Mechanical Failure After Total En Bloc Spondylectomy and Salvage Surgery
Article information
Abstract
Objective
Total en bloc spondylectomy (TES) is a curative surgical method for spinal tumors. After resecting the 3 spinal columns, reconstruction is of paramount importance. We present cases of mechanical failure and suggest strategies for salvage surgery.
Methods
The medical records of 19 patients who underwent TES (9 for primary tumors and 10 for metastatic tumors) were retrospectively reviewed. Previously reported surgical techniques were used, and the surgical extent was 1 level in 16 patients and 2 levels in 3 patients. A titanium-based mesh-type interbody spacer filled with autologous and cadaveric bone was used for anterior support, and a pedicle screw/rod system was used for posterior support. Radiotherapy was performed in 11 patients (pre-TES, 5; post-TES, 6). They were followed up for 59 ± 38 months (range, 11–133 months).
Results
During follow-up, 8 of 9 primary tumor patients (89%) and 5 of 10 metastatic tumor patients (50%) survived (mean survival time, 124 ± 8 months vs. 51 ± 13 months; p=0.11). Mechanical failure occurred in 3 patients (33%) with primary tumors and 2 patients (20%) with metastatic tumors (p=0.63). The mechanical failure-free time was 94.4 ± 14 months (primary tumors, 95 ± 18 months; metastatic tumors, 68 ± 16 months; p=0.90). Revision surgery was performed in 4 of 5 patients, and bilateral broken rods were replaced with dual cobalt-chromium alloy rods. Repeated rod fractures occurred in 1 of 4 patients 2 years later, and the third operation (with multiple cobalt-chromium alloy rods) was successful for over 6 years.
Conclusion
Considering the difficulty of reoperation and patients’ suffering, preemptive use of a multiple-rod system may be advisable.
INTRODUCTION
Total en bloc spondylectomy (TES) is a curative surgical method for both primary and solitary metastatic tumors [1-3]. The concept of en bloc resection of spinal tumors is similar to that of marginal or wide resection for musculoskeletal tumors [1-3]. Weinstein, Boriani, and Biagini (the WBB system) and Tomita et al. suggested staging systems for the proper application of TES, which is a rewarding but challenging surgical technique with a local tumor control rate of more than 80%–90% for both primary and metastatic tumors [3-8]. When it is indicated, the 3 spinal columns are totally resected, and reconstruction of the vertebra and the durability of constructs are of paramount importance [1-3].
Initially, mechanical failure was reported in approximately 5% of patients after TES, but it rarely occurred after the introduction of titanium-based mesh-type vertebral spacers [1,3,9,10]. However, mechanical failure is a concerning issue for long-term survivors, especially for patients who have undergone radiotherapy and/or chemotherapy [3,11,12]. For long-term survivors, mechanical failure was reported in 27%–43% of patients [8,11,13].
Mechanical failure was not an uncommon complication, but it was not sufficiently addressed. The objective of this study was to present the incidence and case of mechanical failure and to suggest strategies for salvage surgery. Although the present study was based on experiences at a single institution, we intended to highlight the issue and share the experience of mechanical failure after TES.
MATERIALS AND METHODS
Nineteen consecutive patients (9 with primary tumors, 10 with metastatic tumors) who underwent TES for primary or metastatic tumors from July 2011 to April 2018 were retrospectively reviewed. TES was indicated for intracompartmental primary tumors or solitary intracompartmental metastatic tumors. Two patients underwent TES for a recurrent primary tumor, and the other patients underwent TES for their first surgical procedure. Two patients with recurrent tumors underwent intralesional resection and radiotherapy 1 year and 6 months before, respectively, and they were referred for recurrent primary tumors (giant cell tumor [GCT] and chondroblastoma). The primary tumors were 6 GCTs, 2 chondroblastomas, and 1 osteosarcoma. Among the patients with metastatic tumors, the primary organs were the kidney in 3 patients, the cervix in 2, the liver in 2, the lung in 2, and the thyroid in 1 (Table 1). This study was approved by the Institutional Review Board of Seoul National University College of Medicine/Seoul National University Hospital (2110-117-1263). The requirement for informed consent was waived because the data were deidentified and all methods were carried out in accordance with relevant guidelines and regulations.
