Therapeutic Approaches Targeting Vascular Repair After Experimental Spinal Cord Injury: A Systematic Review of the Literature
Article information
Abstract
Traumatic spinal cord injury (SCI) disrupts the spinal cord vasculature resulting in ischemia, amplification of the secondary injury cascade and exacerbation of neural tissue loss. Restoring functional integrity of the microvasculature to prevent neural loss and to promote neural repair is an important challenge and opportunity in SCI research. Herein, we summarize the course of vascular injury and repair following SCI and give a comprehensive overview of current experimental therapeutic approaches targeting spinal cord microvasculature to diminish ischemia and thereby facilitate neural repair and regeneration. A systematic review of the published literature on therapeutic approaches to promote vascular repair after experimental SCI was performed using PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) standards. The MEDLINE databases PubMed, Embase, and OVID MEDLINE were searched using the keywords “spinal cord injury,” “angiogenesis,” “angiogenesis inducing agents,” “tissue engineering,” and “rodent subjects.” A total of 111 studies were identified through the search. Five main therapeutic approaches to diminish hypoxia-ischemia and promote vascular repair were identified as (1) the application of angiogenic factors, (2) genetic engineering, (3) physical stimulation, (4) cell transplantation, and (5) biomaterials carrying various factor delivery. There are different therapeutic approaches with the potential to diminish hypoxia-ischemia and promote vascular repair after experimental SCI. Of note, combinatorial approaches using implanted biomaterials and angiogenic factor delivery appear promising for clinical translation.
INTRODUCTION
Traumatic spinal cord injury (SCI) is one of the leading causes of disability and a major burden for healthcare systems. Current therapeutic strategies are limited to acute medical and surgical management and rehabilitation, as there are no therapies available to promote spinal cord regeneration [1].
SCI results from a primary mechanical injury which is amplified by a secondary injury cascade to result in loss or disruption of neural elements and vascular structures. The traumatic vascular disruption in the injured spinal cord plays a key role in modulating secondary injury development by promoting local blood-spinal cord barrier (BSCB) breakdown, inflammation, and neuronal cell death as well as creating a hypoxic-ischemic environment which can spread to initially intact regions [1]. The characterization of endogenous vascular regeneration following SCI-induced vascular injury has been the goal of some recent experimental studies, conducted mostly in rodents. Results show that vascular regeneration occurs spontaneously after SCI, following a definite time schedule – this regeneration process is however deficient and, due to an increased BSCB-permeability, even potentially detrimental to preserved neural tissue [2-6]. But even if this new vasculature is only partially functional, it provides guidance for regenerating axons and is thereby crucial for neural regeneration [7,8]. To find a way to improve vascular repair after SCI and restore a functional blood supply, thereby supporting neural regeneration, is one of the major challenges in experimental SCI research.
A multitude of experimental therapeutic approaches have been applied to restore functional vascularization, consisting of (1) the application of angiogenic factors, (2) genetic engineering therapy, (3) physical stimulation, (4) cell transplantation, and (5) biomaterials carrying various factor delivery [9-11].
The aim of this systematic review is to rigorously summarize and evaluate the present state of research in this emerging field. We first outline a focused overview of SCI pathophysiology with an emphasis on vascular injury and endogenous vascular repair and then shift to an examination of current experimental therapeutic approaches to promote vascular repair with the prospect of future translation of innovations into clinical patient care.
MATERIALS AND METHODS
1. Approach to the Systematic Literature Review
We performed a systematic, qualitative review of the literature, by screening the established MEDLINE databases PubMed, Embase, and OVID MEDLINE. The main search terms included “spinal cord injury,” angiogenesis,” “angiogenesis inducing agents,” and “tissue engineering,” in combination with “rodent subjects.” The search included no time limit. Articles in English were included, meeting the following criteria: experimental research, original studies, rodent subjects, therapeutic intervention, and focus on vascular injury and repair. The systematic literature search was conducted by one researcher from June 2021 to July 2021 and from May 2022 to June 2022. Data analysis was conducted according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines. A total of 614 articles were selected for review, of which 475 were excluded after screening as a result of not meeting the inclusion criteria. An additional 30 articles were excluded due to not meeting the inclusion criteria after full-text assessment for eligibility. For qualitative synthesis 111 articles were assessed in detail and included in this publication (Fig. 1).
2. Relevant Experimental Therapeutic Approaches According to the Literature Search
According to the systematic literature search, 5 main categories of therapeutic approaches to promote vascular repair in experimental SCI research were identified: (1) angiogenic factors, (2) genetic engineering, (3) physical stimulation, (4) cell transplantation, and (5) biomaterials carrying various factor delivery (Table 1).
