Meta-Analysis on the Effect of Hypothermia in Acute Spinal Cord Injury
Article information
Abstract
Objective
Acute spinal cord injury (SCI) can result in debilitating motor, sensory, and autonomic dysfunction. As a treatment option, therapeutic hypothermia has been researched to inadequate pharmaceutical treatment, except for methylprednisolone. In this article, we systematically meta-analyzed to clarify the effect of hypothermia in acute SCI on neurological outcomes.
Methods
The PubMed, Embase, Web of Science, and Cochrane clinical trial databases were systematically searched until June 30, 2022. The proportion of cases with improved neurological status after hypothermia in acute SCI were pooled with a random-effects model. Subgroup analyses for the method of hypothermia and injury level were conducted.
Results
Eight studies with a total of 103 patients were included. Hypothermia in acute SCI improved neurological function by 55.8% (95% confidence interval [CI]: 39.4%–72.1%). The subgroup analysis revealed that the pooled proportion of cases showing neurological improvement was higher with systemic hypothermia (70.9%) (95% CI, 14.9%–100%) than with local hypothermia (52.5%) (95% CI, 40.4%–64.5%), although the subgroup difference was not statistically significant (p=0.53). Another subgroup analysis revealed that the proportion of cases with neurological improvement did not differ statistically between the cervical spine (61.4%) (95% CI, 42.2%–80.6%) and thoracic spine injury groups (59.4%) (95% CI, 34.8%–84.0%) (p=0.90).
Conclusion
This meta-analysis identified that more than 50% of patients showed neurological improvement after hypothermia following acute SCI in general. A multicenter, randomized, double-blind study with larger sample size is necessary to validate the findings further.
INTRODUCTION
Acute spinal cord injury (SCI) is a potentially debilitating neurological disorder that can result in partial or total loss of motor, sensory, and autonomic function. In addition to a poorer quality of life for the patients, SCI also has negative socioeconomic effects on their families and on society [1]. There are currently no effective therapeutic approaches for the treatment of acute SCI, although various potential drugs and treatments are being researched. Surgical treatment with adequate decompression and fixation has the possibility of functional recovery of the neural structures. However, the outcomes of additional therapeutic pharmacological treatment are unsatisfactory, except with methylprednisolone; nevertheless, the evidence in support of methylprednisolone is inconclusive.
Recently, therapeutic hypothermia has emerged as one of the most promising treatments. It has been studied experimentally and clinically for a variety of disorders, including cerebral aneurysm, traumatic brain injury (TBI), aortic arch aneurysm, cardiac arrest, and acute SCI. The purpose of therapeutic hypothermia is to decrease the metabolic rate for oxygen, hence lowering lactate levels and other byproducts of anaerobic metabolism and minimizing cellular acidosis. It is hypothesized that therapeutic hypothermia can inhibit several metabolic pathways, inflammatory responses, and apoptotic processes that occur following an ischemia cascade [2].
Studies on animals have consistently demonstrated its effectiveness in functional recovery after SCI [3-5]. Furthermore, clinical trials on therapeutic hypothermia have shown positive outcomes [6,7]. It can be induced as local and systemic hypothermia, and each has its pros and cons. While systemic hypothermia can lead to rapid and persistent hypothermic status through surface cooling, endovascular heat exchange catheters, or cold intravenous infusions, local hypothermia has the advantage of direct irrigation to the intradural or extradural space with cold saline without the risk of systemic consequences [8]. However, human investigations on the effects of hypothermia have yielded inconsistent results, depending on the clinical circumstances and methods of hypothermia, although animal studies have showed usually positive results.
The purpose of present meta-analysis is to assess the effect of hypothermia on acute SCI. We specifically evaluated whether the influence of hypothermia on neurological outcome varies according to the method of hypothermia and the level of injury.
MATERIALS AND METHODS
This research followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines [9]. We conducted a meta-analysis and systematic review of clinical studies on therapeutic hypothermia for the treatment of acute SCI.