1. Surgery and Perioperative Care
The surgical principles and techniques of TES were the same as those reported [1,2,4,6]. A posterior approach was used for 15 thoracic tumors, and a combined anterior and posterior approach was used for lumbar tumors. The surgical extent was 1 level in 16 patients and 2 levels in 3 patients. A titanium-based mesh-type interbody spacer (SynMesh system, Depuy Synthes, MA, USA) filled with autologous and cadaveric bone was used for anterior support, and a pedicle screw/rod system (Expedium, Depuy Synthes, Raynham, MA, USA; Legacy, Medtronics, Memphis, TN, USA) was used for posterior support. For 1-level TES, the fixation points were 2 levels above and below, and for 2-level TES, they were 3 levels above and below. In the first surgical procedure, a single titanium-based 5.5-mm diameter rod was used on each side. After fixation with cage and rod/screws, the posterior structure was reconstructed to reduce compression by soft tissue and formation of compressive scar tissue on the spinal cord. The cadaveric femur head was tailored to make rectangular bony plates approximately 7 mm thick, and those were connected with metal plates and screws to make a long single rectangular plate. It was attached to the intact cranial and caudal laminae with metal plates and screws for posterior bony fusion (Fig. 1A, B). The trajectory of the previous biopsy was resected from the entry at the skin, and a resection-free margin was pathologically confirmed. Patients were encouraged to ambulate from the day of surgery with a rigid orthosis and were discharged 6–7 days after surgery. Patients were instructed to wear the orthosis for 3 months. Patients were followed-up for 59± 38 months (range, 11–133 months).
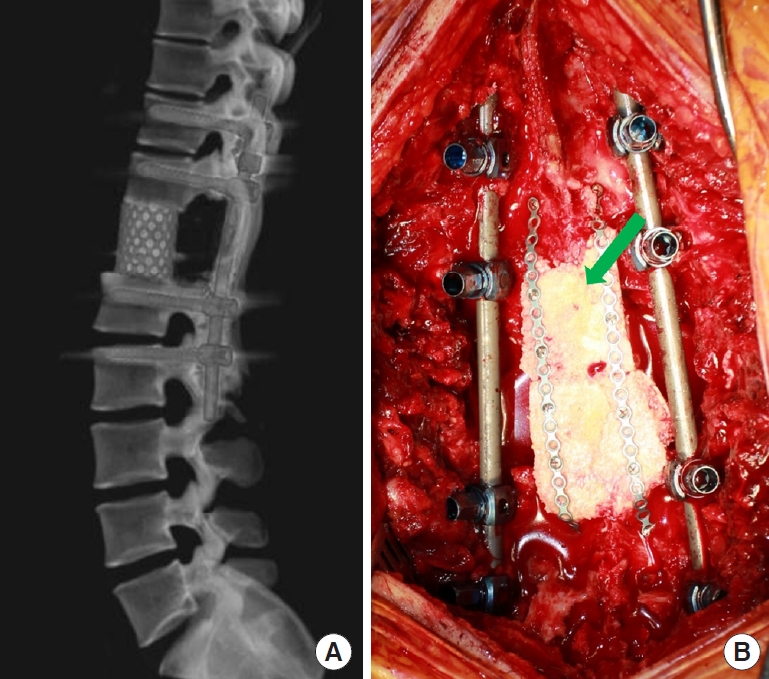
Reconstruction of 3 columns after total en bloc spondylectomy (TES). (A) After TES at T12, the anterior vertebral column is supported by a titanium-based mesh-type interbody cage, and the posterior column is supported by pedicle screws and a 5.5-mm rod on each side. (B) Cadaveric bone was tailored and fixed with a plate and screw system for bone fusion between intact laminas (arrow).
