Relationship Between Types of Warming Devices and Surgical Site Infection in Patients Who Underwent Posterior Fusion Surgery Based on National Data
Article information
Abstract
Objective
Perioperative hypothermia can lead to various complications. Although various warming techniques have been used to prevent perioperative hypothermia, the effect of these techniques on surgical site infection (SSI) during posterior fusion surgery is unclear. The effects of warming devices on SSI rates were therefore analyzed using data complied by the Health Insurance and Review Assessment (HIRA) Service in Korea.
Methods
This study included 5,406 patients in the HIRA Service database who underwent posterior fusion surgery during the years 2014, 2015, and 2017. Factors related to SSI in these patients, including warming devices, antibiotics, and transfusion, were analyzed.
Results
The incidence of SSI was higher in patients who underwent forced air warming than in those who did not undergo active warming (odds ratio [OR], 1.73; p = 0.039), especially above 70 years old (OR, 4.11; p = 0.014). By contrast, the incidence of SSI was not significantly higher in patients who underwent device using conduction. Infection rates were higher in patients who received prophylactic antibiotics within 20 minutes before incision, than within 21 to 60 minutes (OR, 2.07; p = 0.001) and who received more blood transfusions (1 pint < volume ≤ 2 pint; OR, 1.75; p = 0.008, > 2 pint; OR, 2.73; p = 0.004).
Conclusion
SSI rates were higher in patients who underwent warming with forced air devices than with devices using conduction, as well as being higher in patients who older age, received blood transfusions and administered antibiotics within 20 minutes before incision. Devices using conduction have more advantages in preventing SSI than forced air warming device. In addition, the reduction of other risk factors for SSI may improve postoperative results.
INTRODUCTION
Perioperative hypothermia, defined as a core temperature below 36°C, has been associated with adverse outcomes in surgical patients, including intraoperative blood loss, cardiac events, coagulopathy, and increase hospital stay and associated costs [1-3]. Perioperative hypothermia may be prevented by active and passive warming methods. Active methods include warming intravenous fluids, patients warming devices and passive methods include insulation and radiant heat loss prevention using blanket. Among them, forced warming device and device using conduction are currently mainly used as patient warming devices [4].
Although these warming devices are known to be effective in preventing hypothermia, they had to be used around the surgical field, causing many concerns about their relationship with surgical site infection (SSI) [2-4]. However, there are still many controversies about the effect of these active warming devices on the rate of SSI after surgery [5,6]. In addition, there has been no study on the association in posterior spinal fusion surgery, which is performed in a relatively large number compared to other spinal surgeries but has a higher SSI rate [7-9].
Therefore, the present study analyzed the effects of these warming devices during posterior fusion surgery in patients enrolled in a nationwide database. In addition, the present study also assessed the relationships of other factors, such as the timing of antibiotic administration and blood transfusion, with the rate of SSI.
MATERIALS AND METHODS
1. Study Population and Data
The Health Insurance Review and Assessment (HIRA) Service in Korea has been conducting an analysis titled “Evaluation of the Appropriate Use of Prophylactic Antibiotics” in patients who underwent spine surgery in 2014, 2015, and 2017 [10]. The same analysis was not conducted in 2016. This evaluation was designed to prevent SSI and the misuse of antibiotics by selecting and administering prophylactic antibiotics that meet the standards for clean and uncontaminated surgery. The hospitals targeted for evaluation were all tertiary general hospitals, general hospitals, and hospitals in South Korea. Hospitals participating in this evaluation were required to provide the HIRA Service with information related to surgery, such preoperative antibiotics use and duration of spine surgery, excluding emergency surgery. Variables recorded in the database included patient age, sex, use of a warming device (yes vs. no), type of warming device, time of prophylactic antibiotic administration, type of prophylactic antibiotics, duration of operation, blood transfusion (yes vs. no), American Society of Anesthesiologist (ASA) physical status classification grade, type of hospital, and occurrence of SSI (yes vs. no). Patients included in this retrospective nationwide cross-sectional study were defined as those who underwent elective posterior fusion surgery (code number N0469, N1460, N1469, N2470) for lumbar vertebra. Patients with incomplete information were excluded.
