Pneumocephalus and Pneumorrhachis due to a Subarachnoid Pleural Fistula That Developed after Thoracic Spine Surgery
Article information
Abstract
Development of a communication between the spinal subarachnoid space and the pleural space after thoracic spine surgery is uncommon. Subarachnoid pleural fistula (SAPF), a distressing condition, involves cerebrospinal fluid leakage. Here we report an unusual case of SAPF, occurring after thoracic spine surgery, that was further complicated by pneumocephalus and pneumorrhachis postthoracentesis, which was performed for unilateral pleural effusion.
INTRODUCTION
Pneumorrhachis and pneumocephalus due to a fistula are rare. Another rare condition is a communication between the spinal subarachnoid space and pleural space after thoracic spine surgery. Pneumocephalus may manifest as severe headache4579) and acute neurologic changes that mimic brain metastasis, seizure, or stroke. However, the physiology and management of pneumocephalus are not widely recognized. Here we report an unusual case of subarachnoid pleural fistula (SAPF), occurring after thoracic spine surgery, that was further complicated by pneumocephalus and pneumorrhachis postthoracentesis, which was performed for unilateral pleural effusion.
CASE REPORT
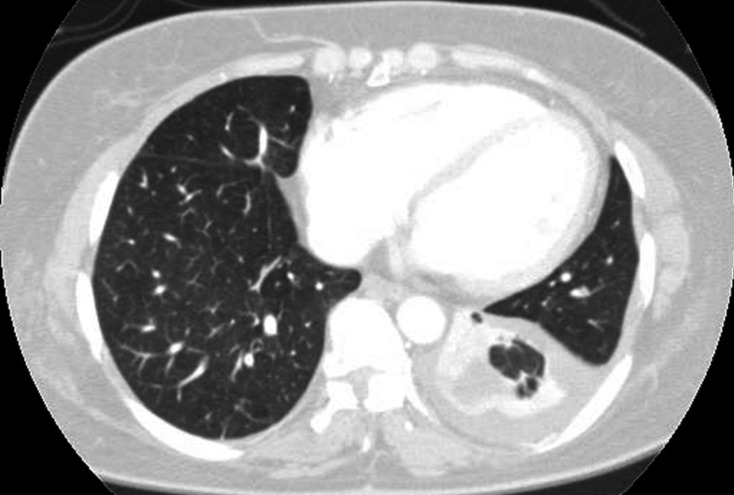
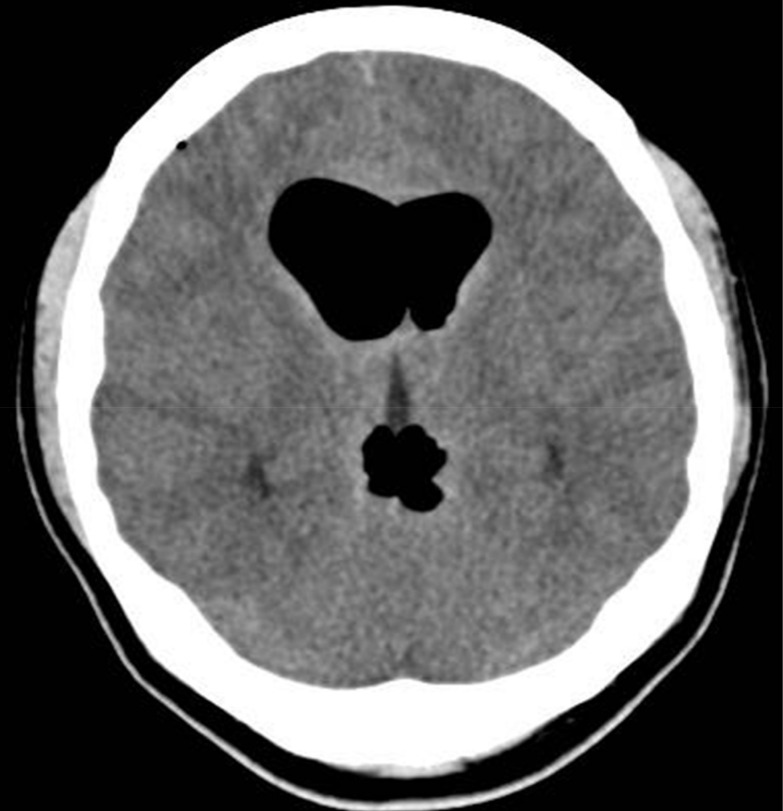
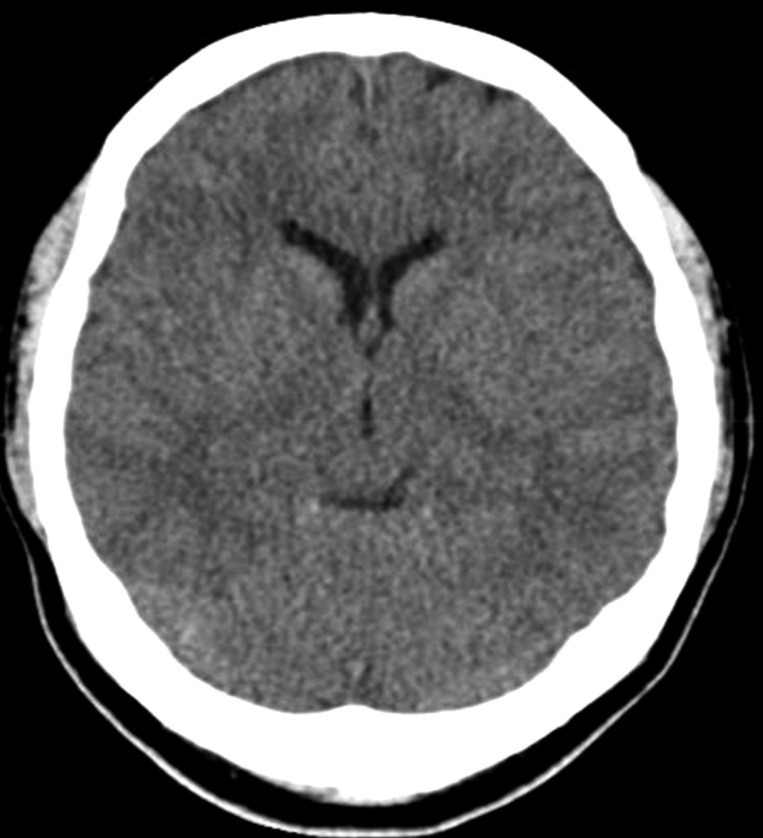