2. Analysis of Clinical and Radiological Outcomes
Patients were scheduled to visit the clinic at postoperative months 1, 3, 6, and 12 and yearly thereafter. At each visit, patients were asked to undergo a plain x-ray to check for mechanical failure, and a computed tomography (CT) scan was performed at 6 months to confirm bony fusion. If solid bony fusion was not visible, the CT scan was planned to be repeated 6 months later and yearly thereafter. For patients with metastatic tumors, the abovementioned schedule was not followed when the chest or abdominal CT scans taken by medical/radiation oncologists were available, and reconstructed coronal or sagittal scans were referenced to evaluate bony fusion. Bony bridging across the spondylectomy site was considered fusion. Asymptomatic mechanical failure was closely observed every 3 months with x-rays, while cases of symptomatic failure underwent reoperation.
Continuous variables are presented as the mean ± standard deviation, and noncontinuous values are presented as numbers and proportions. Categorical data were compared using the chi-square test, and quantitative data were compared using the Mann-Whitney U-test. The survival time, mechanical failure-free time, and time to bony fusion were compared between primary and metastatic tumors using Kaplan-Meier survival analysis and the log-rank test. All statistical analyses were performed using IBM SPSS Statistics ver. 23.0 (IBM Co., Armonk, NY, USA), and a p-value less than 0.05 was considered to indicate statistical significance.
RESULTS
All surgeries were finished without neurological complications. Two patients developed chylothorax, which was controlled with a chest tube and lipid-free diet for 1 week. Any other complications, such as surgical site infection, hematoma collection, or deep vein thrombosis, did not occur. The preoperative treatment was radiotherapy in 5 patients (2 with primary tumors and 3 with metastatic tumors) and conventional chemotherapy in 2 patients with metastatic tumors. Postoperative radiotherapy was performed for 6 patients (1 with a primary tumor and 5 with metastatic tumors) 3 weeks after TES because the tumor involved the bilateral pedicles (n=1) or there was a concern regarding microscopic tumor contamination despite the use of a thread saw to cut the lamina and pedicle (n=5). Two patients with metastatic cancer showed tumor recurrence at postoperative 6 months. One patient with a GCT experienced a malignant transformation of the GCT at postoperative months 69. During the follow-up period, 8 of 9 patients (88.9%) with primary tumors and 5 of 10 patients (50%) with metastatic tumors survived, with mean survival times of 124±8 months and 51±13 months, respectively (p=0.11). Solid bony fusion was achieved in 56% (5 of 9) of primary tumors at a mean of 43±9 months and 6 of 10 (60%) of metastatic tumors at a mean of 20±5 months. Time to fusion was not different between primary and metastatic tumors (p=0.19). A mechanical failure occurred in 3 patients (33%) with primary spinal tumors and 2 patients (20%) with metastatic spinal tumors (p=0.63) during the follow-up period. Fracture of the rod occurred in 3 patients with primary tumors and 1 patient with metastatic tumors. Subsidence of MESH cage occurred in 1 patient with metastatic tumor. A mechanical failure occurred in 27% (3 of 11) of patients with the bony union and 25% (2 of 8) of patients without bony union (p=0.91) (Table 2). The mean mechanical failure-free time was 95±18 months for primary tumors and 68±16 months for metastatic tumors (p=0.90), and the overall mechanical failure-free time was 94.4± 14 months (Fig. 2). The survival time and mechanical failure-free time were not significantly different from each other in either the primary tumor group or the metastatic tumor group (p=0.20 and p=0.35, respectively). Revision surgery was performed in 4 of 5 patients with fractured rods, and the bilateral rods were replaced with new dual cobalt-chromium alloy rods. One patient with subsidence of the cage did not undergo revision surgery due to poor general condition and systematic progression of cancer. Repeated rod fractures occurred in 1 of 4 patients 2 years after reoperation, and the third operation (with multiple cobalt-chromium alloy rods) was successful for more than 6 years.

Event-free time for survival and mechanical failure. The survival curves represent the survival time and mechanical failure-free time for both primary and metastatic tumors. Although statistical significance was not reached, the survival time (124 ± 8 months) was longer than the mechanical failure-free time (95 ± 18 months) in primary tumors.
