Spine Surgical Robotics: Current Status and Recent Clinical Applications
Article information
Abstract
With the development of artificial intelligence and the further deepening of medical-engineering integration, spine surgical robot-assisted (RA) technique has made significant progress and its applicability in clinical practice is constantly expanding in recent years. In this review, we have systematically summarized the majority of literature related to spine surgical robots in the past decade, and not only classified robots accordingly, but also summarized the latest research progress in RA technique for screw placement such as cervical, thoracic, and lumbar pedicle screws, cortical bone trajectory screws, cervical lateral mass screws, and S2 sacroiliac screws; guiding targeted puncture and placement of endoscope via the intervertebral foramen; complete resection of spinal tumor tissue; and decompressive laminectomy. In addition, this report also provides a detailed evaluation of RA technique’s advantages and disadvantages, and clarifies the accuracy, safety, and practicality of RA technique. We consider that this review can help clinical physicians further understand and familiarize the current clinical application status of spine surgical robots, thereby promoting the continuous improvement and popularization of RA technique, and ultimately benefiting numerous patients.
INTRODUCTION
In the past decades, the world’s population is aging rapidly. With the change in people’s work and lifestyle, as well as the widespread use of electronic devices, the number of people around the world suffering from spine-related disorders is increasing annually, which has become one of the most common diseases [1,2]. Surgical treatment is regarded as an important treatment option for spine disorders, and can achieve satisfactory results. However, traditional spine surgery often relies on surgeons’ experience and intraoperative imaging devices, which has many disadvantages, such as high operation risk, low precision, and being difficult to be popularized due to complex surgical procedures. The advent of spine surgical robotics offers a new way to remedy these disadvantages [3-5]. With the rapid advances in science and technology, spine surgical robots are constantly evolving and widely used in clinical practice, which could improve surgical safety [6]. In this review, we systematically outline recent progress in the clinical application of spine surgical robotic platforms in various spine surgical procedures, and summarize their advantages and disadvantages, in order to provide surgeons with more in-depth understanding of robot-assisted (RA) technique in spine surgery, and further promote its popularization.
CLINICAL CLASSIFICATION OF SPINE SURGICAL ROBOTS
Since Nathoo et al. [7] first proposed a 3-type classification system for spine surgical robots based on the robot-surgeon interaction in 2005, a variety of spine surgical robots have flourished in the last decade. According to the spatial location of a spine surgical robot and an operator during surgery, we classify surgical robots into on-site-controlled robots and remote-controlled robots.
