Upper Cervical Compression Myelopathy Caused by the Retro-Odontoid Pseudotumor With Degenerative Osteoarthritis and Calcium Pyrophosphate Dihydrate Disease: A Case Report and Literature Review
Article information
Abstract
The retro-odontoid pseudotumor is often concurrent with atlantoaxial subluxation (AAS). Therefore, the pseudotumor is relatively common in rheumatoid arthritis (RA) but rare in primary osteoarthritis (OA). This is a case report of an elderly male patient suffering from neck pain and compression myelopathy caused by the craniocervical pseudotumor with OA but without atlantoaxial instability. He had long-lasting peripheral and spinal pain treated by nonsteroidal anti-inflammatory drugs. Imaging found upper cervical spondylosis without AAS or dynamic instability but with periodontoid calcifications and ossifications, suggesting calcium pyrophosphate dihydrate (CPPD) crystal deposition. Based on a comprehensive literature search and review, CPPD disease around the atlantodental joint is a possible contributor to secondary OA development and retro-odontoid pannus formation through chronic inflammation, which can be enough severe to induce compression myelopathy in non-RA patients without AAS. The global increase in the aged population advises caution regarding more prevalent upper cervical spine disorders associated with OA and CPPD.
INTRODUCTION
The retro-odontoid pseudotumor around the atlantodental joint is often concurrent with non-traumatic atlantoaxial subluxation (AAS) [1]. Therefore, pannus formation by atlantoaxial instability is a relatively common complication in patients with rheumatoid arthritis (RA), a chronic, systemic inflammatory disease [2]. The cervical spine is a popular focus of ligamentous laxity and joint destruction by RA, and AAS is the most frequently observed involvement [3-5]. The pseudotumor can occur even in patients without RA [6], which is however a rare condition in upper cervical osteoarthritis (OA) [1,7]. Although the development of OA is multifactorial, altered mechanical properties arising from degenerative instability [8,9] with calcification and/or ossification [10,11] are the primary cause [12]. Then, OA can secondarily be developed often more severely by other factors including trauma, inflammation, e.g., gout, and metabolic disorders, e.g., diabetes [13]. Because of less involvement of AAS in OA [14], there is only limited evidence regarding the link between the craniocervical pseudotumor, atlantoaxial instability, and OA [15].
We experienced an elderly male patient case of the retro-odontoid atlantodental pseudotumor with upper cervical OA including the occipitocervical region, thereby causing compression myelopathy. The patient did not have marked AAS but periodontoid calcifications and ossifications, suggesting the involvement of calcium pyrophosphate dihydrate (CPPD) crystal deposition disease, also known as pseudogout and pyrophosphate arthropathy. In this case, CPPD-induced chronic inflammation may be a causative factor of secondary OA and the atlantoaxial pseudotumor. We thus performed a comprehensive literature search and review. We discussed a possible contribution of CPPD to the retro-odontoid pseudotumor in non-RA but OA patients without AAS and also the selection of treatment options.
CASE REPORT
This study was approved by the Institutional Review Board (IRB) at Kobe University Graduate School of Medicine (IRB No. B190002). Written informed consent was obtained from the patient. Further, this patient was informed that data from the case would be submitted for publication, and gave his consent. This study was conducted in accordance with the principles of the Declaration of Helsinki and with the laws and regulations of Japan.
A 55-year-old Japanese man was referred to the authors’ hospital due to complaints of low back, neck, shoulder, elbow, and hip pain. His symptoms lasted long before visiting, but relieved conservatively by nonsteroidal anti-inflammatory drugs (NSAIDs). His low back pain resulted from lumbar spinal canal stenosis with disc herniation as shown by magnetic resonance imaging (MRI). Then, his visiting continued 1 to 3 times a year complaining joint pain without any abnormality reflected on the blood test. Radiographic peripheral joint findings were normal except for hip joint effusion detected by MRI when he was 57 years old.
At 62 years old, neck pain worsened with a limited range of motion. Cervical spine flexion–extension radiographs revealed no apparent atlantoaxial instability but structural changes were obscure because of bony overlapping. Then, MRIs showed slight cervical disc bulging in lower vertebrae and granulomatous soft-tissue swelling around the atlantodental joint that resembled the pseudotumor associated with AAS in RA (Fig. 1). Based on no spinal cord compression and rapid pain relief by NSAIDs, further examinations were not performed. He had medical history of hypertension but not diabetes mellitus, rheumatic diseases, allergic diseases, or metabolic disorders. However, as low back and leg pain by lumbar spinal stenosis had worsen, decompression surgery was performed at 66 years old, facilitating successful postoperative relief of symptoms.