RESULTS
1. PART 1: Pathophysiology of Traumatic SCI With Emphasis on Vascular Injury And Repair
1) Pathophysiology and its progression following SCI
SCI is characterized as a sudden mechanical force impacting the spinal cord, e.g., following a fall, vehicle accident, or sportsrelated injury. It is commonly comprised of both contusion and compression injury causing direct cell death, axonal shearing, and disruption of the microvasculature with traumatic bleeding. This primary injury is not accessible to treatment [1,12] and is immediately followed by a sustained secondary injury cascade [13]. This secondary injury spreads to formerly unaffected regions, leading to further spinal cord damage with the consequences of demyelination and axonal dieback [2,3,14]. The developing secondary injury can be divided into acute (first 48 hours), subacute (48 hours–14 days), intermediate (14 days to 6 months) and chronic phases (> 6 months) [2,12] (Fig. 2).
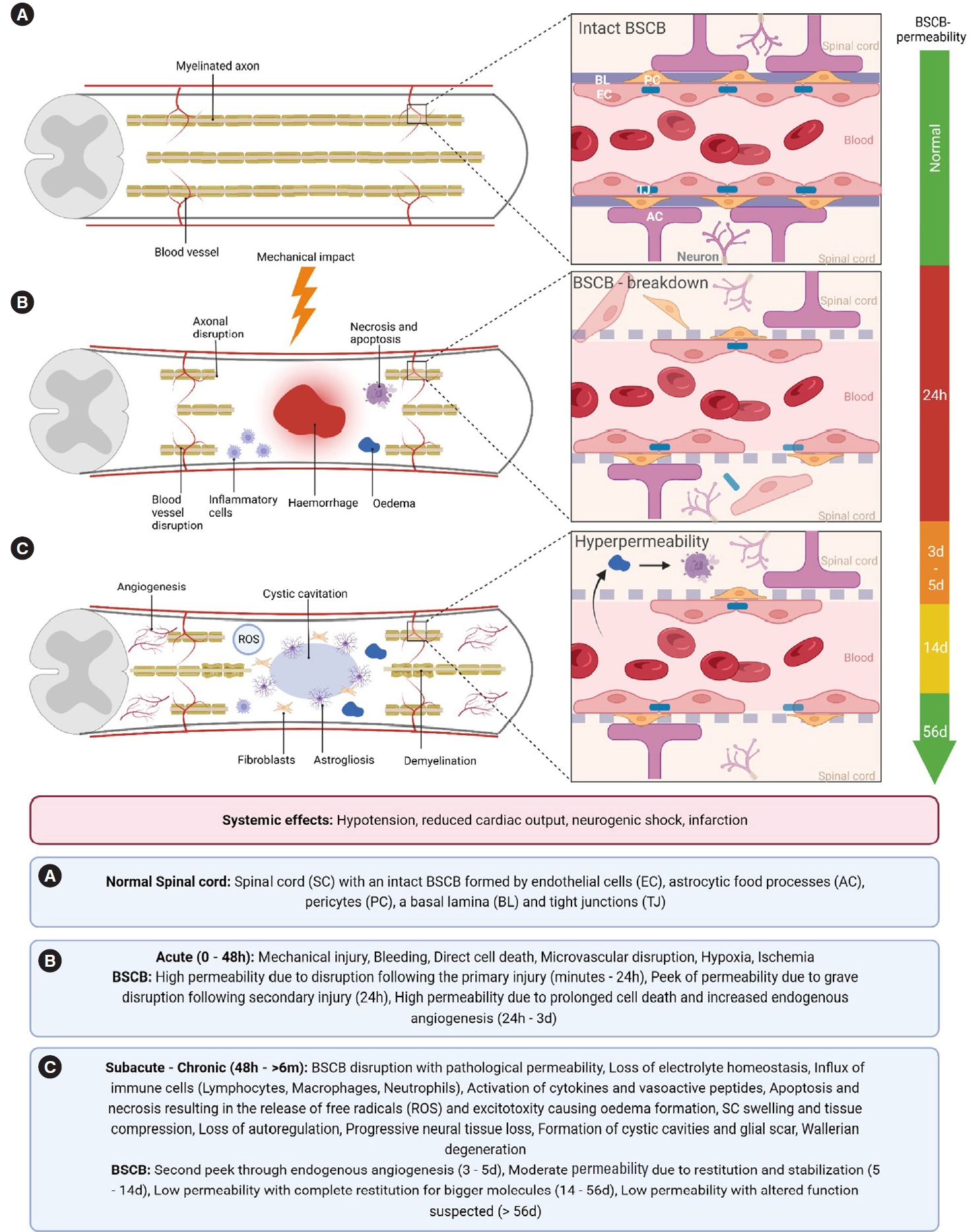
(A-C) Pathophysiology of spinal cord injury and the neurovascular unit in health and its disruption. SC, spinal cord; BSCB, blood – spinal cord barrier; EC, endothelial cells; AC, astrocytic foot processes; PC, pericyte; BL, basal lamina; TJ, tight junction; ROS, reactive oxygen species; h, hours; d, days; w, weeks; m, months.