1. Search Strategy
We searched records in PubMed, Embase, Web of Science, and Cochrane clinical trial since their inception through June 30, 2022. The following search queries included SCI and hypothermia synonyms and related terms: (spine OR “spinal cord” OR vertebral) AND (injury OR injuries OR injured OR trauma OR traumatic) AND (hypothermia OR hypothermic OR cooling OR cooled). Furthermore, the reference lists of the indicated articles were searched for further relevant studies. Two reviewers worked separately on the literature search and selection. The disagreements between the reviewers were resolved after the discussion.
2. Study Selection
Patients, interventions, outcomes, and study design (PICOS) criteria were used to choose inclusion for the study. PICOS criteria in this article are as follows: (1) “patients” with acute SCI, (2) hypothermia treatment as the “intervention,” (3) no relevant “comparator,” (4) neurological outcomes as the “outcome,” and (5) original articles for “study design.” The following were the exclusion criteria: (1) nonoriginal articles, (2) not in the field of interest, (3) not written in English, (4) overlap in study population, (5) the number of populations is less than five, and (6) insufficient information on study outcomes. When research populations are overlapped, we included the study that had the most extensive data.
3. Extraction of Data and Quality Assessment
Characteristics of included studies were extracted using a standardized form, including author, publication year, country, the number of patients, age, injury level, initial neurological status of American Spinal Injury Association (ASIA) A or complete deficit, method of hypothermia, route of hypothermia, time from injury to hypothermia, duration of hypothermia, target temperature, number of patients with other treatments such as surgery or steroid, follow-up period, and final neurological outcome. The methodologic quality of the included studies was assessed using the Risk of Bias in Nonrandomized Studies of Interventions (ROBINS-I) tool [10]. This tool evaluates 7 assessment domains, which include bias due to confounder, subject selection, classification of interventions, deviations in