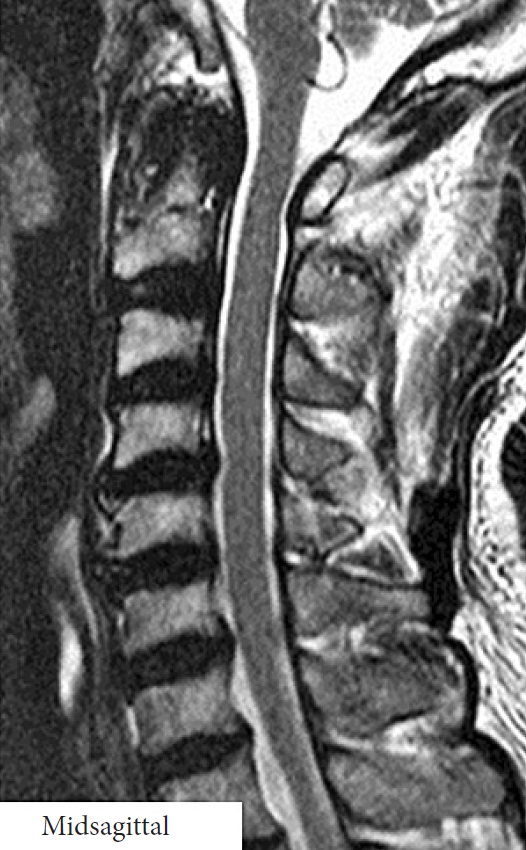
Midsagittal T2-weighted magnetic resonance imaging of the cervical spine in the male patient at 62 years old. Atlantodental joint swelling without spinal cord compression was observed.
At 68 years old, he felt severe neck and occipital pain with limited motion in extension with rotation, shooting pain in both upper extremities with hand clumsiness and numbness, and walking disturbance. Four days after the onset, he visited our hospital. Neurological examination revealed modest muscle weakness in left extremities; however, sensory sensation and deep tendon reflexes were normal except for elevated left ankle jerk. Laboratory blood and urine data were within normal limits including white blood cell count and C-reactive protein. Cervical spine radiographs demonstrated subaxial spondylosis including vertebral osteophytes with disc height narrowing and Barsony’s sign; however, no marked development of AAS or dynamic atlantoaxial instability was observed in flexion and extension positions (Fig. 2). Spinal cord compression by the enlarged retro-odontoid pseudotumor and C1 posterior arch with an intramedullary high signal-intensity lesion was detected on T2-weighted MRIs (Fig. 3). Computed tomography (CT) scan showed degenerative changes with calcifications and osteophytes around the occipitocervical junction but no ossification of the anterior longitudinal ligament (OALL) (Fig. 4). According to neurological and radiological findings with previous disease episodes, this patient was diagnosed with compression myelopathy due to the retro-odontoid pseudotumor associated with OA and CPPD but without RA or AAS.
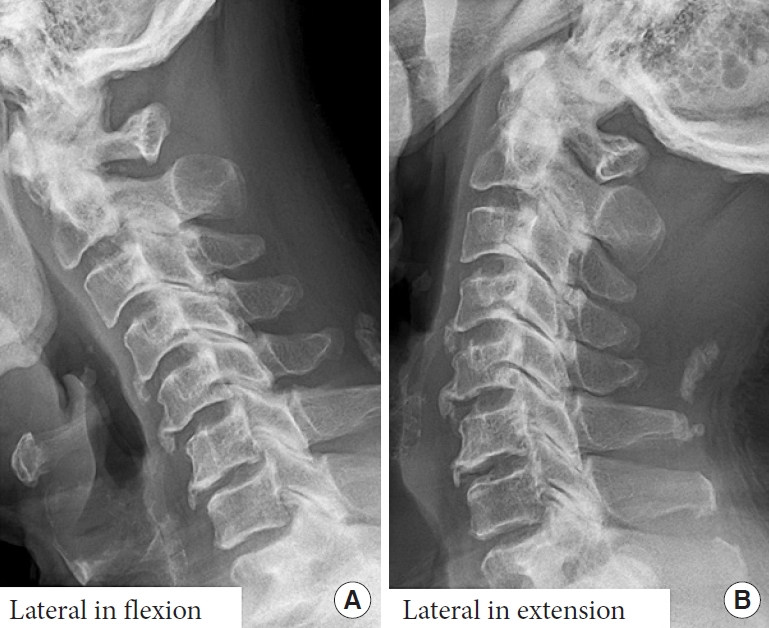
Lateral radiographs in flexion (A) and extension (B) positions of the cervical spine in the male patient at 68 years old. No apparent development of atlantoaxial subluxation but with upper cervical degenerative spondylosis was observed.
