Multimodal Repair of Spinal Cord Injury With Mesenchymal Stem Cells
Article information
Abstract
Spinal cord injury (SCI) is a result of a devastating injury to the central nervous system. Currently, there is no effective treatment available for these patients. The possible use of mesenchymal stem cell (MSC)-based treatment for SCI has been the focus of extensive investigations and is increasingly moving from the bench to bedside. Both experimental observations and clinical studies have shown the safety and efficacy of MSCs in managing SCI. However, the exact mechanism by which MSCs contribute to the repair of the injured spinal cord remains to be elucidated. In this review, we aim to summarize current research findings about the role of MSCs in improving complex pathology after SCI. MSCs exert a multimodal repair mechanism targeting multiple events in the secondary injury cascade. Our recent results showing the perineurium-like differentiation of surviving MSCs in the injured spinal cord may further the understanding of the fate of transplanted MSCs. These findings provide fundamental support for the clinical use of MSCs in SCI patients. Under experimental conditions, combining novel physical, chemical, and biological approaches led to significant improvements in the therapeutic efficacy of MSCs. These findings hold promise for the future of cell-based clinical treatment of SCI.
INTRODUCTION
Spinal cord injury (SCI) is a devastating central nervous system condition, caused by direct physical impact. The lack of effective treatment causes significant public concerns. Following SCI, patients live with some degree of permanent disability and several complications can arise. According to the World Health Organization, the global estimated prevalence of SCI is 40 to 80 cases per million population. For example, in the United States, there were 17,810 new cases reported in 2020, with a total of 294,000 Americans living with SCI [1]. The provision of effective treatment for SCI remains an unmet medical need [2]. Alleviating the suffering of these patients and reducing the need for their extensive care is a challenge to society.
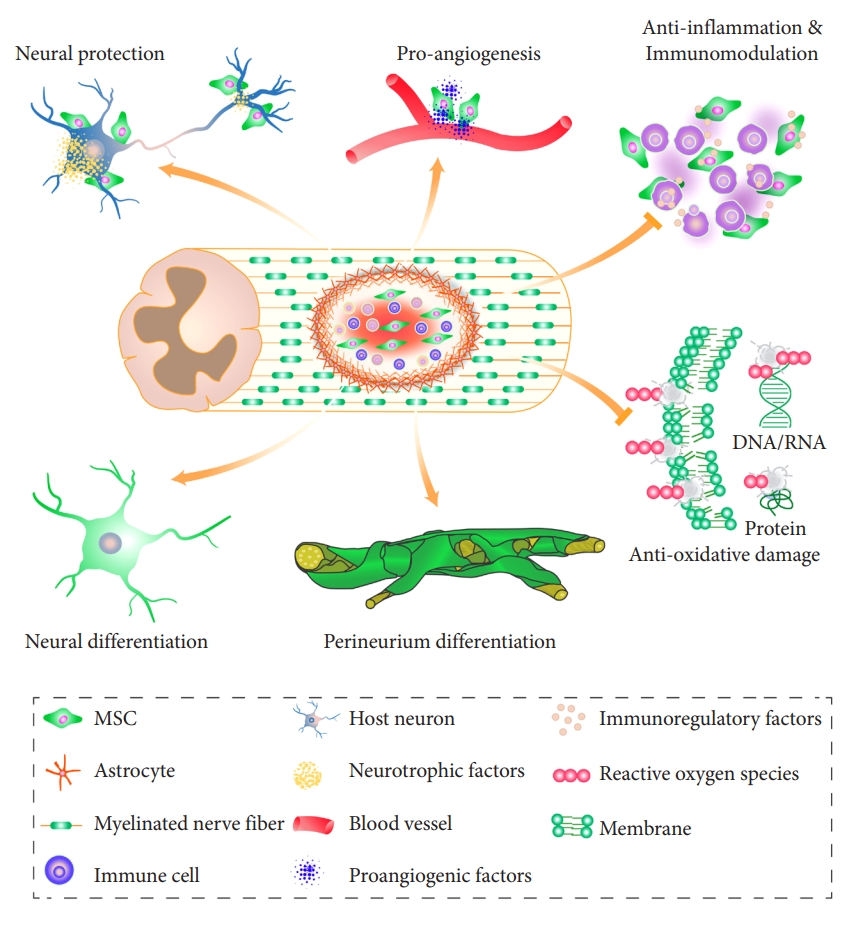
The recent emergence of stem cell-based tissue engineering is a promising strategy for SCI repair. Amongst the possible candidate seed cells, mesenchymal stem cells (MSCs) are the most extensively studied both in basic science experiments and in translational research. According to the Web of Science database, over 2,200 publications appeared on this topic and the National Library of Medicine database contains 40 registered clinical studies in various countries, including China (10), the United States (7), and the Republic of Korea (3). Due to their immunomodulatory, anti-inflammatory, and pro-angiogenic properties, MSCs show a strong potential to promote tissue repair [3-5], with ongoing registered clinical trials [6,7]. Despite this extensive work, the mechanism that allows MSCs to repair SCI remains to be elucidated. Recently, we described a novel mechanism, where MSCs differentiate into perineurium-like sheaths to protect neural tissue damage [8]. It seems that MSCs provide a multimodal repair, addressing several events in the multidimensional pathophysiology seen after SCI. In this review, we summarize the possible roles of MSCs in the treatment of SCI, highlighting multiple mechanisms that underlie their therapeutic benefits.