interventions, missing data, biased measurements, and biased reporting. The potential for bias in each individual domain is rated as either low, moderate, serious, or critical, respectively. After combining the results of these domains, an overall risk of bias judgment was determined to be either low, moderate, serious, or critical. Two reviewers separately extracted the data and evaluated its quality; any discrepancies were resolved through discussion.
4. Statistical Analysis
To analyze the relationship regarding pooled outcomes, we calculated the proportion with 95% confidence interval (CI) for studies. The proportions were pooled using meta-analysis utilizing the random-effects model (DerSimonian-Laird technique) with logit transformation for computing weights. The Cochran Q test and the Higgins I2 test were used to assess heterogeneity between the outcomes of the various studies. To assess the presence of publication bias, funnel plots and Egger tests were used [11]. Stratification by the method of hypothermia and injury level was done for subgroup analyses. Every test was 2-sided, and a p-value of 0.05 or less was regarded as statistically significant. R software was used to conduct the statistical analysis (ver. 4.0.4; The R Foundation for Statistical Computing, Vienna, Austria).
RESULTS
1. Literature Search
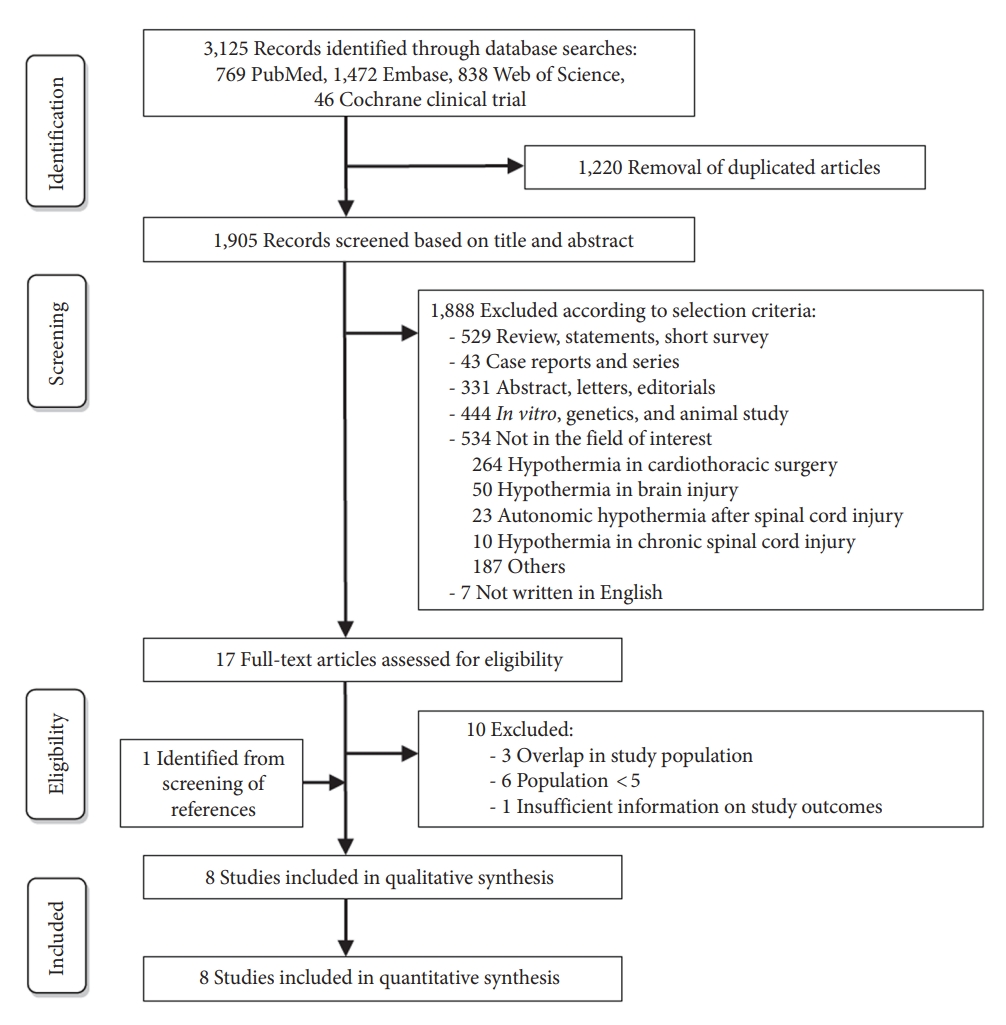
A preliminary literature search utilizing the subject headings revealed 769 articles from PubMed, 1,472 studies from Embase, 838 studies from Web of Science, and 46 studies from the Cochrane clinical trial. There were 1,220 duplicates among these 3,125 studies, thus they were eliminated. After evaluating the remaining 1,905 titles and abstracts, 1,888 were deemed ineligible because they did not match the selection criteria. Ten of the remaining seventeen studies were removed based on the following criteria (Supplementary Table 1): overlap in study populations (n=3), the number of populations was less than five (n=6), and insufficient information on study outcomes (n=1). One paper was added during a manual screening of references. Finally, a total of 8 studies were included in the meta-analysis [6,7,12-17]. Fig. 1 presents the detailed selection process.
