up to 7 mm can be considered normal) are both suggestive of an unstable fracture that requires surgical treatment [48] (Fig. 4A, B).
3) Facet joints/pedicles
The first-line x-ray can detect facet joint luxation but to better characterize facet joints and pedicles situation CT and magnetic resonance imaging (MRI) scans are necessary. The presence of facet joint-pedicle complex subluxation-dislocation-fracture uni- or bilateral is an important factor that can predict outcome, in terms of risk of fracture progression without adequate correction, and to shift for a surgical treatment and which type of surgical treatment [49].
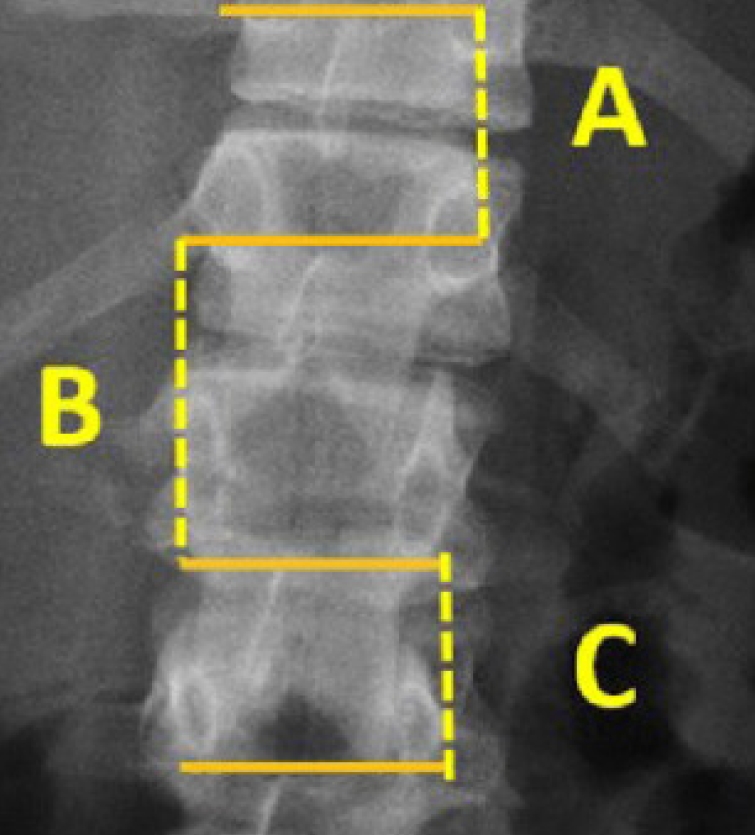
4) Intervertebral disc space
This parameter can be detected on x-ray in terms of narrowing of the discal space and coronal and sagittal displacement, but an MRI scan is necessary to characterize it. It is important because a discal injury is advisable of instable fractures and requires surgical treatment [47,50,51] (Fig. 5).
5) Displacement - dislocation
The presence of spinal displacement or dislocation in coronal, sagittal, and axial planes is advisable of highly unstable thoracolumbar fracture because it implies the affection of all the spinal columns. Its presence requires surgical treatment and is highly suggestive for severe neurological deficits. Its presence negatively affects clinical and radiological outcome [52].
6) Spinal canal diameter
Another important factor that can influence outcome is the neurological damage varies between vertebral levels. Nevertheless, a sagittal-transverse diameter ratio < 0.40 is highly associated to neurological injury and requires surgical intervention to decompress neurological structures [53].