MULTIDIMENSIONAL PATHOPHYSIOLOGICAL FEATURES OF SPINAL CORD INJURY
A full understanding of the pathophysiological events that follow SCI is necessary to identify effective treatments. The primary injury is the physical impact affecting the spinal cord directly as a result of a traffic accident, fall, sports accident, or other violent events. This initial impact can displace bone fragments and components of the intervertebral discs. Alternatively, ligaments bruise or tear into the spinal canal. These mechanisms lead to the penetration, contusion, or compression of the cord tissue [9]. The primary injury activates a rapid cascade of secondary pathophysiological changes that are collectively referred to as secondary injury. There are three early phases of secondary injury: the acute phase (within 48 hours), the subacute phase (2–14 days), and the intermediate phase (14 days–6 months). These are followed by a chronic phase (beyond 6 months) [10]. While characteristic pathophysiological changes occur during each of these phases, these are not entirely independent, but represent a step in a complex series of interlinked events. The main pathophysiological changes after SCI include hemorrhage, ischemia-reperfusion injury, alterations in ion distribution, excitotoxicity, oxidative damage, and axonal degeneration [11]. The homeostasis of the spinal cord internal microenvironment is completely disrupted after injury, leading to a cascade of pathophysiological changes. Despite our current insight into the biochemical reactions and pathways involved in the progression of SCI, much remains to be explored. A fuller understanding of the pathophysiology after injury will help identify potential avenues to improve current therapies, and potentially create new approaches that may promote spinal cord tissue regeneration.
FUNCTIONAL MULTIPOTENCY OF MESENCHYMAL STEM CELLS
In the 1970s a new type of adherent stromal cell was isolated and cultured from the bone marrow [12]. These cells exhibited fibroblast-like morphology and were multipotent cells that had the capacity to self-renew. Importantly, these cells could differentiate into a variety of cell types, including osteogenic, adipogenic, chondrogenic, and myogenic mesenchymal lineages in vitro [13-15]. Based on these characteristics, they were referred to as “mesenchymal stem cells” by Caplan in 1991 [16]. MSCs adhere to plastic under standard culture conditions and express CD90, CD105, and CD73. At the same time, they are CD45, CD34, CD14, CD11b, CD79α, or CD19 negative [17]. To date, no single marker has been identified that could distinguish MSCs from other cell types. Subsequent studies have shown that MSCs can be isolated from a variety of tissues, including bone marrow, umbilical cord, dental pulp, and adipose tissue [18]. The ability to establish MSCs from such a wide range of tissues boosted their potential for clinical applications, as these cells could be obtained without serious ethical concerns. Furthermore, MSCs are considered to be immune-privileged, because of their low immunogenicity. They express very low levels of MHC class I molecules and are completely devoid of MHC class II. There is growing evidence suggesting that MSCs can suppress immune responses [19]. Moreover, clinical studies showed the safety of MSC transplantation, excluding the risks of tumor formation [20]. Apart from proving their safety, studies have also demonstrated that in SCI MSCs do not act via a single mechanism but promote a multimodal repair. This multitude of repair mechanism can be regarded as the functional multipotency of MSCs [21-23], suggesting that they have the capacity to simultaneously treat multiple aspects of the complex pathology that characterizes SCI.