2) Time course of BSCB disruption and restitution following SCI
The BSCB protects the spinal cord, as part of the central nervous system (CNS), from the periphery and maintains a homeostatic environment. The BSCB, formed by endothelial cells (ECs), astrocytic foot processes and pericytes as well as a basal lamina together with adjoining neurons comprise the neurovascular unit (NVU) (Fig. 2A). The unique cell-cell-contacts between ECs containing CNS-specific tight junctions (TJs) provide an immune privilege to the spinal cord compared to the periphery, restricting the passage of neurotoxic molecules and inflammatory cells [2,15].
Following the primary injury, a localized direct disruption of the NVU damages the microvasculature directly surrounding the injury site: Disruption of the protective cell-cell-contacts leads to altered cell permeabilization and consequent oedema formation, causing increased apoptotic and necrotic cell death [14,16,17] (Fig. 2B). These permeability changes not only lead to electrolyte influx and oedema, but to an influx of inflammatory cells (lymphocytes, macrophages, and neutrophils), as well as cytokines (tumor necrosis factor-α, interleukin [IL]-1β, IL-5) and vasoactive peptides. This immune response, commencing within the first minutes postinjury, leads to further oedema formation and thus tissue compression, resulting in further injury development spreading farther from the primary injury site [2,15,18]. These events further destabilize the BSCB and lead to a release of reactive oxygen species, resulting in the dysfunction of the NVU in formerly unharmed regions [2,3,14]. Correlating with these mechanisms, the first peak in BSCB-permeability in the penumbra zone is reached 24-hour postinjury, followed by a smaller peak after 3–5 days [4,5].
Most of the BSCB function is believed to be reinstated after 14 days [4,19] (Fig. 2C). However, experimental studies show that the permeability for small molecules is increased up to the chronic phase of SCI, for as long as 56-day postinjury [6].
3) Endogenous revascularization after SCI
The major form of vascular regeneration in the spinal cord is known to be angiogenesis, with a smaller part displayed by postnatal vasculogenesis. Angiogenesis is defined by the formation of blood vessels out of pre-existing vessels, whilst vasculogenesis describes the formation of new vessels [20]. The hypoxic milieu after SCI results in the release of angiogenic factors from the injury site. Endogenous revascularization following SCI is expected to occur during the first week postinjury, and to disappear shortly afterwards [21-24]. Importantly, the second, smaller peak in BSCB-permeability correlates with the peak of endogenous revascularization after SCI. As new vessels form due to angiogenesis or vasculogenesis, this young vasculature does not necessarily possess a functional BSCB. As some parts of this newly formed vasculature gain a functional barrier and promote a functional NVU during its genesis, other parts never do [4,15]. This results in the increased extravasation of molecules and fluids causing oedematous swelling, inflammation, and necrotic and apoptotic cell death and thus exacerbating the secondary injury [2,3,5,25] (Fig. 2B).
4) Regenerative potential facilitated by vascular repair after SCI
As the spinal cord remains in a state of chronic hypoxia and ischemia postinjury, reinstatement of a steady tissue perfusion would be desirable. This was shown by previous research indicating that neovascularization is crucial for axon regeneration, as these were shown to grow along blood vessels. This suggests that the developing vasculature guides axonal sprouting after injury [7,8]. However, the increased permeability for molecules otherwise excluded from the selective CNS environment might provide a unique chance for therapeutic intervention, with a potential window of opportunity displayed over the first days postinjury [4,26].
To address this multifaceted pathophysiology, several aspects of SCI must be taken into account to allow for tissue regeneration: Inhibition of the inflammatory response and sufficient perfusion might limit the secondary injury, while prevention of the glial scar formation could enable neural regeneration. Therefore, combinatorial approaches accounting for these mechanisms together in the adequate timespan might be promising for spinal cord repair.
2. PART 2: Experimental Therapeutic Approaches To Promote Vascular Repair
According to the systematic literature search, several experimental therapeutic approaches promising to diminish hypoxia and improve spinal cord microvasculature after SCI exist. In summary, 5 categories of experimental therapy were identified: (1) delivery of angiogenic factors, (2) genetic engineering, (3) physical stimulation, (4) cell transplantation, and (5) biomaterials carrying various factor delivery [9] (Fig. 3). In the following section of this study, we aim to provide a comprehensive overview of the literature in this field, comparing and discussing the most promising approaches for clinical translation.