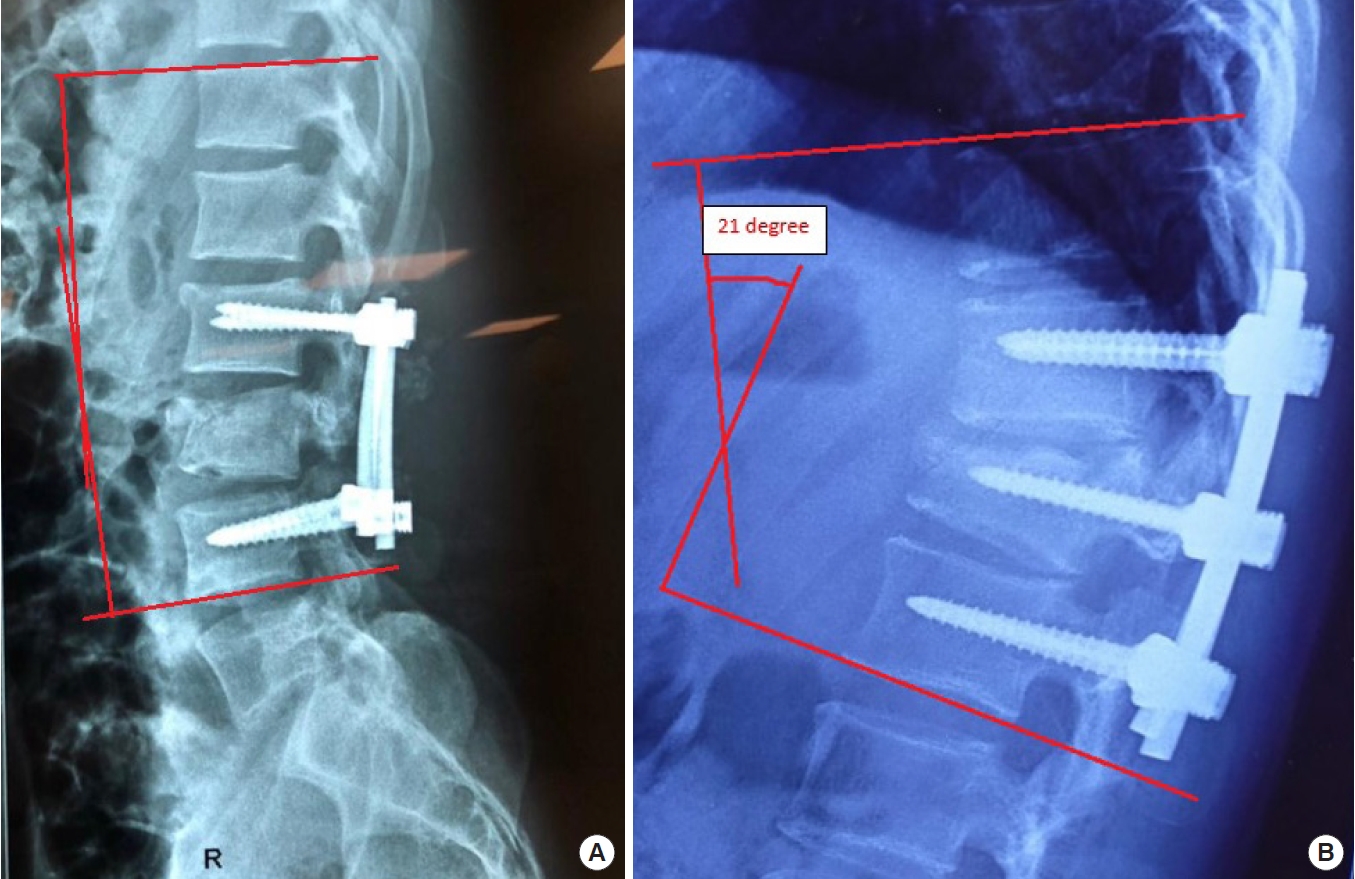
7) Spinal lordosis
Lastly, spinal lordosis should be evaluated. Insufficient correction leads to late progression of kyphosis, the return to the normal range of thoracolumbar curvature is necessary to avoid additional excessive loading to the spine, which might be another secondary complication with chronic pain and disability [54].
The usual target of correction is Cobb angle < 7° for the reasonable functional outcome [55].
Correction loss at the follow-up (Cobb ≥ 20°) has a strong correlation with severe back pain and postoperative disability [56].
Seo et al. [56] correlates insufficient correction to late progression of kyphosis (Cobb angle > 10.5°) immediately after surgery with unfavorable radiological outcome, which is related to a poor clinical outcome (Fig. 6A, B).
CONCLUSIONS
A variety of clinical factors is known to influence outcome of patients with thoracolumbar fractures. Some of these are wellknown established factors such as blood pressure augmentation and patient age, while some are not well studied. Overall, the quality of evidence is low and we need more randomized controlled studies to validate our results.
On the other hand, radiological factors that can predict outcome are well stated and there is a high accordance worldwide. In reverse, still under debate is the application to choose which surgical treatment is advisable based on them.
WFNS SPINE COMMITTEE RECOMMENDATIONS
Based on the presented literature and personal experience, the Steering Committee voted the following statements with a positive consensus and they were converted to recommendations.
• Obesity can worsen segmental kyphosis following surgery for a thoracolumbar burst fracture.
• Increasing age is a predictor of poor outcome.
• Comorbidities, smoking, and long-term high-dose steroid usage predict poor outcome.
• Polytrauma and high injury severity score should not be considered as a contraindication for early surgery.
• Anterior VBH loss more than 50% may lead to progression of kyphotic deformity.
• Detection of injury of posterior ligamentous complex is important, as it significantly influences the outcome.
• Burst fractures with sagittal-transverse canal diameter ratio < 0.40 are highly associated with neurological injury and worse outcome.
• Cobb angle > 10.5° after surgery may predict poor outcome.
Notes
The authors have nothing to disclose.