2. Study Characteristics
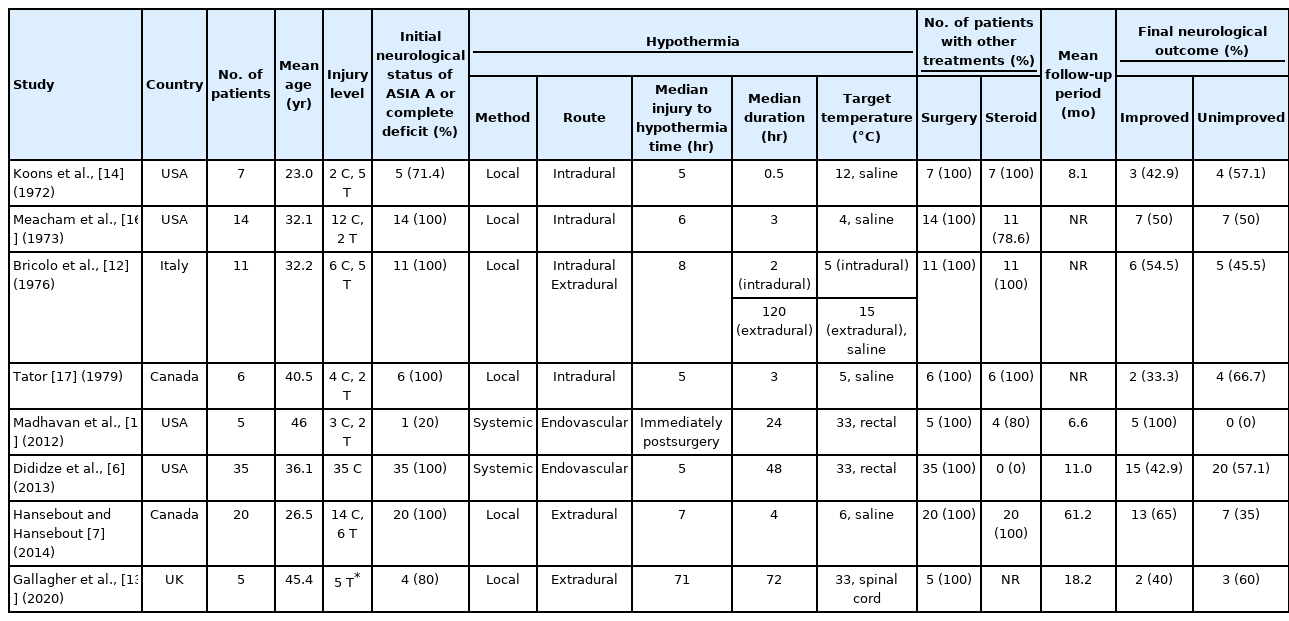
Table 1 summarizes the baseline characteristics of the included studies. The included studies were all retrospective. The studies included a total of 103 patients who were provided hypothermia treatment for SCI. Among the 8 articles included, 6 studies used local hypothermia for 63 patients, and 2 studies used systemic hypothermia for 40 patients.
3. Quality Assessment
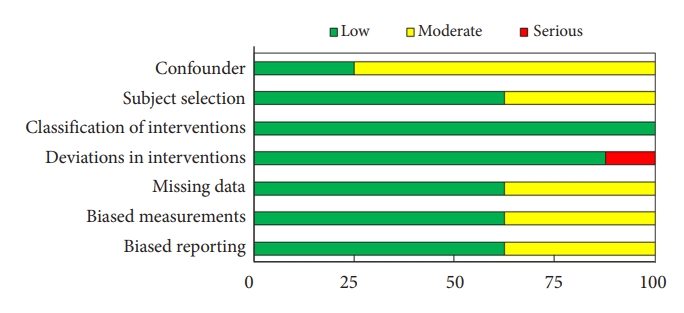
The overall quality assessment using the 7 domains of the ROBINS-I tool is shown in Fig. 2. Most domains were assessed as a low to moderate risk of overall bias. However, 1 study (12.5%) was evaluated as a serious risk of bias as 2 methods of hypothermia were used [12]. Two studies (25.0%) were evaluated as having a low risk of overall bias, and 5 studies (62.5%) were judged as having a moderate risk of overall bias. The detailed results of the quality assessment of each study are presented in Supplementary Table 2.