1. Proangiogenic Features of MSCs
MSCs are potent inducers of angiogenesis and vasculogenesis when transplanted into tissues. This property has been observed under several conditions, including cardiovascular disease [24], wound healing [25], and SCI [26]. Reestablishing the blood supply is a critical requirement for successful tissue regeneration. The molecular mechanisms promoting angiogenesis by MSCs involve the secretion of the tissue inhibitor of metalloproteinase-1, vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), platelet-derived growth factor, interleukin (IL)-6, and IL-8 [27,28]. Apart from the secretion of these cytokines, MSCs also contribute to blood vessel formation via additional mechanisms. These include providing physical contacts supporting growth and the production of components of the extracellular matrix (ECM). In a previous study, we found several transplanted MSCs surrounding newly formed blood vessels while others encircled tightly the epithelial layer, forming pericytes in the injured spinal cord. MSCs surrounding these structures expressed hypoxia-inducible factor 1-alpha and VEGF, and deposited fibronectin (FN) around the blood vessel inside [5]. This topology may contribute to synergistic effects, promoting the formation and maturation of blood vessels after SCI. This angiogenic nature of MSCs may counteract the effects of tissue ischemia developing after injury. By improving blood supply, MSC transplants promote the delivery of nutrients and oxygen necessary for tissue regeneration.
2. Anti-inflammatory and Immunomodulatory Capacity
MSCs can suppress the activity of a variety of immune cells, including T and B lymphocytes, neutrophils, monocytes, and macrophages [29]. MSCs can strongly inhibit the proliferation of mitogen or alloantigen-activated T lymphocytes [30,31], irrespective of whether the T cells were autologous or allogeneic [32,33]. MSCs were also able to modulate immune responses by interacting with regulatory T cells (Treg), involved in the maintenance of immune homeostasis and self-tolerance [34]. Evidence suggests that MSCs can increase the number of Treg cells while simultaneously improving their immune suppressive action [35,36]. Key molecules involved in this interaction include MSC-derived soluble factors, such as prostaglandin E2 (PGE-2), transforming growth factor (TGF)-β1, indoleamine 2,3-dioxygenase (IDO), and HGF [37-40]. Recent studies have also shown that MSCs can inhibit B cells by arresting their proliferation at the G0/G1 phase of the cell cycle [41]. Furthermore, MSCs alter B-cell chemotaxis by significantly down-regulating the expression of CXCR4, CXCR5, CCR7, and their ligands, CXCL12, and CXCL13 [42]. It was also demonstrated that MSCs can directly inhibit the transformation of B cells into plasma cells [43]. The key molecules involved in this inhibition include interferon (IFN)-γ and B-cell activating factor [44]. MSCs can rescue resting and IL-8-activated neutrophils from apoptosis by constitutively releasing IL-6 in vitro [45]. They can also sustain and amplify the function of neutrophils via endogenously produced IL-6, IFN-β, and granulocytemacrophage colony-stimulating factor [46]. MSCs seem to drive macrophage polarization towards the anti-inflammatory M2 phenotype through the suppression of nuclear factor-kappa B p65 and the activation of STAT3 pathways [47]. This extensive immunosuppressive and regulatory action of MSCs has attracted considerable interest for potential clinical use. The infiltration of inflammatory cells into the injured SCI generates a microenvironment that is detrimental to recovery. The anti-inflammatory and immunoregulatory effects of MSCs may provide a promising approach to alleviate this neuroinflammation.
3. MSCs Show Strong Secretory Actions: Production of Nutritional Factors and Exosomes
There is a view that the therapeutic effect of MSCs transplantation does not occur via direct cell replacement, but through the modulation of the host microenvironment [48]. Indeed, the nutritional activity of MSC has been widely confirmed and has become the focus of research in the treatment of several diseases [49]. MSCs secrete a variety of growth factors, neuroprotective cytokines, and chemokines in an autocrine or paracrine manner. These include HGF, VEGF, fibroblast growth factor, brainderived neurotrophic factor (BDNF), and nerve growth factor (NGF). All of these factors have been implicated in supporting the regeneration of damaged spinal cord tissues [50,51]. In addition, our team found that transplanted MSCs can also deposit FN, a well-known component of the ECM necessary for axonal growth. MSCs can also secrete Laminin and TGF-β in the injured spinal cord to promote the recovery [52]. Altogether, these changes result in a microenvironment that is conducive to regeneration [53].