The risk factors for mechanical failure were analyzed. A mechanical failure occurred more frequently in patients who received radiotherapy (4 of 11, 36%) than in patients who did not receive radiotherapy (1 of 7, 13%), but the difference was not statistically significant (p=0.24). Other factors, including fusion status and sex, did not show statistical significance.
1. Case
A 23-year-old man presented with back pain and was diagnosed with GCT at T6 and T7 (Fig. 3A, B). Based on the operation records, the tumor was totally removed based on a gross assessment, but the procedure was intralesional resection, and radiotherapy (6,000 cGy) was administered. A postoperative x-ray showed fixation from T4–8 without total vertebrectomy (Fig. 3C). Several lung masses were detected 9 months after the first surgical procedure; these masses were resected and pathologically confirmed as metastatic GCTs. Adjuvant therapy was not performed. Three additional small lung masses were detected 7 months later (16 months postoperatively). Eventually, the patient was referred to our institution. Positron emission tomography showed high uptake at the index surgery site (Fig. 3D) and a concordant tumor at T6. GCT was confirmed by needle biopsy at the thoracic spine. Considering the patient’s age and oncological outcomes, it was decided to perform revision TES and metastasectomy, followed by chemotherapy with denosumab in a multidisciplinary spinal oncology board meeting. To achieve a tumor-free margin, TES including the residual rib, the vertebral body of T6 and T7, pedicle, and transverse processes was planned (Fig. 3E). The operation took 10 hours, and 2-level TES with a tumor-free margin was performed after resection of the unilateral lamina and pedicle from the right side, where the tumor had not invaded (Fig. 3F). The vertebral column was reconstructed using a mesh-type titanium cage filled with autologous and cadaveric bone, pedicle screws, a rod system from T3 to T10, and a rectangular tailored cadaveric femoral head between laminae (Fig. 3G, H). Lung masses were resected 2 months later, and denosumab was used for 3 months. Although the tumor did not recur, unilateral rod fracture occurred 1.5 years after TES (Fig. 3I), and CT scan showed no solid bony union between the cage and T8 (Fig. 3J). Because patients did not complain of pain, the fracture was closely observed, but bilateral rod breakage occurred 6 months later (2 years after TES) (Fig. 3K). Because the ventral approach and additional anterior support seemed to be risky, the fractured rods were replaced with cobalt-chromium-based rods, and additional rods were applied (Fig. 3L). Images taken 1 year later (37 months after TES) showed no further rod breakage or cage subsidence at either the anterior or posterior spinal columns. The patient had no further mechanical problems until back pain reappeared at 69 months after TES, and radiation-induced osteosarcoma was diagnosed. Because the mass was extensive and not operable, chemotherapy was performed. Finally, the patient died 13 months later (72 months after TES).