The study protocol was approved by the Institutional Review Board (IRB) of Eulji Medical Center, which waived the requirement for patient informed consent because of the retrospective nature of the data analysis (IRB No. EMC 2023-01-003).
2. Variables
SSI was defined as follows: (1) a purulent discharge in the operation wound, (2) identification of an infective organism in wound culture, or (3) a surgical intervention for a wound discharge. The independent variable was the use of intraoperative warming devices. Patients were classified into 3 subgroups according to type of warming device: none; forced air warming devices; or devices using conduction, including warm water garments, electric blankets, and water heating circulation pump (Fig. 1). Patients were also sub grouped according to the time of prophylactic antibiotic administration: within 20 minutes, or 21 to 60 minutes before surgery.
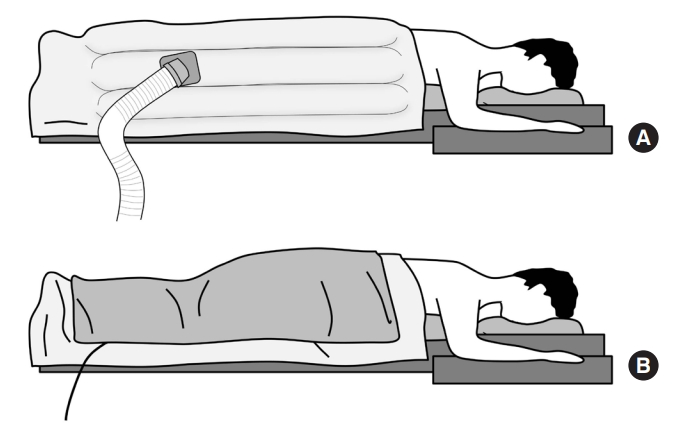
Description of 2 representative warming devices. (A) Forced air warming device. (B) Device using conduction.
Other independent variables affecting SSI were also considered covariates. They included sex, age, cause of surgery (traumatic or nontraumatic), type of prophylactic antibiotics (first and second generation cephalosporins vs. other types of antibiotics, including third and fourth generation cephalosporin, quinolone, and glycopeptides), the length of the operation (< 2.5 hours vs. ≥ 2.5 hours), blood transfusion (yes vs. no), ASA physical status classification grade, type of hospital (tertiary general hospital, general hospital or hospital), and Charlson Comorbidity Index (CCI, 0, 1, 2, ≥ 3), and year of surgery. CCI was calculated by weighting and scoring comorbid conditions using Quan’s method, with additional points given to comorbidities that affect the health outcomes [11].
3. Statistical Analysis
We used the chi-square tests for categorical variables and analysis of variance for continuous variables to evaluate differences between patients according to warming devices. Multiple logistic regression analysis was conducted to estimate adjusted odds ratios (aORs) and 95% confidence intervals (CIs) to assess the association between warming devices and SSI. In other words, active warming techniques were divided into forced air warming devices and devices using conduction, and whether warming devices were associated with SSI was analyzed. Subgroup analyses were performed to evaluate the interactions between warming techniques and time of prophylactic antibiotics administration in relation to SSI after adjusting all covariates. Moreover, we analyzed the interaction between prophylactic antibiotics and timing of prophylactic antibiotic administration in relation to SSI for providing actionable insights to medical professionals. Finally, the association between warming devices and SSI stratified by age group and blood transfusion was evaluated and we adjusted all covariates excluding stratified variables. All calculated p-values were 2-sided, and p-values < 0.05 were considered statistically significant. All analyses were performed using SAS Enterprise Guide ver. 7.1 software (SAS Inc., Cary, NC, USA).