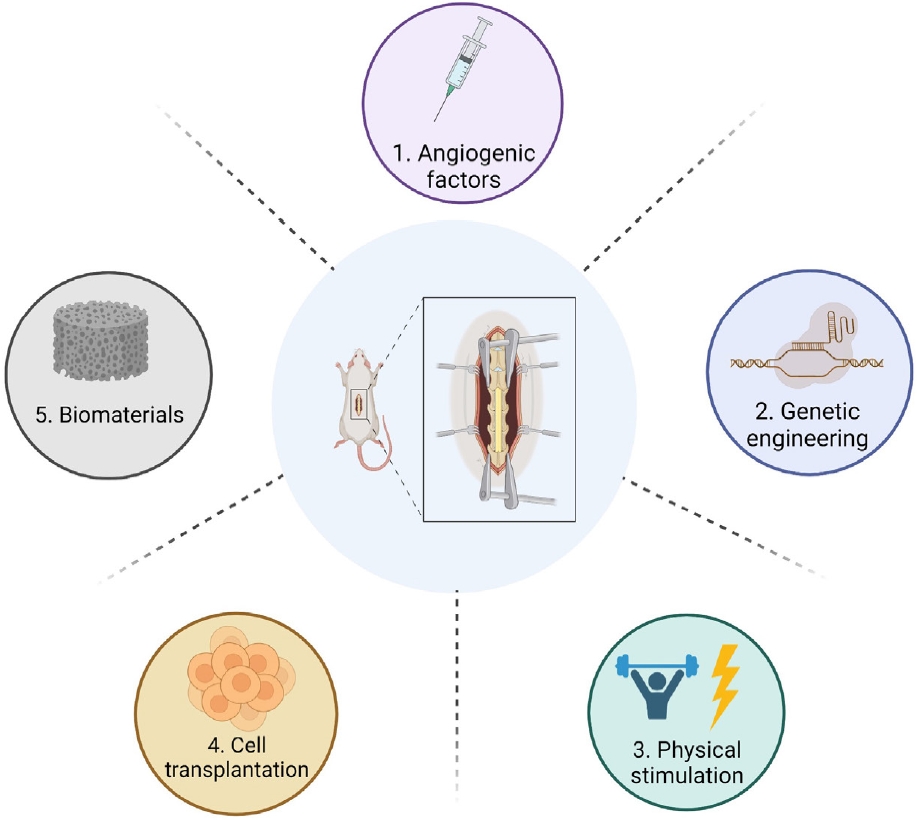
Therapeutic approaches for vascular repair after experimental spinal cord injury: (1) angiogenic factors, (2) genetic engineering, (3) physical stimulation, (4) cell transplantation, and (5) biomaterials carrying various factor delivery.
1) Angiogenic factors
Many different therapeutic agents evoking an angiogenic answer, either directly or indirectly, have been investigated in experimental SCI models, often in combination with the implantation of biomaterials. In the following, we give an overview of different angiogenic factors and their previous experimental use in SCI research (Fig. 4A).
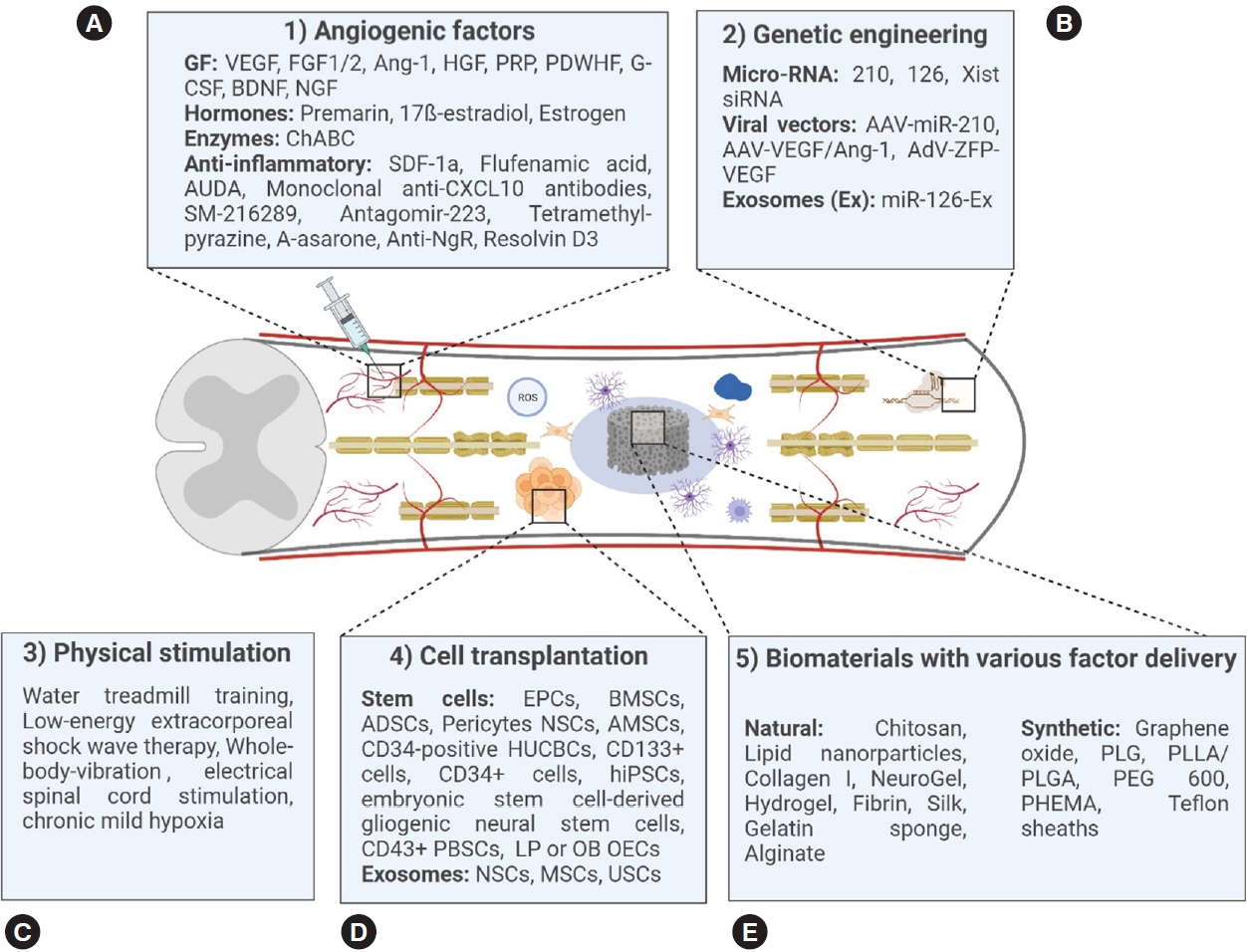
(A-E) Overview of therapeutic approaches inducing spinal cord repair after experimental spinal cord injury. PLG(A), polylactide-co-glycolide (acid); PLLA, poly-l-lactic acid; PEG600, polyethylene glycol 600; PHEMA, poly(2-hydroxyethyl methacrylate); VEGF, vascular endothelial growth factor; FGF, fibroblast growth factor; Ang-1, angiopoietin-1; HGF, hepatocyte growth factor; PRP, platelet-rich plasma; PDWHF, platelet-derived wound healing formula; G-CSF, granulocyte colony-stimulating factor; BDNF, brain-derived neurotrophic factor; NGF, nerve growth factor; ChABC, chondroitinase ABC; SDF-1a, stromal cellderived factor-1 alpha; AUDA, soluble epoxide hydrolase inhibitor; Anti-NgR, anti-nogo receptor antibody; Xist, X-inactive specific transcript; AAV, adeno-associated virus; AdV-ZFP-VEGF, adenoviral vector with zinc-finger protein coding for VEGF; EPC, endothelial progenitor cell; BMSC, bone mesenchymal stem cell; ADSC, adipose-derived stem cell; HUCBC, human umbilical cord blood stem cell; NSC, neural stem cell; AMSC, amniotic mesenchymal stem cell; hiPSC, human-induced pluripotent stem cell; PBSC, peripheral blood stem cell; LP OEC, lamina propria-derived olfactory ensheathing cell; OB OEC, olfactory bulbderived ensheathing cell; MSC, mesenchymal stem cell; USC, human urine stem cell.