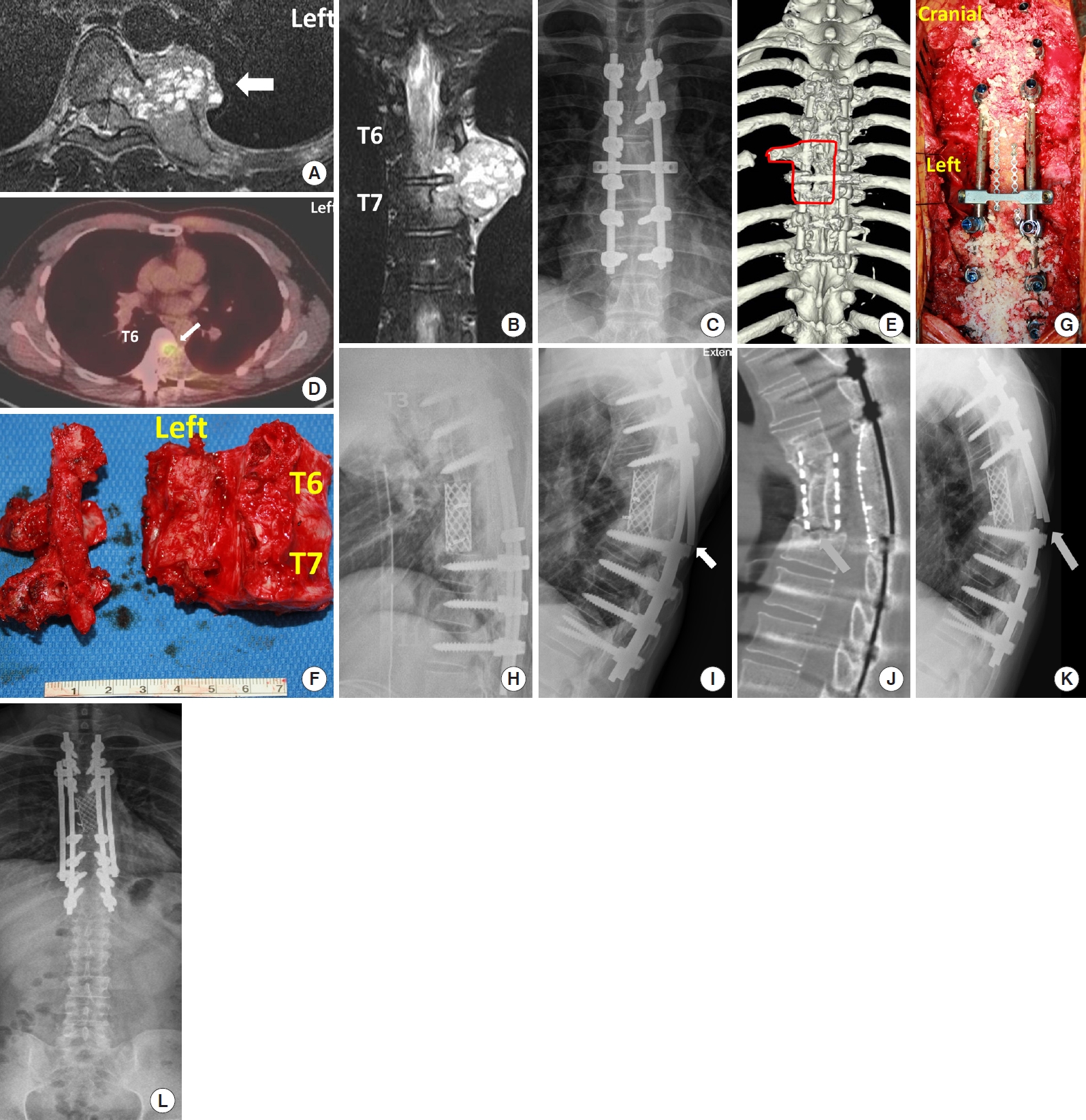
Total en bloc spondylectomy for recurrent giant cell tumor. T2-weighted magnetic resonance images show a mass across T6/7 in axial (A, arrow) and coronal (B) views. The tumor extended to the vertebral body, epidural space transverse process, and ribs. A postoperative x-ray shows instrumentation from T4 to T9, but the vertebral bodies are not resected (C). Positron emission tomography shows high uptake (D, arrow) in the left T6 vertebral body. Presurgical planning is marked on a 3-dimensional posterior (E) image with bold lines. (F) An intraoperative specimen shows a resected vertebra. The tumor was in the left vertebral body, pedicle, and transverse process, and those structures were removed in an en bloc fashion after resecting posterior spinal elements, as marked with a bold line (E). (G) An intraoperative photo shows posterior reconstruction with a pedicle/screw system and cadaveric bone bloc and chips. (H) A postoperative x-ray shows anterior column support with a mesh-type interbody space. (I) X-ray shows a unilateral rod fracture (arrow). (J) Sagittal computed tomography shows nonunion between the cage and T8 (arrow). (K) X-ray taken 6 months later showed a bilateral rod fracture (arrow). (L) The broken screws were replaced with cobalt-chromium alloy rods, and additional rods were applied.