4. Neurological Status According to the Method of Hypothermia
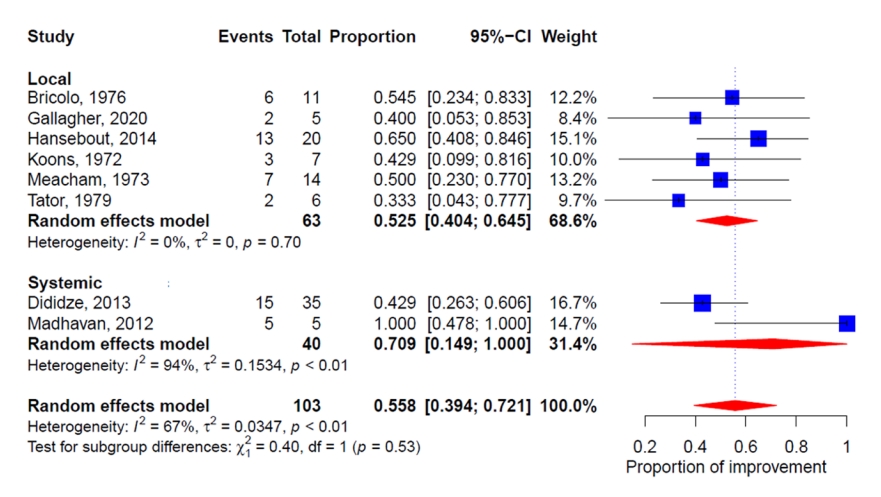
All 8 studies investigated the neurological status after therapeutic hypothermia in patients with SCI. The changes in neurological status after hypothermia in all studies were stratified by the method of hypothermia and are illustrated in Fig. 3. The proportion of patients with neurological improvement across all 8 studies was 55.8%. (95% CI, 39.4%–72.1%). Higgins I2 statistics revealed significant heterogeneity (I2=67%). The subgroup analysis revealed that the proportion of systemic hypothermia patients with neurological improvement was greater than that of the local hypothermia group. (proportion, 70.9%; 95% CI, 14.9%–100.0% and proportion, 52.5%; 95% CI, 40.4%–64.5%, respectively); however, the difference between the groups was not statistically significant (p=0.53). The funnel plot and Egger test showed no publication bias (Supplementary Fig. 1; Intercept=-0.34, p=0.48).
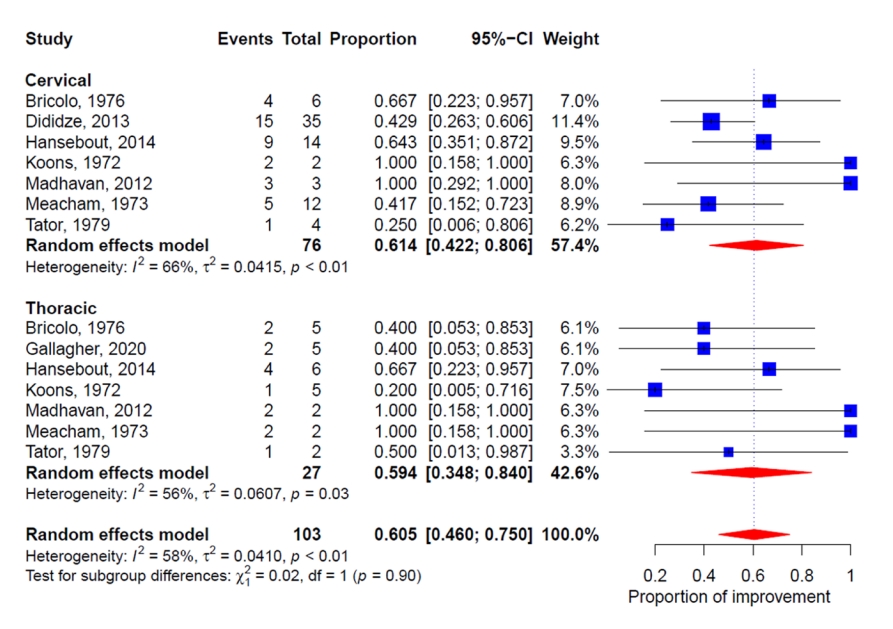
5. Neurological Status According to the Injury Level
All 8 studies demonstrated the neurological status according to the injury level. The changes in neurological status after hypothermia in the included studies based on the injury level are illustrated in Fig. 4. Higgins I2 statistics revealed significant heterogeneity (I2=58%). The subgroup analysis suggested that the proportion of patients with neurological improvement was not statistically different between groups with cervical and thoracic spine injuries (p=0.90) (proportion, 61.4%; 95% CI, 42.2%–80.6% and proportion, 59.4%; 95% CI, 34.8%–84.0%, respectively). The funnel plot and Egger test showed no publication bias (Supplementary Fig. 2; Intercept=-0.51, p=0.18).