Recently, an increasing number of studies confirmed that MSCs exert part of their therapeutic efficacy by secreting exosomes. These specialized membrane-coated nano-sized vesicles are secreted in large quantities by MSCs [54]. Exosomes contain biologically active molecules, various proteins, mRNA, transfer RNA, long noncoding RNAs, microRNAs, and even mitochondrial DNA. They affect cellular function via different routes [55]. Apart from MSCs a variety of cell types, including immune cells, can produce exosomes [56,57]. However, MSC-derived exosomes have unique characteristics; they exhibit immunomodulatory properties [58], elicit anti-inflammatory responses, and promote angiogenesis [59]. These actions of MSCs-derived exosomes replicate functions that the MSCs themselves provide [60]. Their natural cell membrane packaging protects the content of the exosomes against systemic degradation, making them suitable for systemic administration in living organisms [61,62]. Utilizing this feature attempts have been made to use MSC-derived exosomes for the treatment of SCI. Liu et al. [63] found that exosomes from bone-derived MSCs can repair traumatic SCI by suppressing the activation of A1 neurotoxic reactive astrocytes. Intravenously delivered MSC-derived exosomes can repair SCI by targeting M2-type macrophages at the site of injury [64]. The systematic administration of exosomes from MSCs can reduce apoptosis and inflammatory response, promote angiogenesis and functional recovery after SCI [65].
4. Transdifferentiation Into Neuron-Like Cells
Like other stem cells, MSCs have the potential to differentiate into a range of tissues. As progenitors of a mesenchymal lineage, they intrinsically differentiate into mesoderm-derived tissues such as chondrocytes, osteocytes, and adipocytes [66]. Whether MSCs can be reprogrammed to transdifferentiate into neurons remains controversial [67]. Decades ago, a number of studies suggested that such transdifferentiation was possible under specific conditions. In early studies of human MSCs, a chemical induction formula, containing a combination of dimethylsulfoxide (DMSO), butylated hydroxyanisole (BHA) and β-mercaptoethanol (BME) yielded cells that phenotypically appeared neuron-like [68]. However, this ‘neuronal differentiation’ of MSCs after chemical induction was an artifact due to cytotoxic cell changes. ‘Chemical induction,’ by BME, BHA, and DMSO, detergents, high sodium concentration, and extreme pH values, can shrink the cell bodies of MSCs within a few hours, leaving them with a neuronlike morphology. This change may even be accompanied by an increase in the level of neuronal markers, such as neuron-specific enolase and neuronal nuclear antigen. However, as reverse transcription-polymerase chain reaction experiments show, these changes in protein levels are not due to the up-regulated expression of corresponding mRNAs [69]. Changes induced by cytotoxicity happen rapidly, do not last long, and do not indicate a true transdifferentiation of MSCs. A more reliable approach to induce neuronal differentiation by MSCs involves the use of morphogens and/or neurotrophic factors. When MSCs were treated by epidermal growth factor, retinoic acid (RA), or a combination of RA and BDNF, they differentiated into neuronal cells expressing markers such as nestin and NeuN [70]. Using similar induction regimens, independent studies reported the neuronal differentiation potential of MSCs both in vitro and in vivo. A number of studies have shown evidence for action potentials being fired by MSC-derived neuron-like cells [71,72]. It was hypothesized that the increase of intracellular cAMP concentration may be a key factor responsible for this transdifferentiation of MSCs [73]. Our group was the first to report that a combination of neurotrophin-3 (NT-3) and RA could induce the differentiation of MSCs into neuron-like cells [74]. Furthermore, the proportion of neuron-like cells was greatly increased when NT-3 expressing modified Schwann cells were cocultured with MSCs that were genetically modified to express tyrosine kinase C (TrkC), the receptor for NT-3 [75]. By tissue engineering, we have constructed MSC-derived neural networks on 3-dimensional (3D) scaffolds. Neuron-like cells in this environment exhibit characteristic electrophysiological behaviors, including the firing of action potentials, and postsynaptic currents [76,77]. Furthermore, MSC-derived neuron-like cells formed on these scaffolds retained neuronal phenotypes and integrated into host neural circuits with synapse-like connections. More importantly, transplantation of MSC-derived neural network tissue into the injured spinal cord significantly improved motor function in the paralyzed limbs both in a rat and a canine model. We have ruled out the possibility of the morphological changes being cytotoxicity induced, as MSC-derived neurons survived for prolonged periods in culture (up to 14 days) and in vivo (up to 6 months) [76,77]. It has been argued that neuronal differentiation of MSCs is the result of cell fusion with host neurons, resulting in a neuronal phenotype of the fused cells [78]. However, using karyotyping, we excluded this possibility [76]. Nonetheless, MSC-derived neuronal cells do not possess all features of genuine neurons. The structure of synapses formed between MSC-derived neuronal cells or between MSC-derived neuronal cells and host neurons did show some of the characteristic features of a chemical synapse, when studied by electron microscopy. Thus, we prefer to call MSC-derived neuronal cells MSC-derived neuron-like cells, based on their functional similarity to neurons, rather than morphological reasons.