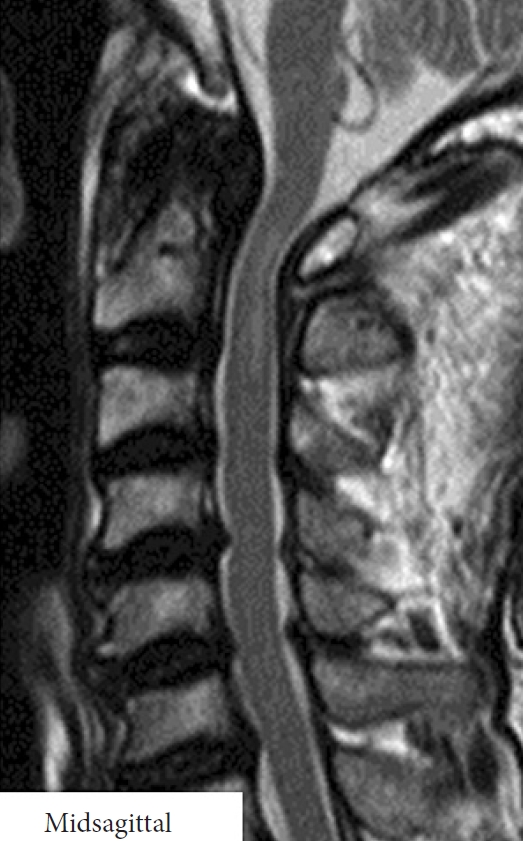
Midsagittal T2-weighted magnetic resonance imaging of the cervical spine in the male patient at 68 years old. Marked spinal cord compression with an intramedullary high-signal intensity lesion between the enlarged retro-odontoid pseudotumor and C1 posterior arch was observed.

Computed tomography images of the occipitocervical joint in the male patient at 68 years old. (A) In a midsagittal image, small but multiple calcifications (arrowhead) and osteophytes around the atlantodental joint were observed. (B) In a parasagittal image, calcifications (arrowheads), and narrowed joints with osteophytes (arrows) in the upper cervical spine were observed. (C) In a coronal image, ossifications of the transverse ligament (arrowheads) and degenerative joints (arrows) were observed. (D) In an axial image at C1–2, osteophytes and ossifications along with the transverse ligament (arrowheads) were observed. R indicates the right side of the body.
Because of the presented long tract sign and difficulty of daily activities, surgical resection of the posterior arch of the atlas was performed. No apparent atlantoaxial instability indicated decompression alone. His symptoms immediately disappeared after C1 laminectomy. No remarkable AAS progression in radiographs, maintained spinal cord decompression with a decreased intramedullary abnormal signal at C1–2 level on T2-weighted MRIs, although the size of the retro-odontoid pseudotumor remained relatively unchanged, and increased periodontoid calcifications and osteophytes in CT images, suggesting sustained CPPD inflammation, were monitored at postoperative 2-year follow-up (Fig. 5).