A 47-year-old woman presented with severe headache, nausea, and vomiting. A known case of T1-T8 posterior longitudinal ligament ossification, she had undergone decompressive laminectomy at T1-T2 and T8, 7 years ago and posterior decompression at T11-L2, 2 years ago at another hospital. At that time, dural tear was unknown. She was in her usual state of health until 10 days before admission to the respiratory department for violent coughing and sputum. A chest radiograph and computed tomography (CT) scan (Fig. 1) revealed left-sided pleural effusion. Thoracentesis was performed, following which she developed sudden headache, nausea, and vomiting. Brain CT scan revealed pneumocephalus, with air in the ventricle and the subarachnoid space (Fig. 2). A chest CT showed a communication between the pleural cavity and the T2 spinal canal with intraspinal air (Fig. 3). The precise location of the SAPF was identified on a CT myelogram with iohexol. The patient refused surgical repair of the SAPF; therefore, conservative management including bed rest with decubitus position was adopted as treatment. Her symptoms improved dramatically. A follow- up brain CT scan after 27 days showed complete resolution of the ventricular and subarachnoid air (Fig. 4). Consecutive chest radiographs showed left-sided pleural effusion, but she had no cough, difficulty in breathing, or sputum. She was discharged and sent home after 7 weeks since admission, and was followed-up in the outpatient department.
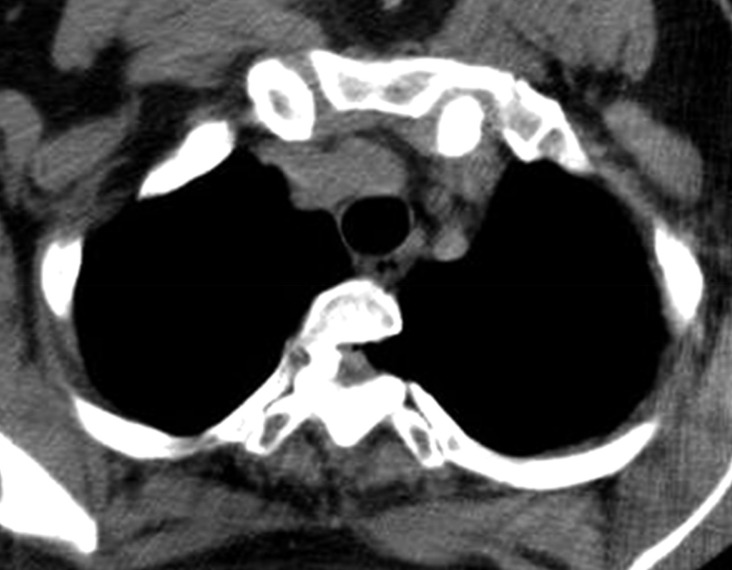
Chest computed tomography scan after thoracentesis showing air collection in the T2 spinal canal and a fistula.