The therapeutic effects of vascular endothelial growth factor (VEGF) in SCI have been variable. VEGF-A is used in many studies and is a promising growth factor that binds to the tyrosine kinase receptors (VEGFRs) 1 and 2, resulting in the proliferation of ECs and angiogenesis. VEGF-A may also have neurotrophic, neuroprotective, and neuroproliferative effects [27]. A common application of VEGF-A in experimental SCI is the direct injection into the lesion site which results in increased blood vessel density and blood vessel diameter [28-29]. This effect has been confirmed in studies of controlled release of VEGF from biomaterials, such as gel foam [30]. On the downside, increased BSCB-permeability was observed after application of VEGF-A indicating the growth of non- or only partially functional blood vessels. Changes in vessel architecture, resulting in tumor-like blood vessels, were described in human studies [27,31].
To improve the angiogenic and neurotrophic effect through the application of VEGF, a release from biomaterials in combination with other growth factors such as fibroblast growth factor-2 (FGF2) [32], angiopoietin 1 and FGF [2,33] or brain-derived neurotrophic factor [34] was explored, also resulting in increased blood vessel density and associated localization of mature oligodendrocytes and axons.
FGF2 has also been frequently applied in experimental SCI. FGF2 was found to be highly overexpressed after SCI [35] and FGF2 is known to play a role in proliferation, differentiation and BSCB-integrity [36,37]. The application of FGF2 via biomaterials has been shown to increase the number of blood vessels [38-40], whereas no significant vascular changes were observed when FGF2 was infused at the injury site [41].
Further angiogenic factors that were previously tested for application in experimental SCI are hepatocyte growth factor (HGF) and the whole blood concentrates platelet-rich plasma (PRP) and platelet-derived wound healing formula (PDWHF). The number of blood vessels has been shown to increase after the application of activated PRP [42], PDWHF alone, or PDWHF in combination with nerve growth factor and of HGF [42-44].
Apart from these often-applied factors, other factors such as hormones, especially oestrogen [45], enzymes [46], and a multitude of anti-inflammatory substances [47] listed in (Fig. 4A) also showed potential to induce angiogenesis or to increase BSCB-integrity following SCI.