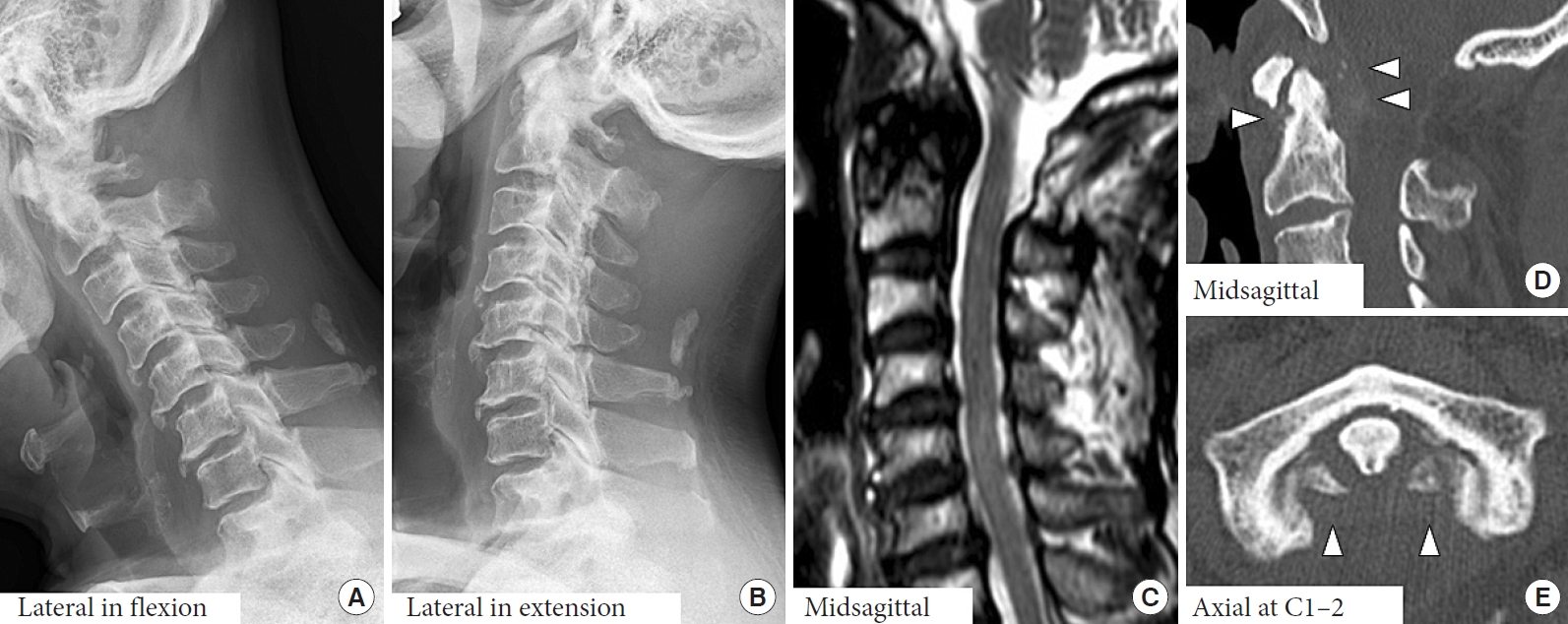
Lateral radiographs in flexion (A) and extension (B) positions, midsagittal T2-weighted magnetic resonance imaging (C), and midsagittal (D) and axial at C1–2 (E) computed tomography images of the cervical spine in the male patient at 70 years old. No remarkable progression of atlantoaxial subluxation was observed 2 years after laminectomy of the C1 posterior arch. Surgical decompression of the spinal cord at C1–2 level with an improved intramedullary high-signal intensity lesion was obtained. Meanwhile, elevated levels of periodontoid calcifications (D, arrowheads) and ossifications (E, arrowheads) were found, indicating calcium pyrophosphate dihydrate crystal deposition.
LITERATURE SEARCH
Literature search of scientific articles published between 1977 and 2019 was performed in PubMed (https://pubmed.ncbi.nlm.nih.gov/). Three primary keywords of “pseudotumor” (107), “OA” (531), and “CPPD” including “pseudogout” and “chondrocalcinosis” (372) were examined with the combination of “retro-odontoid,” “atlantoaxial,” atlantodental,” “atlantoodontoid,” “atlantodens,” and “cervical spine.” Numbers in the parenthesis showed in-relevant articles. Important articles regarding RA, AAS, and diffuse idiopathic skeletal hyperostosis (DISH) were additionally obtained by hand search. The abstract was evaluated and discussed by 2 authors (TY and TI), and 96 articles were selected eligible for the inclusion in this literature review. Based on 4 major topics of craniocervical “pseudotumor,” “OA,” “CPPD,” and “treatment” related to the presented patient case, 70 articles were finally referenced.
DISCUSSION
This is a case report of an older male patient suffering from neck pain and compression myelopathy due to chronic CPPD inflammation-induced secondary upper cervical OA and atlantodental pseudotumor even without AAS. Few prior papers reviewing the retro-odontoid “pseudotumor” with “OA” and “CPPD” have been published. The “treatment” is also undetermined. Therefore, we performed an in-depth literature review and discussed this patient case based on these 4 issues.