5. Perineurium-Like Differentiation
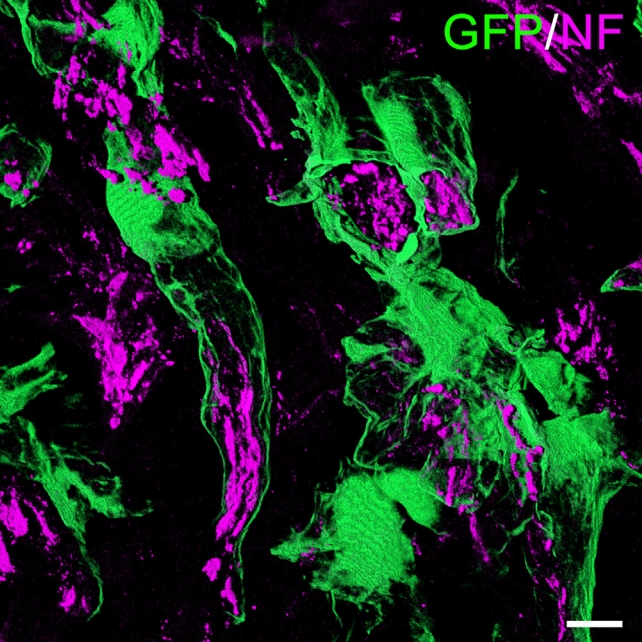
Our group recently identified a novel function of MSCs in treating SCI, the formation of perineurium-like sheaths protecting nerve fibers [8]. MSCs used in these experiments were derived from transgenic rats expressing green fluorescent protein (GFP). These cells were grafted into the transected spinal cord tissue of allogeneic rats, using a biocompatible scaffold. With the use of immunosuppression, the transplanted MSCs survived for up to 8 weeks and formed tube-like structures wrapped around nerve fibers at the injured site (Fig. 1). When examined by electron microscopy, nerve fibers covered by these tubes showed more intact morphology and contained less 4-hydroxynonenal and Nitrotyrosine. These molecules are key indicators of lipid peroxidation and protein nitration in an inflammatory microenvironment. MSCs that formed perineurium-like sheaths around neurons expressed BDNF, HGF, VEGF, glial cell line-derived neurotrophic factor (GDNF), and SOD3, a secreted antioxidant enzyme [79]. The latter indicates that perineurium-like MCS sheaths may protect nerve fibers from oxidative damage. Such MSC-derived perineurium-like sheaths also provide a physical barrier, isolating the enclosed nerve fibers from the deleterious microenvironment surrounding the injury. We performed a retrospective analysis of previously published studies transplanting MSCs to treat SCI, where donor MSCs survived beyond 3 weeks in the host spinal cord. The perineurium-like sheath structures were invariably visible on microscopic images, but were never identified or commented on by the original authors [80-87]. These findings indicate that the formation of perineurium-like sheaths by transplanted MSCs is not a fortuitous phenomenon. As discussed earlier, the microenvironment surrounding the injured cord causes excitotoxicity, oxidative damage, and is affected by deleterious immune responses. The formation of a perineurium-like sheath by MSCs can provide protection against these, aiding the regeneration of nerve fibers.