DISCUSSION
Acute SCI is a serious neurological event that can have adverse effects on health, finances, relationships, and quality of life. Many studies and guidelines have been researched for the treatment of acute SCI [18,19]. However, there are no effective treatments to prevent the deleterious consequences of acute SCI; the evidence regarding the efficacy of methylprednisolone is inconclusive, and it is recommended only for specific indications [20]. In this meta-analysis, we evaluated the impact of therapeutic hypothermia on the outcomes of patients with acute SCI. The pooled proportion of patients in whom hypothermia for acute SCI led to neurological improvement was 55.8%. Analysis of the outcomes based on the method of hypothermia showed that the pooled proportion of patients with neurological improvement was 52.5% with local hypothermia and 70.9% with systemic hypothermia, respectively (p=0.53). Furthermore, an analysis based on the levels of injury showed that the pooled proportion of patients with neurological improvement was 61.4% in the cervical spine injuries and 59.4% in the thoracic spine injuries group (p=0.90). These findings suggest that hypothermia for acute SCI showed an improvement in neurological status in more than 50% of the patients in general.
Although the result of an experimental study varies depending on the protocol of the study, hypothermia for acute SCI has shown generally beneficial effects in experimental animal models [8,21-25]. Ok et al. [21] reported that both local (28°C for 48 hours) and systemic hypothermia (32°C for 48 hours) had neuroprotective effects after acute SCI in rats, and systemic hypothermia showed a higher neuroprotective effect due to antiapoptotic and anti-inflammatory effects. In another study on local hypothermia, Casas et al. [26] found no significant additive therapeutic effect of local hypothermia (24°C–35°C) in terms of locomotor outcomes or tissue preservation in rats with moderate thoracic spinal cord contusions. In contrast, systemic hypothermia has shown a consistently beneficial effect on neurological function. Kao et al. [23] revealed that moderate systemic hypothermia (33°C for 2 hours) resulted in lower levels of apoptosis, infarction volume, activated inflammation, and hind limb locomotor dysfunction in a rat SCI contusion model. Moreover, Maybhate et al. [24] showed that moderate systemic hypothermia (32°C for 2 hours) produced improvements in somatosensory evoked potentials and motor function in a rat SCI model.
Local hypothermia has been widely performed in acute SCI. Of the 8 studies included in our analysis, 6 used local hypothermia. There were 2 common methods to achieve local hypothermia: intradural irrigation after durotomy or extradural saline cooling. Intradural irrigation was performed in 4 studies for 0.5 to 3 hours with saline at 4°C to 12°C [12,14,16,17]. Intradural irrigation was performed for a shorter duration with a lower temperature of saline compared to extradural cooling. This method was used only until the 1970s as intradural irrigation causes herniation of the injured spinal cord through the durotomy incisions [27]. Extradural saline cooling was performed in 3 studies for 4 to 120 hours with saline at 6°C to 33°C [7,12,13]. Recent studies used cooling of the extradural space method, which achieves constant temperature by placing the heat exchanger tubing system in the epidural space.
Before the 2000s, systemic hypothermia was used in the treatment of some conditions such as cerebral aneurysm, TBI, or aortic arch aneurysm, following its use for cardiac arrest in the 1950s [28-31]. In early 2010, clinical studies for acute SCI were published from the University of Miami group [6,32]. They used systemic endovascular hypothermia for 48 hours with a target rectal temperature of 33°C. Dididze et al. [6] reported the effect of systemic hypothermia after SCI as 42.9%. After obtaining promising results from their retrospective data, they conducted prospective research and incorporated their findings in this paper [6,32,33]. However, as they did not utilize steroids, it is difficult to directly compare the results with those of previous SCI trials that did use steroids. In addition, Madhavan et al. [15] used systemic hypothermia for iatrogenic SCI after surgery and reported its efficacy as 100% effect. When there was a traumatic event and loss of intraoperative evoked potential during the surgery, which resulted in neurological deterioration, they promptly began systemic hypothermia following the operation. The immediate response to the neurological alteration and previously performed surgery may have contributed to the superior results. Because the time from injury to hypothermia was substantially shorter than in other research, and surgical decompression was accomplished during the initial surgery, recovery may have been reached in all patients. In addition, the initial neurological status of ASIA A patients before hypothermia was present in only one patient (20%), but in another research, this ranged from 71.4% to 100%. As incompletely injured spinal cord has functional reserve for recovery, there might be more opportunity to recover after hypothermia. Although previous studies on systemic hypothermia usually used surface cooling with water-circulating blankets above and below the patient or cold intravenous fluid infusion [34,35], recent studies used endovascular cooling, which is superior to other techniques for the maintenance of systemic hypothermia [36]. Endovascular cooling entails percutaneously inserting a catheter into the inferior vena cava, and saline chilled to 4°C to 5°C is pumped into the catheter balloons. The catheter cools the blood and has a temperature probe for blood temperature monitoring.