1. Non-RA Retro-Odontoid Pseudotumor
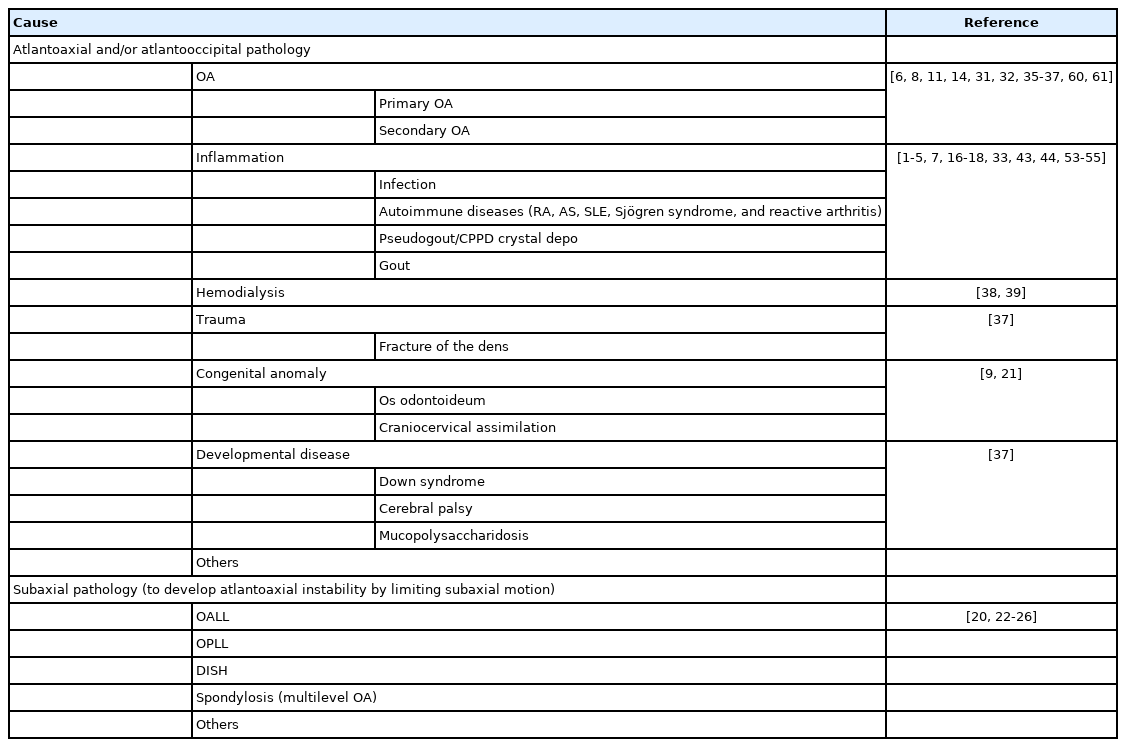
The retro-odontoid pseudotumor and/or AAS can be developed in patients with autoimmune diseases including RA [1-5], ankylosing spondylitis (AS) [16], systemic lupus erythematosus (SLE) [17,18], and Sjögren syndrome [18], which is also observed in non-RA patients with gout, pseudogout, hemodialysis, pigmented villonodular synovitis, and odontoid fracture nonunion [1]. Factors related to cervical spinal cord compression are synovial cyst, epidural lipoma and hematoma, and ossification of the posterior longitudinal ligament [1]. Although the generalized incidence of the retro-odontoid pseudotumor is unknown because of its rarity, the pseudotumor was detected by MRI in 23.2% of 164 patients with AAS surgically treated [19]. In more recent registry data from a consecutive MRI study of 105 patients with the pseudotumor, RA diagnosis was only 27.6% [7], indicating a common involvement of non-RA disease. It is noteworthy that 44.7% of non-RA patients, who were older and male-dominant, had clinical CPPD or imaging evidence for tissue calcification [7]. Then, the pathomechanism of the pseudotumor development in non-RA is considered as transverse ligament degeneration [6,20] due to the altered biomechanics of the craniocervical junction from congenital atlantooccipital assimilation anomaly [21] as well as subaxial ankylosis in severe spondylosis [22], OALL [23], Forestier disease [24], DISH [25,26], and AS [16]. A systematic review of the pseudotumor without radiographic instability failed due to the limited number of cases available, which although had different etiologies including atlantoaxial hypermobility, deposition of substances, and probably disc herniation [15]. Reported causes of AAS and the retro-odontoid pseudotumor are summarized in Table 1.