2) Genetic engineering
Genetic engineering is not widely used in experimental SCI but was shown to hold the potential to increase vascular repair and functional outcome after SCI [26,48-51] (Fig. 4B). Proofs-of-concept were brought forward in experiments using RNAs like micro-RNA-210 [48], micro-RNA-126 contained in exosomes [49] and X-inactive specific transcript-RNA [50]. The endogenous overexpression of VEGF using viral vectors also increased the number of blood vessels at the injury site [26,51]. The combination of VEGF165 and Ang-1 using an adeno-associated viral vector furthermore decreased the BSCB-permeability at the injury site [51]. Therefore, genetic engineering could become a promising approach to support vascular repair after experimental SCI [51].
3) Physical stimulation
As the only noninvasive therapeutic approach, physical stimulation was also shown to increase angiogenesis and functional outcome after experimental SCI. Methods under investigation include water treadmill training [52], low-energy extracorporeal shock wave therapy [53], whole-body vibration [54], chronic mild hypoxia [55], and electric stimulation [56] (Fig. 4C). Especially water treadmill training seems to improve the regeneration of functional blood vessels and improve functional outcome after SCI [52]. Using low-energy extracorporeal shock wave therapy, not only increased vessel density but also enhanced functional outcomes suggesting that the regenerative potential is higher than the risk of further damage to the spinal cord. Electrical stimulation has made tremendous progress as experimental therapy in both preclinical and clinical studies, although only few studies focus on the evaluation of angiogenic effects. The application of transspinal direct current stimulation increases the blood temporarily and is able to stimulate cell proliferation and migration [56]. Another experimental therapeutic approach is the induction of chronic mild hypoxia after SCI. The induction of mild hypoxia results in vascular remodelling, with endothelial proliferation and vascular expansion being more pronounced in the white matter. Interestingly, newly formed blood vessels grow towards neurons and show the upregulation of TJ proteins, indicating the enhanced formation of functional blood vessels [55]. These results are promising to increase angiogenic repair after SCI, but further research needs to address the effect of these approaches in the intermediate and chronic injury phase.
4) Cell transplantation
Cell transplantation is widely used after experimental SCI. Stem cells of different origins can result in a significant increase in angiogenesis and have previously been injected, infused, or implanted following SCI with the result of potently increasing the angiogenic response and improving functional outcome (Fig. 4D). Endothelial progenitor cells (EPCs) [57], bone marrow mesenchymal stem cells [58], adipose-derived stem cells [59], human umbilical cord blood stem cells [60], neural stem cells (NSCs) [61], amniotic mesenchymal stem cells [62,63], human-induced pluripotent stem cells [64,65], peripheral blood stem cells [66], lamina propria-derived olfactory ensheathing cells, olfactory bulb-derived ensheathing cells [67], and mesenchymal stem cells (MSCs) [68] increase angiogenesis when applied after SCI by expressing a wide range of growth factors and modulating the microenvironment. The effect of these cell types is shown in Table 2.
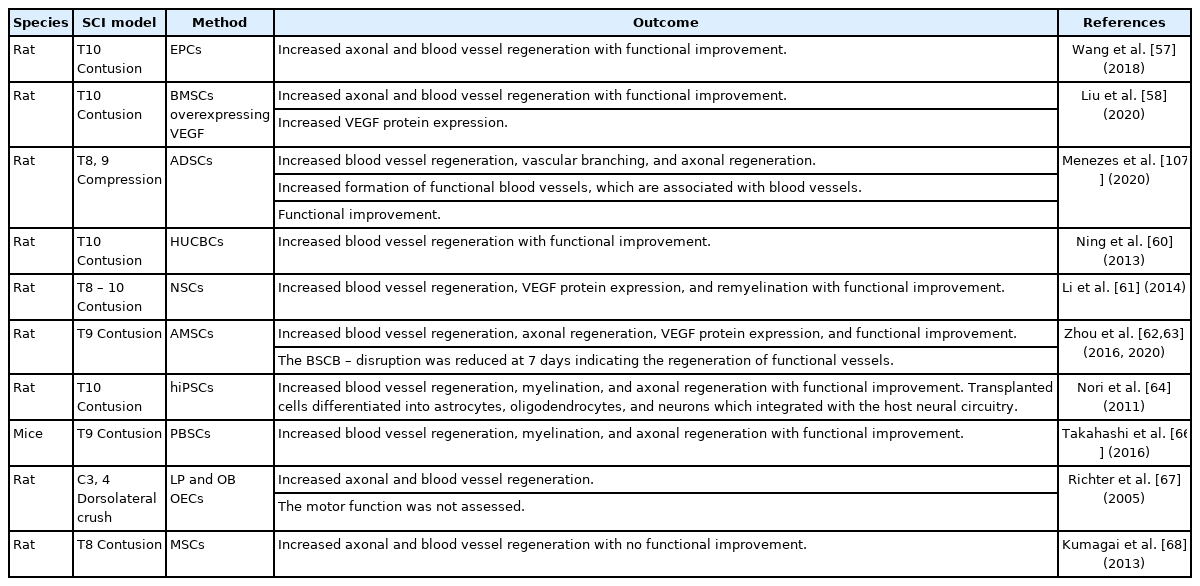
Study overview of experimentally transplanted cells to promote vascular repair after experimental spinal cord injury (SCI)
As not all implanted cells survive the hypoxic environment at and around the injury site, cell-based therapies to date can only unfold a fraction of their potential [69]. To overcome this limitation, other strategies rely on indirectly utilizing the therapeutic potential of stem cells. Especially the isolation of exosomes from different stem cells and their injection after experimental SCI showed similar results in terms of angiogenesis and functional outcome [70-72]. Exosomes derived from many different cell types result in increased angiogenesis after injection. Amongst these are NSC-derived exosomes [70], exosomes derived from human placenta-derived MSCs [71], and mesenchymal stromal cells-derived exosomes [72].