RESULTS
Of the 6,423 patients who underwent posterior spinal fusion surgery during the study period, 5,405 patients met the inclusion criteria. Reasons for excluding of the remaining 1,018 patients included preoperative infection in 68 patients and inadequate data in 950. Subclassification by type of warming device showed that 1,825 patients (33.8%) were not treated with and intraoperative warming device, 2,764 (51.1%) were treated with forced air warming devices, and 816 (15.1%) were treated with devices using conduction. Subclassification by time of prophylactic antibiotic administration showed that 2,377 patients (44%) were administered antibiotics within 20 minutes before surgery and 3,028 (56%) received antibiotics 21 to 60 minutes before surgery. SSI occurred in 88 patients (1.6%) (Table 1), including 21 who were not treated with an intraoperative warming device, 56 who were treated with forced air warming devices, and 11 who were treated with conduction devices. Of the 88 patients with SSI, 55 received prophylactic antibiotics within 20 minutes and 33 received prophylactic antibiotics 21 to 60 minutes before surgery.
Table 2 presents the factors associated with SSI in patients undergoing posterior fusion surgery for lumbar vertebrae. Compared with those not treated with intraoperative warming devices, those treated with forced air warming devices were 1.73-fold more likely (95% CI, 1.02–2.89; p = 0.039) to develop SSI after adjusting for all covariates. SSI was also 2.07-fold more likely (95% CI, 1.33–3.22; p = 0.001) in patients receiving prophylactic antibiotics within 20 minutes than 21 to 60 minutes before surgery, after adjusting for all covariates.
Subgroup analysis of SSI risk according to the type of intraoperative warming device and time of prophylactic antibiotic administration showed that, among patients administered antibiotics 21 to 60 minutes before surgery, the risk of SSI did not differ in patients who no intraoperative warming device and those with devices using conduction (p = 0.211) (Table 3). By contrast, an analysis of patients administered antibiotics 21 to 60 minutes before surgery showed that the risk of SSI was 5.17-fold (95% CI, 1.79–14.97; p = 0.002) higher in patients treated with forced air than with no intraoperative warming device. Moreover, administration of prophylactic antibiotics within 20 minutes before surgery increased the risk of SSI in all subgroups (p < 0.05).

Interactions between warming techniques and time of administration of prophylactic antibiotics in relation to surgical site infection
Table 4 shows the interactions between prophylactic antibiotics and time of prophylactic antibiotics and time of prophylactic administration in relation to SSI. Within 20 minutes before surgery, even if a generally recommended antibiotics (1st or 2nd generation cephalosporins) was used, the rate of SSI increased 2.01-fold more likely (95% CI, 1.27–3.17; p = 0.003), and if a nonrecommended antibiotics was used, the rate of SSI increased 2.64-fold more likely (95% CI, 1.01–6.95; p = 0.047).

Interactions between prophylactic antibiotics and time of prophylactic antibiotics administration in relation to surgical site infection
Table 5 shows the results of the stratified analyses according to independent variables. The risk of SSI was 4.11 times (95% CI, 1.33–12.64; p = 0.014) higher in patients aged >70 years treated with than without forced air warming devices. Additionally, the risk of SSI was higher in patients who received blood transfusions (aOR, 2.61; 95% CI, 1.11–6.17; p = 0.029) when forced warming devices were used.
DISCUSSION
There are 2 main types of active warming technique: forced air warming devices and devices using conduction. Forced air warming devices suck in air from the surroundings and warm this air with electric coils. A blower circulates the warm air through a blanket that warms patients through convection. Device using conduction involve the use of conductive polymer fiber sheets that produce heat and warm patients through conduction [3,4].
Forced air warming systems may increase the risk of SSIs by acting as a vector or causing unwanted airflow disturbances. McGovern et al. [5] reported forced air warming disrupts laminar flow ventilation and significantly increases SSI in patients undergoing arthroplasty surgeries. Belani et al. [12] reported forced air warming device significantly increase the bubble counts compared to device using conduction in operating room. By contrast, other studies have reported that the type of warming device did not significantly affect bacterial counts at any sampling sites in operating room [6,13]. Although the relationships between warming devices and SSI rates remain unclear, few studies have compared results in large numbers of patients, and no studies to date have used nationwide data. Therefore, the results of the present study showing that use of forced warming devices increases SSI rates are clinically meaningful.