1. On-Site-Controlled Robots
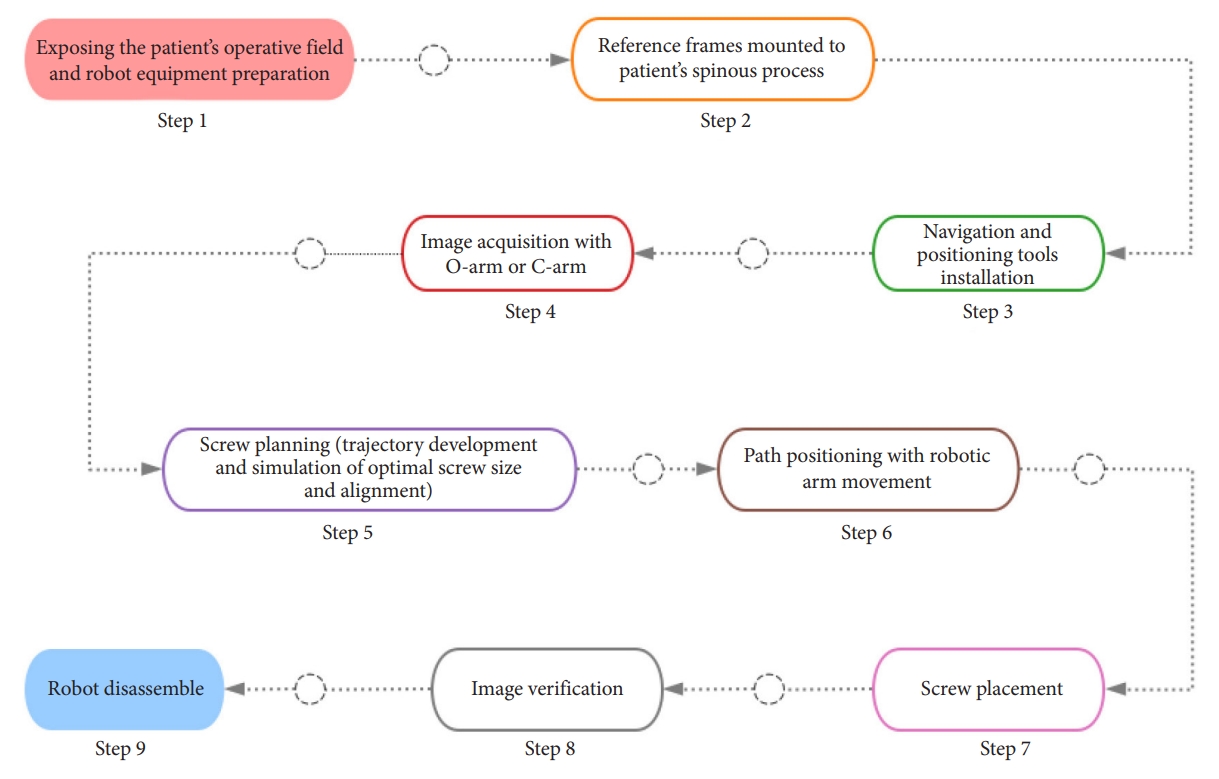
On-site-controlled robots are robots used on-site, which can collaborate with an operator in the same space, allowing them to work together to complete the surgical procedures (Fig. 1). The currently available commercial spine surgical robotic systems including TiRobot (TINAVI Medical Technologies Co. Ltd., Beijing, China), Mazor (Mazor Robotics Ltd., Caesarea, Israel), Davinci (Intuitive Surgical, Sunnyvale, CA, USA), ROSA (Zimmer Biomet Robotics, Montpellier, France), Excelsius GPS (Globus Medical, Inc., Audubon, PA, USA), and Orthbot (Xin Junte, Shenzhen, China) series are on-site-controlled robots [8,9]. On one hand, these robotic systems allow surgeons to complete a rigorous surgical plan before the operation, then the robot executes the plan under the supervision of the surgeon during the operation. On the other hand, the robotic systems can also be used as surgical instruments to provide stable mechanical arm support, and enable surgeons to perform the operation with more accuracy and precision, thus avoiding the influence of surgeons’ physiological factors on surgical precision [7,10].
2. Remote-Controlled Robots
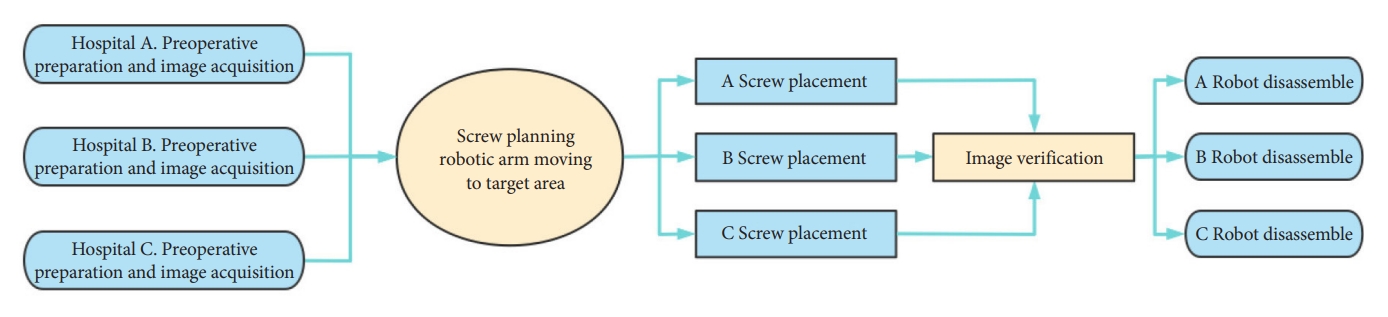
Remote-controlled robots are installed adjacent to the operating table, and remotely controlled by an operator seated at the surgeon console in a control room. The console consists of a monitor that provides a live display of the operating area through a camera mounted above the surgical table. The operator can control the robot instruments remotely to perform the surgery through a haptic feedback tool on the control panel, receive realtime feedback via the camera to guide surgical procedures intraoperatively, and complete the remote surgery successfully. When a patient and an operator are not in same region, the robotic system allows an operator to perform surgery on the patient by receiving instructions from the surgeon console remotely via internet connection (Fig. 2). Tian et al. [5] controlled TiRobot system remotely via 5G network to perform spinal fusion surgery in 12 patients with spine disorders, a total of 62 pedicle screws were placed, the pedicle screw placement accuracy classified using Gertzbein-Robbins criteria (grade A, screw placed within the pedicle without cortical breach; grade B, less than 2 mm breach; grade C, 2- to 4-mm breach; grade D, more than 4-mm breach) showed that 59 screws were grade A, and 3 screws were grade B, the accuracy of screw placement was as high as 95%, confirming the accuracy and reliability of 5G remote RA pedicle screw placement in spinal surgery. However, the sample size of the above-mentioned study is small, and the surgical procedures may be affected by the performance of the networks, thus resulting in failure of robotic procedures [11], so multicenter and large-sample clinical studies are needed to confirm the reliability of 5G remote RA spinal surgery. We consider that the continuous optimization of remote-controlled robotic systems can effectively address the problem of uneven distribution of medical resources.