Another promising strategy is the combinatorial release of stem cells via biomaterials to enhance cell survival at the injury site by modulating the environment and by providing guidance for the regenerating tissue [69,73].
5) Biomaterials carrying various factor delivery
Biomaterials are materials engineered to interact with biological tissue and can be divided into natural materials sourced from plants or animals, synthetic materials and hybrid biomaterial combinations [10]. Many different biomaterials improve angiogenesis when implanted in combination with stem cells or angiogenic factors or even without concomitant application of other therapeutics. The implantation of biomaterials alone already holds the potential to support the ingrowth of blood vessels into the lesion site. This angiogenic potential was shown using many different biomaterials, such as a reduced graphene oxide scaffold [74], an oxygen-generating hydrogel scaffold [75,76], a nanofiber-hydrogel composite [77], “NeuroGel” [78], an aligned fibrin hydrogel [79], and a collagen type I scaffold [80] (Fig. 4E). Biomaterials can be combined with other therapeutics at will, such as angiogenic factors and/or stem cells to potentiate their regenerative potential.
Both natural and synthetic biomaterials were previously used successfully in animal studies to promote vascular repair after SCI with different advantages and disadvantages. Natural biomaterials are often intrinsically biodegradable and therefore useful to modify the delayed release of angiogenic factors or cells [81]. Synthetic biomaterials, unless modified accordingly, are nondegradable and guarantee more structural and long-lasting stability as well as reduced batch-to-batch variance. For implantation and combination with factor delivery, they can be sterilized and chemically modified to allow for an optimal release profile [10,81] (Table 3). Due to their high modifiability, both natural and synthetic biomaterials can be adapted to provide optimal release kinetics of incorporated factors at a defined location [10]. Release kinetics can be modified by using the intrinsic degradation rate of the biomaterial itself [82] or noncovalent interactions between the incorporated therapeutics and the biomaterial [40].
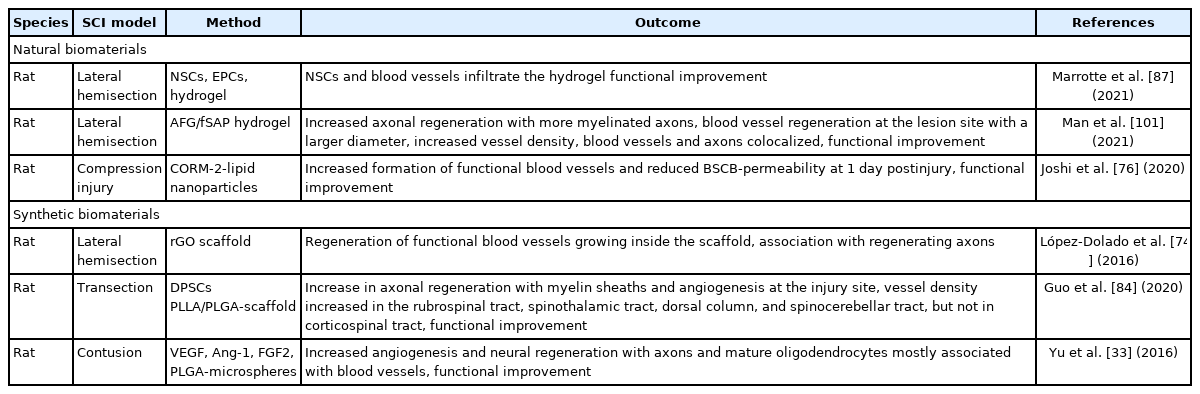
Study overview of experimentally used biomaterials to promote vascular repair after experimental spinal cord injury (SCI)
After implantation or injection, biomaterials can occupy the space of a posttraumatic cavity and provide a cell-friendly environment with reduced infiltration of inflammatory cells [77,81]. Therefore, biomaterials are often used in combination with stem cells, which highly depend on such an environment [83].