CURRENT CLINICAL TRIALS USING MESENCHYMAL STEM CELLS IN THE TREATMENT OF SPINAL CORD INJURY
A number of clinical trials using MSCs from autologous (mostly bone marrow-derived) or allogenic (mostly umbilical cord-derived) sources have been carried out on patients with SCI [88-90]. The safety of MSCs transplantation in humans was first demonstrated in 1995. In the initial studies, adherent stromal cells isolated and cultured from bone marrow samples of patients suffering from hematological malignancies were transfused back to the donors. This early demonstration of the safety of MSCs led to an increase in the number of translational studies and clinical applications of these cells [20]. To date, dozens of registered clinical trials have been initiated to investigate the MSCs-based treatment of SCI around the world, and this number is continuously growing. Despite some encouraging observations, the fate of donor MSCs inside the human body remains largely unknown due to the absence of biopsy data, the limited number of postmortem studies, and lack of relevant imaging. Animal studies suggest that these cells only survive for a short period within the spinal cord, typically less than 2 weeks [91]. In a clinical study, autologous bone marrow-derived MSCs were labeled with superparamagnetic iron oxide nanoparticles and injected intrathecally to treat a single SCI patient. Magnetic resonance imaging detected the focal accumulation of signal 48 hours after administration. This signal faded 2 weeks later and disappeared completely 1 month after transplantation [92]. Despite their low immunogenicity, transplanted MSCs fail to persist long-term in vivo without any immunosuppression, and are thought to provide therapeutic benefits via a “Touch-and-Go”/“Hit-and-Run” mechanism [42,93 94]. However, with most of the conducted clinical studies being phase I, I/II, or II trials, the therapeutic efficacy of MSCs still needs further investigation [95]. A phase III clinical trial showed limited efficacy of a single dose of autologous MSCs in treating chronic SCI [96]. However, the administration of multiple doses of MSCs was effective in a previous study during long-term observation [89], suggesting that the therapeutic efficacy of MSCs may be dose-dependent. Thus, any intervention that increases the number of donor MSCs or promotes their survival, may produce better clinical outcomes. However, it appears that achieving this will remain a technical challenge in the treatment of SCI. There are several factors affecting therapeutic efficacy: (1) Heterogeneity of MSCs from different tissues: MSCs from the umbilical cord matrix, adipose tissue, or bone marrow show differences in their ability to inhibit peripheral blood B cells, T cells, and NK cells [97]. There are similar differences in the differentiation and proliferation of MSCs derived from different tissues [98,99]. (2) Differences in passage numbers: MSCs lose their stem cell characteristics and their telomeres get gradually shorter as the number of in vitro passages increases [100]. Although the use of cells from an early passage is recommended for clinical use, it is difficult to control for changes in this parameter when comparing trial data. (3) Differences in cell preparation processes: The lack of reproducibility in producing the transplanted MSCs in vitro may also lead to different results [101]. (4) Different routes of administration: Currently, MSCs are administered via intrathecal injection, intravenous dosing, intramuscular injection, injection in situ, or being delivered using scaffolds [102]. Intrathecal, intravenous, and intramuscular administration is less invasive but relies on the homing effect of MSCs [103]. According to animal studies, intrathecal injections appear more effective than intravenous administration [104]. Injecting MSCs directly into the spinal cord contusion cavity seems beneficial for the resolution of the glial scars and bridging axon regeneration. Thus, in situ injection to the intramedullary injury area may also achieve better outcomes [105]. More work is certainly needed to establish the optimal route of administration. (5) Different dose ranges: In various trials, the frequency of administration and the injected number of MSCs varied widely. Cell doses range from 0.5×106 to 10×106/kg, or even higher were tried using single or multiple dosage regimes [106]. (6) Individual differences: Notable differences were observed in the response of individual patients. Some appear to show significant improvements while others do not [107]. These differences could be due to several poorly quantifiable factors, including the differing extent and location of the damage, the patient’s age, prior physical condition, and the presence or absence of underlying diseases. Currently, reports on completed phase III clinical trials are scarce. Although weaknesses in study design have triggered criticism and debates [108-110], the efficacy of MSCs in the treatment of SCI demonstrated in a trial led by Stemirac is promising. In summary, although the safety of MSCs in clinical has been verified, their efficacy in treating SCI remains controversial. Data available from current clinical trials are clearly inadequate to draw final conclusions from. Much more effort will be needed before the large-scale application of MSCs for the treatment of SCI will become reality.