CLINICAL APPLICATION SCENARIOS FOR SPINE SURGICAL ROBOTS
Currently, the use of spinal surgical robots mainly involves screw placement, guiding puncture and complete resection of spinal tumor tissues [12-14]. In terms of screw placement, spine surgical robots have been used for the placement of cervical, thoracic, and lumbar pedicle screws, cortical bone trajectory (CBT) screws, cervical lateral mass screws, and S2 sacroiliac screws [15-17]. RA placement of cervical, thoracic, and lumbar pedicle screws is currently the most widely used in spine surgery [18]. Targeted puncture is an essential step in vertebroplasty and percutaneous transforaminal endoscopic discectomy (PTED), robotic assistance can obviously improve puncture accuracy [19]. Due to the fact that the anatomy of spinal tumor tissues is complex and the surgical field vision for tumor removal is limited, it is quite difficult to excise spinal tumor tissues completely, and avoid vascular and nerve damage [20]. The use of robotic visualization in clinical settings could markedly improve the success rate of complete tumor resection while minimizing trauma [21]. Additionally, some scholars have conducted experimental studies on RA decompressive laminectomy, but no clinical application has emerged [22].
1. RA Spinal Implant Placement
1) RA thoracolumbar screw placement
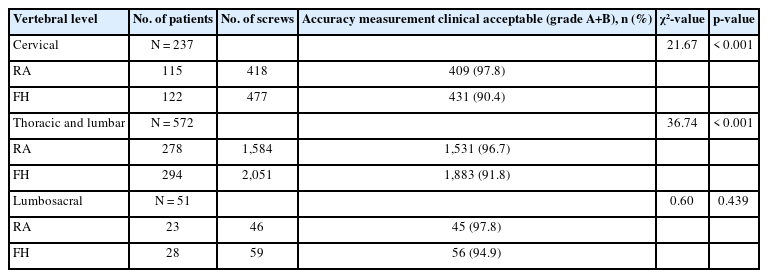
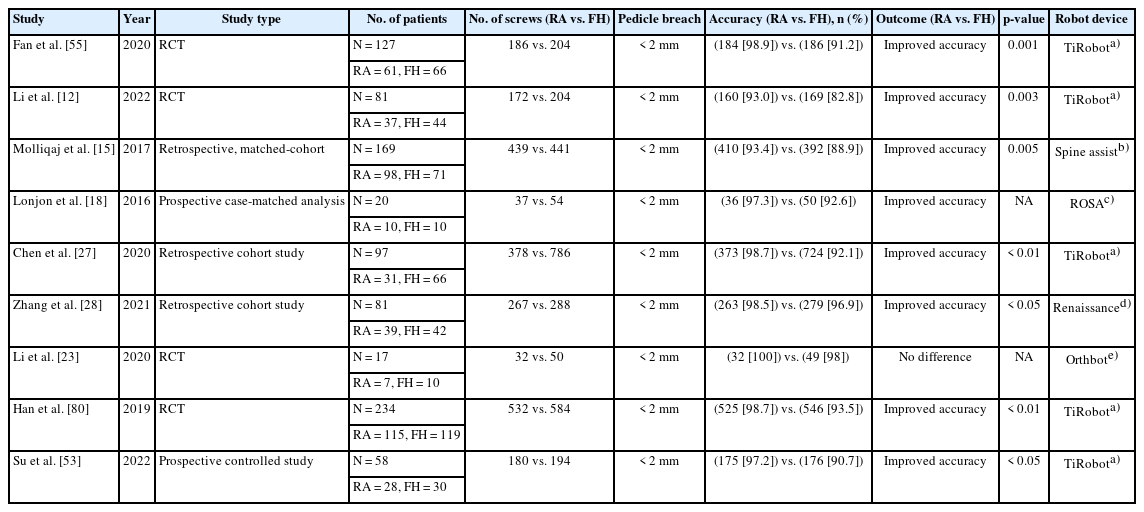
At present, the primary application of robotics in spine surgery is pedicle screw placement, followed by CBT screw placement. A number of studies have confirmed that RA thoracolumbar pedicle screw placement has obvious advantages over traditional fluoroscopic techniques, resulting in an accuracy of 93%–100% [12,15,18,22,23]. Zhang et al. [24] performed RA and freehand (FH) pedicle screw placement in 36 patients, and found that RA screw placement exhibited significantly higher accuracy than FH technique (98.1% vs. 90.3%). A multicenter, large-sample study of 2,046 patients undergoing RA surgery conducted by Wei et al. [25] compared the accuracy of thoracolumbar pedicle screw placement assisted by various types of spine surgical robots, they found that TiRobot showed the highest screw placement accuracy, followed by Mazor Renaissance, while Orthbot, Mazor SpineAssist, ROSA robotic systems were similar to conventional technique in terms of screw placement accuracy. A cohort study by Khan et al. [26] included 22 patients