Exemplary, the number and density of blood vessels could be increased after implantation of an oligopolyethylene-glycol-fumarate-hydrogel combined with Rapamycin and Schwann-cells [83], of a prevascularized poly-L-lactic acid (PLLA)–polylactide-co-glycolide acid (PLGA) scaffold containing dental pulp stem cells [84], a fibrous porous silk scaffold containing human umbilical vein ECs [85], a hydrogel PLGA-scaffold containing NSCs and ECs [73], a gelatine sponge containing MSCs [69], and a PLGA-scaffold containing human MSCs [86]. The implantation of a hydrogel containing NSCs and EPCs also increased the formation of blood vessels inside the scaffold [87]. These findings show the great potential of combinatorial approaches using biomaterials containing angiogenic factors and/or stem cells of different types.
The application of biomaterials to promote vascular repair after SCI varies in time, location, and mode of application in the analysed studies (Table 3, Supplementary Table 1). In nearly all studies showing vascular changes, biomaterials are applied directly after induction of the injury with monitoring for up to 8 months [30,32-34,38-40,43,44,47,57,69,73-76,78,80,82-104]. Only one study reports an increased blood vessel density after the implantation of a nanofiber-hydrogel composite 3 days after injury [77]. Biomaterials are mostly applied at the injury site as scaffolds replacing the lesion after implantation [32,34,38,40,43,44,47,69,73,74,78,80,83-90,92-94,96-105] or injection[33,75,77,82,95] with only a few hydrogels being implanted on top of the injured spinal cord [30,39,91] or injected systemically [76].
POTENTIAL FOR CLINICAL IMPLEMENTATION
Taken together, all 5 experimental approaches discussed in this review show the potential to improve vascular repair and angiogenesis. The application of angiogenic factors (1) is a well-studied approach. Especially growth factors like VEGF have shown a promising potential to regenerate functional blood vessels. Genetic engineering (2) allows for direct activation of the endogenous expression of VEGF and other growth factors. Even if it is not applied as frequently as other approaches after SCI, it holds the potential to regenerate functional blood vessels. As physical stimulation techniques (3) also displayed the ability to induce vascular repair, they might be a useful supplementary therapy in a clinical setting as soon as intensive rehabilitation is possible. Cell transplantation (4) is another promising strategy to regenerate functional blood vessels after traumatic SCI, especially as they can be taken from a variety of sources. Their potential is inhibited by the toxic environment, which needs to be overcome to allow the cells to integrate at the injury site. The implantation of cells still poses a promising approach when combined with the implantation of biomaterials (5). These materials can be taken from many sources and are easily modifiable. Some have an inherent angiogenic potential and are biodegradable. As they can be modified to release incorporated therapeutics in the desired time frame at defined locations and provide guidance for regenerating axons, these combinatorial approaches seem promising for future research and potential clinical implementation. But before implementing promising biomaterials into clinical trials, several questions need to be addressed. For most biomaterials, especially when integrated in a combinatorial approach, the exact degradation time in SCI is neither known for humans nor for animals. As degradation byproducts might cause immunological reactions, biomaterials need to be actively characterized in preclinical and clinical studies [106]. Furthermore, most of the biomaterials summarized are solid materials filling a transection or hemisection injury, but contusion SCI is most common clinically. This and the overall limited comparability from controlled animal models into the individual human SCI makes it difficult to translate the results of preclinical studies into the design of clinical trials and shows the need for using clinically relevant SCI animal models with contusion or compression injuries.
Before applying these approaches in a clinical setting, future research needs to evaluate the ideal combinations to accurately address the multifaceted pathophysiology and the time course of BSCB de- and regeneration to allow for the regeneration of functional blood vessels as a basis for neural regeneration.
CONCLUSION
SCI is characterized by the injury of both neural and vascular components and results in extensive tissue loss. The disruption of the vasculature and its insufficient regeneration with a prolonged state of BSCB-permeability exacerbate the primary injury by amplification of secondary injury mechanisms. To restore a functional vasculature to prevent neural loss and to promote neural regeneration is one of the most important challenges in SCI research. Several therapeutic approaches show an improved vascularization after SCI, like the application of angiogenic factors, genetic engineering, physical stimulation, cell transplantation, and biomaterials carrying various factor delivery. Combinatorial approaches, like implanted biomaterials with the ability to release angiogenic factors or therapeutic cells in a temporally and spatially controlled manner seem most promising to restore functional vasculature and to be translatable into clinical patient care in the future.
SUPPLEMENTARY MATERIALS
Supplementary Table 1 can be found via https://doi.org/10.14245/ns.2244624.312.
Complete list of studies using biomaterials to promote vascular repair after spinal cord injury identified by the systematic literature search.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contribution
Conceptualization: LR, VH, MGF, PV; Data curation: LR, VH; Formal analysis: LR, VH; Project administration: VH, MGF; Visualization: LR; Writing - original draft: LR, VH; Writing - review & editing: VH, JS, LKS, MGF
Acknowledgements
Figures were created using BioRender.com.