APPROACHES TO ENHANCE THE EFFICACY OF MESENCHYMAL STEM CELLS IN SPINAL CORD INJURY REPAIR
The fact that MSCs can target multiple pathological changes associated with the secondary injury after SCI promises tantalizing benefits. Although the efficacy of MSCs for the treatment of SCI patients is still being studied, several strategies are being developed to enhance their effectiveness in pre-clinical studies. This work may produce new MSC-derived products with improved therapeutic characteristics.
1. Preconditioning MSCs
IFN-γ pretreatment could promote the secretion of soluble factors responsible for the immunosuppressive and immunomodulatory effects of the transplanted cells [106,111]. Hypoxic preconditioning followed by reoxygenation for 30 minutes improved the ability of MSCs to proliferate and migrate [112]. Research showed that culturing the cells with the oxygen level reduced to 1% could significantly increase their survival and angiogenic capacity while reducing their sensitivity to the ischemic microenvironment of the damaged spinal cord. These effects were achieved without changing the biological behavior, immunophenotype or karyotype of MSCs [113]. Transplantation of hypoxia preconditioned MSCs enhanced their protective effect during spinal cord ischemia/reperfusion injury [114]. It appears that these therapeutic benefits are due to the up-regulation of the secretion of cytokines, such as HGF and VEGF [115]. In other experiments, preconditioning with sevoflurane, an inhaled anesthetic, improved the survival and therapeutic potential of MSCs during serum deprivation and hypoxia. This was mediated through the up-regulation of HIF-1α, HIF-2α, VEGF, and p-Akt/Akt attenuating the initiation of apoptosis and the loss of mitochondrial membrane potential [115]. Other small molecules, such as IFN-γ, IL-1β, and lipopolysaccharide have also been reported to enhance the immunomodulatory properties of MSCs, causing prominent PGE-2 secretion [116].
2. Three-Dimensional Culture
Biomaterial scaffolds can provide a 3D milieu for embedded cells, conferring enhanced biological function during SCI repair. The physical properties of such scaffolds may alter the functional status of the seeded MSCs. The stiffness of the biomaterials can affect the morphology, proliferation, and differentiation of MSCs. MSCs cultured in alginate hydrogels of varying stiffness showed changes in gene expression profiles. The production of inflammatory regulators, including IDO1 and PGE-2, increased in stiffer matrices [117]. Other features, such as surface characteristics, or the pore size fundamentally changed the biological function of MSCs [118,119]. Furthermore, chemical modification of the scaffold may introduce new features. A hydrogel scaffold modified with the bioactive peptide PPFLMLLKGSTR significantly improved the survival and adhesive growth of MSCs in 3D cultures in vitro. This translated into better hindlimb motor function in the treated animals [120]. Coculturing MSCs with immune cells in the 3D matrix also affected their immunomodulatory potential [121]. MSCs being cultured in a polystyrene scaffold produced more anti-inflammatory cytokines, such as PGE-2 and tumor necrosis factor-stimulated gene 6. At the same time the secretion of proinflammatory cytokines, such as IL-6, monocyte chemotactic protein-1, macrophage colony-stimulating factor and receptor activator of nuclear factor κ-B ligand were reduced in cocultures with macrophages [122].
3. Genetic Modification
Genetic modification of MSCs could introduce new features that are useful for therapeutic purposes [123]. Transplanted MSCs genetically modified to produce insulin-like growth factor-1 have shown better survival with enhanced immunoregulation. This promoted myelination, leading to significant functional improvement after SCI [124]. MSCs modified to produce VEGF and GDNF improved angiogenesis in the injured area. This, in turn, increased the survival of transplanted cells and the extent of axonal regeneration in a rat model [125]. MSCs transduced with the BDNF gene protected the spinal cord tissue more, inhibited glial scar formation, and alleviated inflammatory responses [126]. Grafting spheroids formed by MSCs expressing BDNF promoted the retention of myelinated axons in the area of SCI and led to a significantly enhanced recovery of hindlimb motor function in a mice model [127]. Our group cocultured NT-3 expressing Schwann cells and MSCs expressing its receptor, TrkC, in a gelatin sponge scaffold. The constructed MSC-derived neural network tissue was used to repair SCI in rat and canine models [76,77]. We found that combining tissue engineering with the genetic modification of MSCs improved neural differentiation and helped the functional recovery of animals. However, the use of vectors necessary for genetic modifications, such as modified adenovirus, lentivirus, and adeno-associated virus particles, causes concerns about the safety of such genetically modified MSCs in human trials. Thus, although promising, the safety of genetically modified MSCs should be fully investigated to reduce potential harm. If their safety could be guaranteed, genetic modifications of MSCs may enhance their therapeutic utility in the treatment of SCI.