who underwent RA CBT screw placement using the Mazor X robot and 18 patients who underwent computed tomography (CT) navigation-guided CBT screw placement, the results showed that in the CT-navigation group, a total of 74 CBT screws were inserted, 69 of which were grade A and 5 were grade B, with an accuracy of 93%, while in the Mazor X robot group, a total of 92 CBT screws were inserted, which were all marked as grade A, with an accuracy of 100%. A randomized controlled study by Li et al. [12] included 81 patients with lumbar degenerative diseases who underwent CBT screw fixation using RA (n = 37) and FH techniques (n = 44), they found that screw placement accuracy was 93% (160 of 172) in the RA group, which was significantly higher than the FH group (83%, 169 of 204) (p = 0.003). Furthermore, the number of screws without superior facet joint violation (FJV) in RA group (78.4%, 58 of 74) is more than in FH group (63.6%, 56 of 88) (p = 0.041). We included 8 high-quality clinical studies comparing 2 screw placement techniques [12,15,18,24,26-29]. Among them, the RA group had 278 cases, with a total of 1,584 pedicle screws implanted, while the FH group had 294 cases, with a total of 2,051 pedicle screws implanted. The analysis results showed that the RA group (96.7%) had significantly better screw placement accuracy than the FH group (91.8%), and the difference was statistically significant (Table 1) (p < 0.001). The specific applications of RA screw placement in complex thoracolumbar spine surgery and minimally invasive spine surgery are summarized below:
(1) The use of robots for the treatment of scoliosis: Currently, the use of RA techniques for surgical treatment of adolescent and adult patients with scoliosis has received satisfactory clinical results [30]. Several previous studies have demonstrated high accuracy, low screw misplacement rate, and excellent surgical outcomes of RA pedicle screw placement in the treatment of adolescent idiopathic scoliosis [31-34]. However, robotic systems are rarely used in the surgical treatment of adult degenerative scoliosis. Chen et al. [27] conducted a study on 97 patients with adult degenerative scoliosis, and treated them with RA surgery (n = 31) and conventional open surgery (n = 66), the results showed that the RA group had less intraoperative blood loss, and exhibited significantly greater screw placement accuracy when compared to the conventional group (98.7% vs. 92.2%).