DISCUSSION
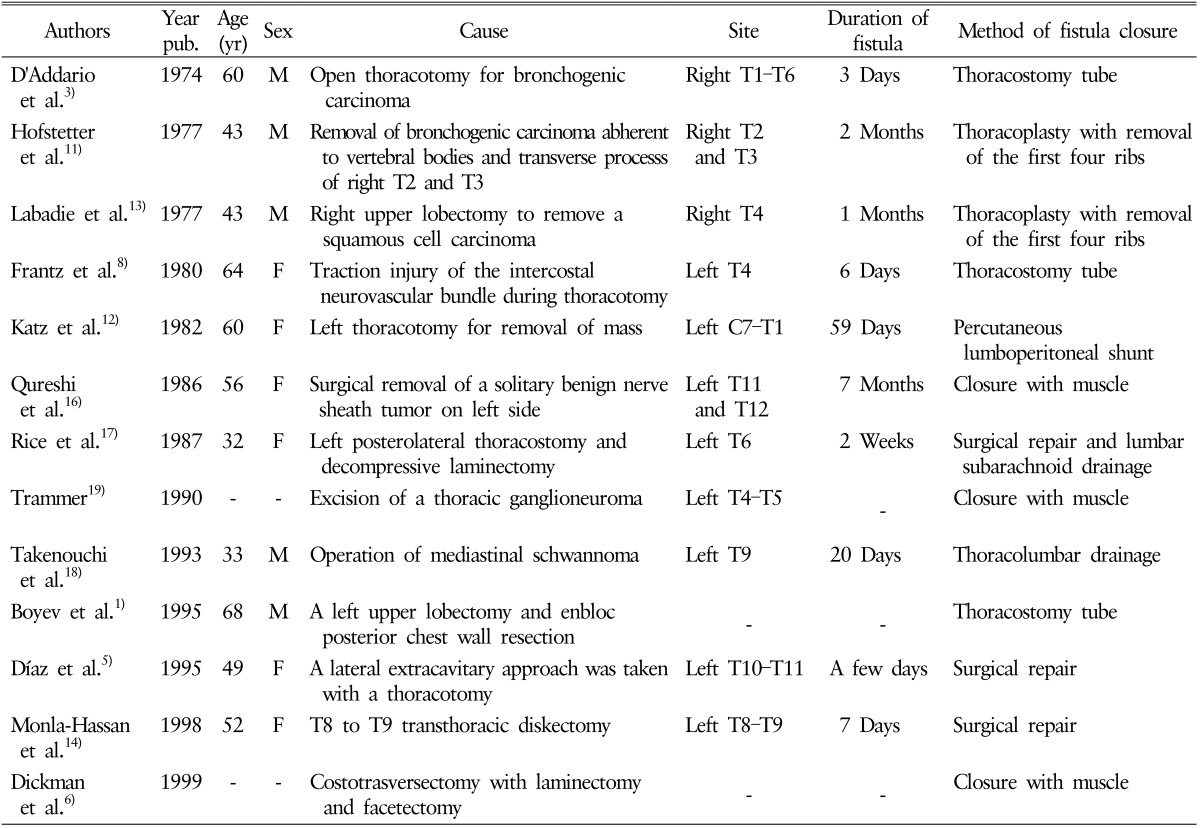
SAPFs are rare and difficult to diagnose. Only 13 cases of SAPF following surgery have been reported in the literature135681112131416171819) (Table 1). Postoperative pneumocephalus could result from combination of a dural tear and a pneumorrhachis. An SAPF may remain asymptomatic when small, or may cause pleural effusion and subsequent dyspnea and pleuritic chest pain, in addition to postural headaches4579), nausea, and vomiting secondary to intracranial hypotension1). An SAPF once developed causes a pressure gradient, which allows cerebrospinal fluid (CSF) to flow from the positive-pressure spinal subarachnoid space to the negative-pressure pleural space. The low thoracic pressure created during inspiration causes CSF to flow into the chest cavity, and the high intrathoracic pressure during expiration forces air into the CSF space. This is further complicated by an upright head position that causes an increase in air in the subarachnoid space, resulting in pneumocephalus. The diagnosis of SAPF requires an appropriate clinical setting and relies mostly on the aforementioned symptoms and signs, although imaging modalities are of great value in confirming the presence of the fistula. The commonly used imaging modalities are radionuclide cisternography11), CT myelography10), and magnetic resonance imaging. Management of SAPF depends not only on the size of the fistula defect but also on the progression of symptoms, and choosing a treatment strategy is the most challenging part.
Several reports showing that conservative management is available in most of patients618).
In a review of 19 cases of SAPF secondary to trauma, 63% were treated surgically, 26% via chest tube drainage or lumbar drainage, and 11% conservatively15). Furthermore, 27% of the fistulae developing postthoracotomy closed spontaneously2). Conservative therapy, which includes bed rest, external lumbar drainage, and chest tube insertion, has been advocated as the initial treatment strategy. Surgical repair of the fistula should be considered after the failure of conservative treatment; however, the appropriate timing for chest tube drainage or surgical intervention remains controversial. Early detection and appropriate therapy will decrease morbidity associated with this rare complication.
CONCLUSION
Clinicians must be aware of the possibility of SAPF in patients with unilateral pleural effusion who have undergone thoracic spine surgery. Conservative management may be feasible in patients with SAPF, and long-term follow-up is imperative.
Notes
CONFLICT OF INTEREST: No potential conflict of interest relevant to this article was reported.