4. Combination With Neurorehabilitation
MSCs transplantation has been combined with various forms of neurorehabilitation therapy. There are reports that combining MSC transplantation with treadmill training [128], electrical stimulation [129], electroacupuncture [130], transcranial magnetic stimulation (TMS) [131], ultrashort wave therapy [132,133], and swimming training [134] resulted in improved therapeutic outcomes. Several theories were put forward to explain the increased benefits of these combined treatment regimes. (1) Tissue sparing: The combination of MSCs and TMS displayed synergistic effects on alleviating SCI-induced spinal cord lesions and neuronal apoptosis in a rat model. Increased GAP-43, NGF, and BDNF expression levels, downregulated glial fibrillary acidic protein (GFAP) expression, and reduced activation of the Raf/MEK/ERK signaling pathway was reported with this combined treatment [131]. MSCs transplantation together with physical activity such as treadmill training showed better tissue preservation, fewer microcavitations, and reduced degeneration of nerve fibers after SCI [128]. (2) Promoting donor MSCs survival: Electrical stimulation and acupuncture can promote the survival and differentiation of MSCs in rats following SCI. We have previously found that electrical acupuncture could efficiently promote the survival and differentiation of bone marrow-derived MSCs. This could lead to better axonal regeneration and locomotor recovery [130]. MSC transplantation combined with electroacupuncture therapy can also improve Basso-Beattie-Bresnahan scale scores and evoked motor potentials. The number of neurofilament-positive and Biotinylated dextran amine-labeled axons increased, leading to improved outcomes [135]. There is evidence that treadmill training improves the survival of neural precursor cells in the post-SCI microenvironment, through the involvement of MSCs [136]. (3) Modulation of neuroinflammation and reduction of glial scarring: Other experiments combined human umbilical cord MSCs with ultrashort wave therapy in SCI. This combination improved motor function, and decreased the number of infiltrating CD3+ T cells, while decreasing microglia and astrocyte inflammation [132,133]. Moreover, decreased GFAP and chondroitin sulfate proteoglycans expression was detected in this combination treatment group [135]. What is the mechanism behind the improved outcomes when MSCs transplantation is combined with neurorehabilitation? Our study showed that electroacupuncture increased the secretion of NT-3 from the injured spinal cord tissue. This, in turn, can promote the survival, differentiation, and migration of grafted MSCs expressing the TrkC gene [137]. Subsequently, we explored the mechanism that allows electroacupuncture to promote the increased secretion of NT-3 by the activation of the CGRP/RAMP1/αCaMKII pathway [138]. In a recently published review, we summarized the potential mechanisms explaining the benefits of combining electroacupuncture with MSCs transplantation in the treatment of SCI [139].
CONCLUSIONS AND PERSPECTIVE
The exact mechanism by which MSCs improve outcomes after SCI remains to be explored further. This work will offer further insights into enhancing their safe and effective therapeutic use. In this review, we have shown that MSCs have multiple functional properties that can target a variety of pathological consequences of SCI (Fig. 2). One of these is a novel finding, the perineurium-like differentiation of MSCs, that warrants further investigation. Future basic research needs to be focused on developing MSC-based treatment strategies that improve the efficacy of treatment in SCI.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
This research was supported by grants from the Chinese National Natural Science Foundation (Grant No. 31900975 to Y. H. Ma); the Natural Science Foundation of Guangdong Province (Grant Nos. 2018A030310110 and 2020-A1515011537 to Y. H. Ma); the Start-up Fund Projects of Guangzhou First People’s Hospital (Grant No. KYQD20210017 to Y. H. Ma) and the Startup Grant of Guangdong Hospital of Chinese Medicine (Grant No.2022KT1032) to Xiang Zeng.
Author Contribution
Conceptualization: YM, XZ; Funding acquisition: YM, XZ; Writing - original draft: YM; Writing - review & editing: QL, YD, IH, XZ