Forced air warming device were the most common type of active warming method in 2014, 2015, and 2017, being used in 51.1% of patients who underwent posterior spinal fusion surgery in South Korea. According to McGovern et al. [5], however, a change of warming technique from forced air warming to devices using conduction around 2010 reduced the rate of SSI in patients underwent hip and knee replacement cases from 3% to less than 1%. Forced air warmers may interrupt the flow of filtered air toward the area of the wound and may allow dust particles containing pathogenic organisms to contact with the wound. Because SSI is relatively common in patients undergoing posterior spinal fusion compared to other surgeries, it is recommended that these patients undergo intraoperative warming with devices using conduction.
Many studies have evaluated the relationships between SSI rates and prophylactic antibiotic administration, including the type of antibiotics and optimal timing. Canseco et al. [14] reported that the risk of SSI was higher in patients administered antibiotics ≥ 61 minutes than 0–15 minutes before incision, with each additional 1 minute delay increasing the likelihood of SSI 1.05-fold. Garey et al. [15] reported the incidence of SSI in patients who underwent cardiac surgery was much lower in patients administered antibiotics 16–60 minutes than 0–15 minutes before incision (3.4% vs. 26.7%). Most studies have recommended that antibiotics be administered within 1 hour before incision, but the optimal time has not been determined. The present study, however, found that too early administration was detrimental to patients, as these antibiotics do not have sufficient time to reach the incision site, suggesting that the optimal time of incision be at least 20 minutes after antibiotic administration [16-19].
Fisahn et al. [20] reported that the SSI rates are significantly higher in patients who did than did not receive blood transfusions during major spinal fusion surgery (> 8 levels, 36% vs. 10%, p = 0.03). In addition, 8% of these patients had wound infections, all of whom received blood transfusions. A meta-analysis of 34,185 patients reported that the rate of SSI was about 2.9-fold higher in patients who did than did not receive transfusions [21]. The present study confirmed that transfusion was associated with an increased SSI rate.
The present study had several limitations. First, it did not analyze the ability of warming devices to maintain body temperature during surgery, as data on body temperatures immediately prior to surgery were unavailable. Since hypothermia is also a risk factor for infection, it would have been better if the body temperature maintenance following warming device was also investigated in this study. In addition, we did not analyze the patient factor including smoking, glucose level, weight loss, the surgeon factors including skin preparation methods, hand hygiene, irrigation, and the operating room factors including temperature, laminar flow setting could not be determined. Because this study evaluated prospectively collected nationwide real-world data, it likely reflects all the differences in facilities among hospitals in South Korea. However, since this study was conducted in one country and targeted many numbers of patients at the general hospital level or higher, we believed that it could be meaningful even if some biases were considered. Although the results of this study are not absolute due to the theses biases, we thought that this study suggests some direction for the relationship between warming devices and SSI. In addition, the data were adjusted according to the type and timing of administration of preoperative antibiotics as well as the underlying diseases that can affect the incidence of SSI. Finally, the cross-sectional nature of this study requires cautious interpretation of the causal relationship between the use of active warming devices in spine surgery and SSI.
CONCLUSION
Although this study has limitation in not confirming the effect of the warming device on body temperature, devices using conduction seemed to have more advantages in preventing SSI than forced air warming device. In addition, blood transfusion and early administration of prophylactic antibiotics might be also associated with increased SSI rates. The reduction of other risk factors, including avoiding blood transfusion and proper timing of antibiotic administration, may also improve postoperative results.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author Contribution
Conceptualization: YC; Data curation: BYK, HJL, GOK; Formal analysis: SHK; Methodology: SHK; Visualization: SYS; Writing - original draft: SYS; Writing - review & editing: JHC.
Acknowledgements
This study was conducted as part of a Joint Project on Quality Assessment Research of Health Insurance Review & Assessment (HIRA) Service. This study was based on data from the HIRA (research data number: M20220219833).