Here we reported a non-RA male patient with the atlantodental pseudotumor and upper cervical compression myelopathy. He complained neck pain and arthralgia, showing craniocervical OA and periodontoid calcifications and ossifications without AAS. Based on our literature review, CPPD crystal deposition is suggested to be involved.
2. Upper Cervical OA
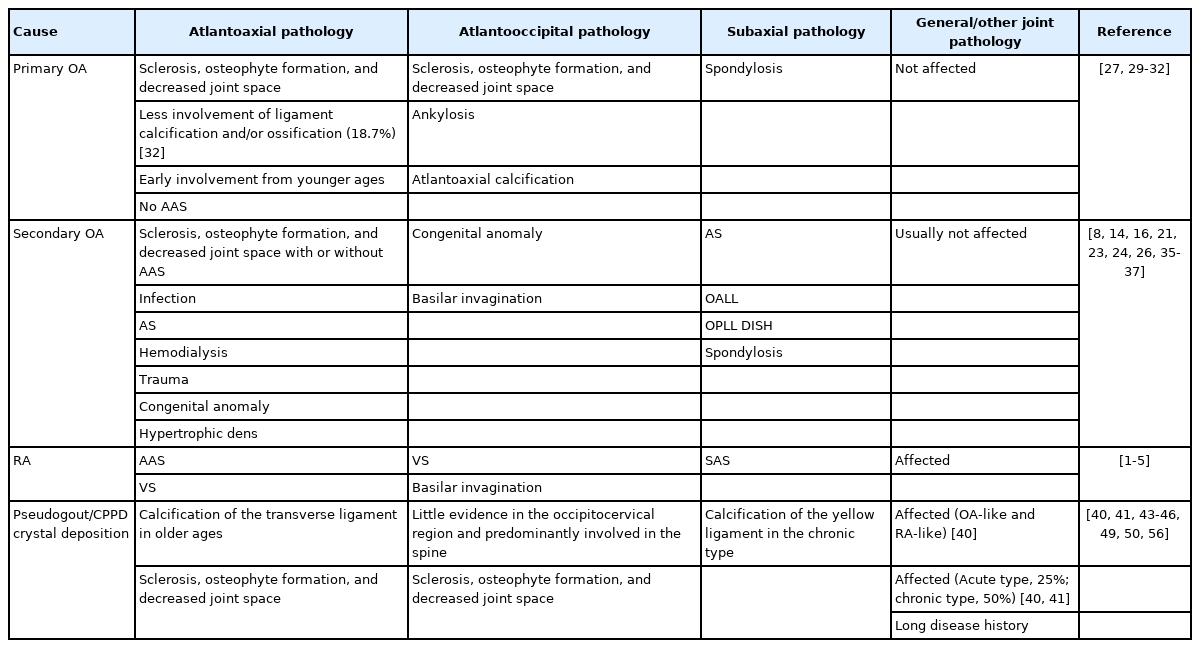
In 31 patients with atlantoaxial OA, both the atlantoodontoid and lateral mass joints, only the atlantoodontoid joint, and only lateral mass joints were radiologically involved in 71.0%, 16.1%, and 12.9%, respectively [27]. The importance of CT evaluation, identifying atlantoodontoid OA and transverse ligament calcification, was later emphasized in middle-aged and older patients with occipitalgia and limited neck motion because of the difficulty in radiographically assessing overlapping craniocervical bony structures [28]. Upper cervical CT examination of 700 patients without trauma clarified an age-dependent increase in the prevalence of atlantoodontoid OA: 16% in 18–25 years, 23% in 25–30 years, 33% in 30–40 years, 54% in 40–50 years, 70% in 50–60 years, 87% in 60–70 years, and 93% in > 70 years [29]. A CT study of 1,543 patients at a trauma center showed an age-dependent decrease in the atlantodental interval and increase in bone cyst formation and synovitis with calcium deposition around the dens in > 40 years old [30]. Consecutive 700 patients undergoing brain or paranasal sinus CT exhibited increased transverse ligament calcification with age and advanced atlantoodontoid degeneration [31]. Furthermore, mean 32.6-year-old male porters carrying loads on the head radiologically presented joint space narrowing with osteophytes, interspinous and transverse ligament calcifications, and occipito-atlantoaxial joint ankylosis, indicating primary OA, but did not all develop AAS or pseudotumor [32]. Based on these findings, OA is common with age in the atlantoodontoid and lateral mass joints. Then, age-related increase in retro-odontoid soft-tissue thickness was found on CT [33] and MRI [34], particularly in male patients with OA and/or undergoing dialysis [34]. Cervical compression myelopathy also resulted from degenerative AAS [14] and/or dens hypertrophy [11,35,36]. The periodontoid soft-tissue mass resembling to the pseudotumor was detected in 90% of 108 surgically treated patients with degenerative atlantoaxial instability resulting from trauma and congenital anomaly without RA or CPPD [37]. The pseudotumor with amyloid deposition can be caused by atlantoaxial instability due to secondary OA in hemodialysis patients [38,39]. Consequently, secondary OA is often associated with the development of atlantoaxial instability and the retro-odontoid pseudotumor.