DISCUSSION
1. TES and Mechanical Failure
Because of the proximity of the spinal cord, wide or marginal resection of spinal tumors is a challenging surgical option. Due to this issue, Weinstein, Boriani, and Biagini (the WBB system) and Tomita et al. suggested staging systems for the proper application of TES [4,6]. The overall local control rate was reported to be approximately 95%, and the 5-year survival rate was 67% for primary malignant spinal tumors and 100% for primary aggressive benign tumors [3-9]. Although TES was mostly applied for the initial surgery, TES may also be performed for recurrent primary spinal tumors, as the present study showed [14].
One of the major issues after TES is the reconstruction of the spinal column. Solid bony fusion has been established as the most important factor to prevent mechanical failure when the 3 spinal columns are destabilized [15-17]. Even in the first report of TES by Dr. Tomita et al. [1], a case of mechanical failure and a displaced pedicle screw were reported. Bony union of the construct can be a concern in long-term survivors, especially for patients requiring adjuvant radiation and/or chemotherapy [3,10]. Several options are available for interbody constructs, such as allografts, titanium-mesh cylindrical cages, polyetheretherketone cages, expandable cages, and autologous free vascularized grafts [3]. Bongers et al. [18] showed that an autologous free vascularized fibular graft was an applicable option for reconstruction after TES, and the complete union rate was 76%. However, revision surgery was necessary for 26% of patients due to mechanical failures, such as fracture of graft and implant failure. The strength of the vascularized fibular graft was sufficient for the cervicothoracic spine, but it was not strong enough to support the lumbar spine [18]. Of those options, titanium-based interbody cages filled with auto or allo-bone materials, and pedicle screw/rod systems have been commonly used for reconstruction [1,3,9,19-21]. Mechanical failure occurred in 7% of patients after multilevel TESs and reconstruction with cages filled with iliac crest and rib autografts and pedicle screw/rod systems [7]. Autografts are the graft material of choice, but allograft or synthetic graft materials are generally the most feasible options considering the morbidity of harvesting a large amount of autograft [3].
Nonetheless, several recent reports have presented results on mechanical failure after TES, such as screw loosening, screw back-out, cage breakage, screw fracture, and rod fracture; these studies identified rod fracture as the most common type of failure, and it often led to reoperation [8,11,12,22,23]. Shinmura et al. [8] showed that instrumentation failure occurred in 43% of patients at a mean interval of 32 months (range, 11–92 months). Li et al. [11] also showed that mechanical failure occurred in 27% (9 of 30) of patients at a mean interval of 32 months (range, 13–64 months). In accordance with their findings, this study also showed that mechanical failure occurred in 26% of patients at approximately 3 years. In the present study, all patients uniformly underwent TES with a titanium-based interbody spacer, a titanium-based pedicle screw, and a 5.5-mm rod system. As described above, the challenging case of 2-level TES after previous intralesional resection and radiotherapy eventually ended up needing additional surgery due to mechanical failure, not due to tumor recurrence. Although the high occurrence of mechanical failure may have been caused by an inappropriate selection of interbody grafts and posterior rod systems, the issue of mechanical failure is noteworthy.
The causes of rod fracture varied, and radiotherapy and surgery at the lumbar spine were common causes [8,11-13,18,22,23]. Although wide resection is the goal of TES, preoperative or postoperative radiotherapy and/or chemotherapy may be necessary for better oncologic outcomes. In such cases, preemptive measures may not be overtreatment considering the suffering of patients, the morbidity of revision surgery, and the overall cost of multiple operations [14,15]. As the present case showed, mechanical failure occurred after reoperation with a single rod on each side, and it was salvaged with a multiple-rod system. To prevent mechanical failure, solid bony union was important, but it was not very successful after TES and radiotherapy [8,11-13,19,20]. In addition, mechanical failure could occur despite solid bony fusion [21]. In this study, mechanical failure occurred in 27% of patients with bony fusion. The discrepancy between solid bony fusion and mechanical failure may imply the pitfalls of the current definition of bony fusion or insufficient strength of fused bone grafts to prevent mechanical failure from occurring [18]. These issues need to be verified in further studies.