(2) The use of robots in revision thoracolumbar spine surgery: Due to disruption of local anatomical structure and the disappearance of anatomical landmarks caused by failed previous surgery, and the presence of severe lumbar degeneration in some patients, the probability of screw misplacement and postoperative complications during revision surgery has greatly increased. CBT screws are particularly suitable for use in revision surgery because CBT screws have special screw structure (small diameter and length, with tight threads) and can be inserted from the inner and inferior aspects to the outer and superior aspects through the pedicle, which allows close screw contact with cortical bone of the pedicle, and results in low rate of FJV [16,35]. Rho et al. [36] reported the use of RA CBT screw placement using Mazor X robot in the treatment of a patient with adjacent segment degeneration after thoracolumbar surgery, they inserted CBT screw again in the same pedicle without removal of the original pedicle screw, the results showed that bilateral screw placement was completed successfully, without the occurrence of bony destruction, RA CBT screw placement was more minimally invasive and had shorter hospital stay when compared to traditional revision surgery. Zhang et al. [28] conducted a retrospective study on 81 patients who underwent lumbar spine revision surgery via a posterior approach, 39 patients received pedicle screw placement using Mazor Renaissance, the remaining 42 patients received conventional FH pedicle screw placement, they found that the number of screws inserted was 267 in the Mazor Renaissance group and 288 in the conventional FH group, lumbar pedicular screw placement accuracy was 93% in the Mazor Renaissance group, and 87.1% in the conventional FH group. The rate of proximal FJV (grades 1, 2, and 3 assessed according to the Babu scale) [16] was 24% in the conventional FH group, and only 6% in the Mazor Renaissance group. Several clinical studies have documented higher accuracy, less screw adjustment and fluoroscopy times, less intraoperative blood loss, as well as low rate of superior FJV for RA pedicle screw placement in lumbar revision surgery [28,37].
(3) The use of robotics in anterior lumbar spine surgery: The DaVinci robotic system is now one of the most widely used surgical robotic systems worldwide, which has been used in gastrointestinal, urological, gynecological and spine surgeries [38-40]. Due to the magnification of the operative field view and flexibility of the mechanical arm, the DaVinci robotic system offers unique advantages for use in anterior lumbar spine surgery. In a case report, Lee et al. [41] used the Davinci robot for the first time to assist surgeons in performing laparoscopic anterior lumbar interbody fusion, and found that the Davinci robot has obvious advantages in visualization of the intervertebral disc and surrounding tissues during surgery. Lippross et al. [42] used the Davinci robot to treat a fracture of L3, the robot equipped with a high-definition 3-dimensional (3D) display screen enable surgeons to perform surgeries with high safety. Although surgeons can control the endoscope and perform the procedures without an assistant during the operation, the robotic arm are moving counterwise on the screen, special training of surgeons is necessary to perform the surgery counterwise. Balboni et al. [43] used the Excelsius GPS robotic navigation system to assist with anterior lumbar total disc replacement surgery, and found that the implants inserted with the assistance of the Excelsius GPS robotic system showed less screw displacement from the center line, and greater screw placement accuracy compared with the non-robotic navigation group. Currently, only the 2 aforementioned robotic systems have been reported for use in anterior lumbar spine surgery.
(4) The use of robotics in minimally invasive spinal fusion surgery: Minimally invasive transforaminal lumbar interbody fusion (MIS-TLIF) is an ideal surgical procedure for the treatment of single-segment lumbar spondylolisthesis, and can further reduce iatrogenic injury to low back muscles, but percutaneous pedicle screw placement is more likely to invade the superior articular process, leading to spontaneous facet joint fusion, the development of adjacent segment diseases, and increased rates of revision surgery. In recent years, TiRobot-assisted MIS-TLIF is shown to be associated with high accuracy of pedicle screw placement, because it can assist in developing preoperative planning, include clear visualization of screw trajectory and selection of ideal screw diameter and length [44,45]. It has been shown that RA pedicle screw placement in TLIF had a screw loosening rate of only 4.3%, which was lower than conventional surgery (10.2%), it also had lower rates of pedicle wall penetration and proximal FJV [29,46-48]. Good et al. [49] have demonstrated the benefits of RA MIS-TLIF using Mazor robot, including a 5.8-fold reduction in the risk of complications, an 11-fold reduction in the risk of revision surgery, and an 80% reduction in fluoroscopic time per screw. A prospective cohort study by Chang et al. [50] compared the clinical outcomes of RA percutaneous endoscopic TLIF (n = 26) and conventional MIS-TLIF (n = 32) for the treatment of lumbar spondylolisthesis, they found that the accuracy of pedicle screw placement with robotic assistance was up to 96.2%, the RA technique was more minimally invasive with faster postoperative recovery. Chen et al. [46] included 104 patients with lumbar degenerative disease, and performed RA MIS-TLIF (n = 52) and FH open TLIF (n = 52), a total of 208 pedicle screws were implanted in each group, respectively. Based on the Gertzbein-Robbins grading standard, 192 screws placed were grade A, the accuracy of screw placement was 98.6% in the RA group, whereas 161 screws were grade A with an accuracy of 77.4% in the FH group. Compared to the preoperative scores, the postoperative visual analogue scale scores decreased 4.83 points in the RA group, and 4.77 points in the FH group. The mean Oswestry Disability Index improvement was 29.21% in the RA group and 29.40% in the FH group. The results indicates that robotic assistance offers high accuracy in screw placement, and induces less tissue damage during surgery, thus enabling better prognosis for patients [51,52].