The presented patient had long-term episodes of neck pain without episodes of chronic mechanical stress in the upper cervical spine. Imaging examination displayed no OALL, DISH, or abnormal biomechanics but spondylosis with periodontoid calcifications and ossifications, suggesting CPPD as a cause of secondary OA.
3. CPPD Disease
The CPPD disease comprises a variety of clinical phenotypes including OA-like and RA-like [40]. Crystals of CPPD are known to induce joint inflammation, bony erosion, and cartilage destruction, possibly resulting in degenerative OA [40]. The prevalence of the chronic polyarticular type of CPPD is roughly 50% while the acute type is approximately 25% [41]. A national study of United States veterans also showed the chronic progression in more than half of cases [42]. While the crowned dens syndrome is a common acute-type CPPD disease in the craniocervical junction [43,44], Chronic CPPD crystal deposition of the ligamentum flavum occurs frequently in the cervical spine [45,46]. On craniocervical CT for acute trauma, a prevalence of atlantoaxial CPPD was 12.5% of 513 patients, increasing with age [33]. Another study detected a similar CT-based prevalence of periodontoid CPPD as 13.5% of 296 patients suspected of brain disease, showing an age-dependent increase [47]. Although calcification of the transverse and alar ligaments around the atlantoodontoid joint was observed in 60%–70% of patients with pseudogout, the majority were asymptomatic with normal serological findings while only a small percentage of those exhibited neck pain and fever [48-51]. Recurrent sterile spondylodiscitis and epidural abscess by atlantoaxial CPPD were also observed [52]. The retro-odontoid pseudotumor in patients with CPPD often displays iso-signal intensity on T1-weighted MRIs and iso-signal to high-signal intensity on T2-weighted MRIs [53,54]. Histopathologically, CPPD crystal deposition can be confirmed from surgical specimens through transoral resection [44,53,55]. The CPPD disease causes inflammatory responses more predominantly in the craniocervical junction than in the subaxial spine, demonstrating occipital pain, numbness, and paresthesias as well as lower cranial nerve deficits [43,44]. Due to CPPD inflammation, atlantoodontoid and occipitocervical OA changes with narrowed joint spaces, osteophytes, and transverse and alar ligament calcifications and/or ossifications were observed [49,50,56]. Despite no reports comparing the severity between primary and secondary OA, CPPD-induced secondary OA should manifest more extensive degeneration than primary OA because of persistent inflammation. Reported radiological characteristics of primary causes of the retro-odontoid pseudotumor are summarized in Table 2.
The current patient with long-standing periodical neck pain without serological inflammation would suffer from chronic CPPD with OA progression in the craniovertebral region. The identified retro-odontoid pseudotumor had iso-signal intensity on T1-weighted MRIs and low-signal intensity on T2-weighted MRIs, showing a similar pattern to OA rather than to CPPD [24,37]. Therefore, this pseudotumor can be developed by secondary OA-mediated biomechanical alteration rather compared to CPPD inflammation.