When mechanical failure occurs, the surgical strategy may vary. Although adding strong anterior support with an anterior approach could be a solution, it has a high risk of approach-related complications. Instead, previous studies have suggested posterior revision surgery using multiple rods and an additional bone graft as an option [8,24-27]. A multiple-rod system has been suggested to be useful in deformity surgery, especially when 3 columns were resected to correct the deformity [15-17,28-32]. This suggestion is also applicable after TES [21-24]. Wei et al. [12] preemptively used multiple rods after TES and showed no implant-related complications during a mean follow-up of 31 months. Although cost was a concern, they addressed this issue by using multiple rods cut from a long rod, and it did not increase costs to the expected extent [12]. Preemptively using a multiple-rod system for patients expecting a long survival period may be a rewarding surgical strategy. Another unaddressed issue was the management of cases with unilateral rod fracture, as shown in the illustrated case. As the case showed, a tolerable time with a unilateral rod would be less than 6 months, and revision with a multiple-rod system is recommended in an urgent manner when a unilateral rod fracture occurs. However, this suggestion was based on anecdotal experience and could not be generalized. Further study is warranted to suggest a robust idea to address unilateral rod fracture. In our institution, we reconstructed the posterior spinal element after surgery for spinal pathology, including tumors. The purpose of reconstruction was to protect the spinal cord from compression by muscle or scar tissue. In addition, it facilitated revision surgery when the tumor recurred. In TES, the autologous lamina was resected together with the tumor, and allobone was used for reconstruction. Because we could not obtain a commercialized long rectangular allobone, we used a head of femur and carpentered it to make rectangular plates. Although bone fusion seemed to occur, the strength of the reconstructed posterior bone plate was not enough to support the spinal column, as shown in the illustrated case [21]. However, I should note that posterior reconstruction procedure was not an essential one for TES.
2. Limitations
Although this study underscored the possibility of mechanical complications after challenging TES, it has several limitations. First, this study included only a small number of patients treated in a single institution and may have been affected by selection bias, type I error, and type II error. Most importantly, the number of patients in this study was too small to draw any strong suggestions. Although the surgical procedures were in accordance with previous studies, the large difference in the rate of mechanical failure among several papers merits further attention. Second, although we recommended using a multiplerod system for patients expecting a long survival period, especially in revision surgery, this suggestion is not based on strong evidence [12]. Chemotherapy, radiosurgery, and proton-beam therapy have recently been suggested as primary or adjuvant treatment, but discussing those issues was beyond the scope of this paper [16,33,34]. A systematic analysis would provide a better idea of how to use limited medical resources efficiently. Finally, although we suggested using a multiple-rod system as a preemptive measure after TES, this study did not investigate how to schedule other factors related to mechanical failure, such as radiotherapy, chemotherapy, ambulation time, and return to work. Despite these limitations, this study made a meaningful contribution by sharing the experience of mechanical failure and proposing the use of preemptive measures and the use of a dual or multiple-rod system, especially for patients expecting a long survival duration.
CONCLUSION
The occurrence of mechanical failure after TES was a major cause of revision surgery. It could occur even after following a standard surgical technique: the use of anterior support and a pedicle screw/rod system. Considering the difficulty of reoperation and patients’ suffering, the preemptive use of a multiple-rod system may be advisable, especially for patients expecting a long survival duration. Further studies are required for the efficient use of limited resources.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This work was supported by the New Faculty Startup Fund from Seoul National University. This study was supported by grant from Seoul National University Hospital research fund (grant No. no. 04-2021-0540).
Author Contribution
Conceptualization: CKC, SBP, SHY, CHL, JR, KK, CHK; Data curation: SWK, YI, WTY, CHK; Formal analysis: CHK; Funding acquisition: CHK; Methodology: CHK; Project administration: CHK; Visualization: CHK; Writing - original draft: SWK, CHK; Writing - review & editing: CKC, YI Won, WTY, SBP, SHY, CHL, JR, KK, CHK.