2) RA lower cervical pedicle screw placement
The robotic systems were initially used only for the placement of thoracolumbar pedicle screws [15,18]. With continuous optimization of the systems, the new generation of robots has been adapted to assist with cervical screw placement [53,54]. A prospective randomized controlled study by Fan et al. [55] performed cervical lateral mass screw placement in 127 patients, 61 patients underwent RA screw placement, the remaining 66 patients underwent FH screw placement, they found that a total of 186 and 204 screws were inserted, respectively, in the RA and FH group, RA cervical lateral mass screw placement had higher accuracy (87.6% vs. 60.8%, p = 0.001) and less blood loss (200 mL vs. 350 mL, p = 0.002) when compared to conventional FH technique. A meta-analysis by Beyer et al. [56] showed that among a total of 482 cervical screws placed using a spinal surgical robot, 378 were cervical lateral mass screws, the accuracy of screw placement was up to 97.7%, with an average screw deviation of 0.95 mm. We included 3 high-quality clinical studies comparing 2 techniques [53-55], a total of 418 cervical screws were implanted in RA group, and 477 screws were implanted in FH group. The analysis results showed that the RA group (97.8%) had significantly better screw placement accuracy than the FH group (90.4%), and the difference was statistically significant (Table 1) (p<0.001).
3) RA upper cervical screw placement
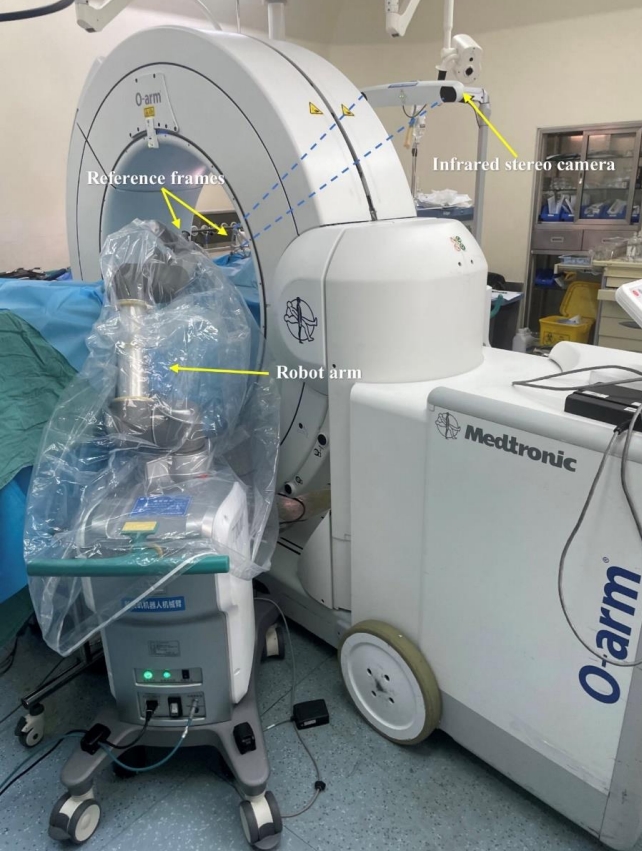
It is more difficult to place screws into the upper cervical spine due to its proximity to important blood vessels and nerves, the surgical risk is high. At present, only few studies reported the application of robotics in upper cervical screw placement, there is a lack of multicenter clinical studies with large samples to confirm its effectiveness [57,58]. Tian [59] reported for the first time the application of RA upper cervical spine surgery, they performed posterior C1–2 transarticular screw fixation using the TiRobot system in a patient, postoperative CT revealed that deviation of the planned screw trajectory and actual trajectory was only 0.8798 mm. Since then, TiRobot can be used to assist in screw placement in spine surgeries of all segments (Fig. 3). Asuzu et al. [60] performed percutaneous C2 pedicle screw placensed using Mazor X robot in a patient with a Levine-Edwards type II C2 fracture who had failed conservative treatment, postoperative x-ray confirmed good screw placement and satisfactory fracture reduction. During 6-month follow-up period, the patient’s neck symptoms subsided completely, and the range of motion was normal with no special discomfort. The clinical application of other robotic systems in upper cervical spine surgery has not been reported.