4. Treatment
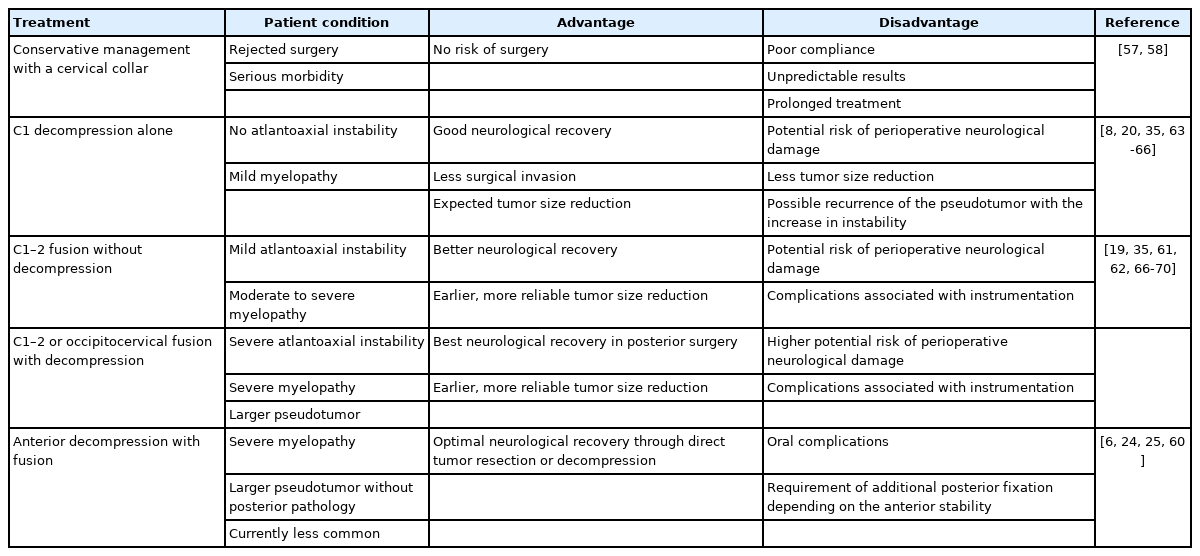
Conservative treatment by neck collar was adapted for patients who had the difficulty in undergoing surgery due to serious complications and/or who rejected surgery, as the size reduction in the retro-odontoid pseudotumor and recovery of symptoms have been reported [57,58]. Nevertheless, surgery is the first selection for patients suffering from pseudotumor-induced compression myelopathy [59]. Anterior decompression by transoral odontoid process and pseudotumor resection (combined with C1–2 posterior fusion) achieved good clinical and neurological outcomes [6,24,25,60]. More recent papers presented a remarkable pseudotumor size reduction even with the disappearance by C1–2 posterior fusion only [19,61,62]. Currently, posterior approach is the primary strategy based on pseudotumor pathologies of soft-tissue swelling and atlantoaxial instability. Then, spinal cord compression by the anterior pseudotumor or posterior C1 arch even after manual AAS reduction may require decompression with fusion. Moreover, C1 laminectomy alone is an acceptable option with good clinical results including pseudotumor size reduction, similar to decompression and fusion, and nonworsened AAS [63-65]. In a comparative study between the retro-odontoid pseudotumor between posterior fusion and decompression alone, recovery rate at the mean 54-month final follow-up did not differ but pseudotumor regression was more frequent in the fusion group (100% vs. 42%), resulting in the recommendation of fusion irrespective of atlantoaxial instability [66]. Further comparative studies regarding the need for stabilization and decompression are required. Reported advantages and disadvantages of treatment for the retro-odontoid pseudotumor are summarized in Table 3.
In our patient, posterior C1 laminectomy was selected because of mild myelopathy without marked AAS. Advantages of decompression alone are less invasive and avoidable from complications according to bone grafting and/or fusion surgery [67-70]. Disadvantages would be residual neck pain and nonexclusive future atlantoaxial instability and also pseudotumor progression [20]. Careful follow-up is necessary.
CONCLUSION
This is a case report of an elderly male patient suffering from neck pain and compression myelopathy caused by the retroodontoid pseudotumor without RA or AAS. Although prior articles described the atlantoodontoid pseudotumor with upper cervical spondylosis, most cases were associated not with primary OA but with secondary OA [32]. Based on periodontoid calcifications and ossifications, the pseudotumor would occur with chronic inflammatory CPPD crystal deposition. Subclinical CPPD progression around the atlantoaxial joint facilitates secondary OA development and retro-odontoid pannus formation, which can be enough severe to induce compression myelopathy in non-RA patients without AAS. The elderly population rapidly increases in the world; therefore, more careful attention around the craniocervical region should be paid to identify compression myelopathy associated with OA and CPPD.
Notes
The authors have nothing to disclose.