Existing research indicates that hypothermia is not an innocuous treatment. Complications of hypothermia could include pulmonary, cardiac, and thromboembolic events. These also occur or are aggravated due to SCI itself [37]. The complications might vary based on the methods of hypothermia. Local hypothermia involves risks related to the techniques used to achieve local cooling. Gallagher et al. [13] used an epidural cooling catheter to induce local hypothermia, following which a surgical site infection occurred in 3 of their 5 patients. The trial was prematurely terminated due to the surgical site infection. Deep systemic hypothermia (24°C–30°C), performed previously, had complications such as cardiac arrhythmias, hypotension, coagulopathies, systemic infections, and electrolyte abnormalities [27]. However, modest systemic hypothermia (32°C–34°C) provides the benefits of hypothermia without additional significant morbidity associated with deep systemic hypothermia [32]. Furthermore, a recent prospective multicenter study showed that modest systemic hypothermia was not associated with an increased risk of complications after SCI [38]. A clinical trial on systemic hypothermia (33°C for 48 hours) following acute SCI (≤ 24 hours) is currently underway [38].
This meta-analysis has some limitations. First, the number of included studies was few, and the number of included patients was small. However, this may be the first meta-analysis to offer a broad overview of this subject. Second, every study that was included was retrospective study. To confirm the outcomes of our meta-analysis, prospective investigations may be required. Third, the neurological change was assessed as only improved or not, rather than the change of specific grades. This may affect the results of the study, as the neurological improvement can be affected by the initial neurological status. Fourth, the follow-up period varies between studies. As the neurological recovery could be acquired in long term follow-up, the difference of follow-up period could affect the evaluation of the neurological status. Fifth, there was no standardization among the included studies about the basic characteristics of hypothermia, including method of hypothermia, route of hypothermia, time from injury to hypothermia, duration of hypothermia, and target temperature. Therefore, caution is necessary while interpreting our pooled estimates. Lastly, combined treatments other than hypothermia were different between the studies. The use of steroid and the surgical intervention were different between the studies.
CONCLUSION
This present meta-analysis and systematic review found that more than half the patients (55.8%) showed neurological improvement after hypothermia following acute SCI in general. In recent times, intradural local hypothermia is no longer performed, and extradural local hypothermia has shown clinical efficacy. In addition, modest systemic hypothermia using the endovascular cooling technique is currently used without an increased risk of complications. Although systemic hypothermia showed higher neurological improvement rates (70.9%) compared to local hypothermia (52.5%), there was no statistical difference, which might be due to the small number of patients included in the study. A multicenter, randomized, double-blind study with a significantly larger number of patients and a standardized defined methodology is necessary.
SUPPLEMENTARY MATERIALS
Supplementary Materials: Supplementary Tables 1, 2 and Figs. 1, 2 can be found via https://doi.org/10.14245/ns.2244444.222.
Studies excluded at the full-text articles assessment
Risk of bias using the Cochrane ROBINS-I Tool
Funnel plot of the subgroup analysis by the method of hypothermia.
Funnel plot of the subgroup analysis by the injury level.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contribution
Conceptualization: HKS, JHP, SWR, SRJ; Data curation: HKS; Formal analysis: HKS; Methodology: HKS, JHP, SWh, SRJ; Visualization: HKS; Writing - original draft: HKS, SRJ; Writing - review & editing: HKS, SRJ