4) RA sacroiliac screw placement
The S2-alar-iliac (S2AI) technique, first proposed in 2007 [61], has been shown to provide strong internal fixation at the lumbosacral junction, which is particularly suitable for long-segment spinal fusion in patients with spinal deformity [62,63]. However, S2AI screw placement is difficult, because the screw trajectory is adjacent to important blood vessels and nerves, and the screws need to traverse 3 layers of dense cortical bone of the sacroiliac joint, so complications such as screw displacement and breach of cortical bone are more likely to occur. Many studies have shown that RA techniques can increase accuracy and reduce the complexity of S2AI screw placement [17,64]. Lee et al. [65] used the MazorX robotic (Fig. 4) system to insert 65 S2AI screws, and found that accuracy of screw placement (grade A) was 93.8%, without the occurrence of intraoperative neurologic, vascular, or visceral complications. A retrospective cohort analysis of 51 patients who underwent S2AI screw placement by RA (n = 23) and conventional FH technique (n = 28) conducted by Shillingford et al. [66] showed that a total of 105 S2AI screws were placed, including 59 screws in the FH group and 46 screws in the RA group, robot assistance showed improved accuracy of S2AI screw placement compared to the FH group (97.8% vs. 94.9%), however, after statistical analysis, there was no difference between the 2 methods (Table 1) (p = 0.439). For patients with lumbosacral metastatic lesions, S2AI screw cannot be placed due to severe destruction of the sacrum, and only iliac screws can be placed. Park et al. [67] successfully performed RA percutaneous iliac screw fixation in 2 patients with lumbosacral metastatic lesions, and confirmed the feasibility and safety of robotic assistance in percutaneous iliac screw placement.
2. RA-Targeted Puncture and Placement of Endoscope via the Intervertebral Foramen
Currently, percutaneous vertebroplasty (PVP) and percutaneous kyphoplasty (PKP) are mainstream approaches for treating osteoporotic vertebral compression fractures (OVCF). However, conventional PVP and PKP both have the disadvantages of low puncture accuracy, high intraoperative radiation exposure and high risk of bone cement leakage. In a retrospective cohort study, Wang et al. [68] included 60 patients with single-segment OVCF of the thoracolumbar spine who underwent unilateral PKP with the use of TiRobot robotic system (n = 30) and the traditional approach (n = 30), they found that TiRobot group had significantly less number of intraoperative fluoroscopies compared with the traditional group (9.80 ± 1.74 vs. 31.53 ± 5.72), suggesting that the use of TiRobot robotic system is associated with decreased radiation exposure. During the postoperative follow-up period, bone cement leakage was noted in 23% (7 of 30, including 3 patients who developed neurological symptoms due to cement leakage) of the patients in the traditional group, while bone cement leakage was not observed in the TiRobot group. The use of TiRobot robotic system in PKP can markedly reduce puncture deviation, radiation exposure, and bone cement leakage risk, resulting in better prognosis for patients. A comparison study of TiRobot-assisted PKP versus traditional fluoroscopy-assisted PKP in the treatment of multi-segmental OVCF performed by Lin et al. [69] showed that the optimal puncture rate was 91.5% (Gertzbein and Robbins grade A) in the TiRobot group, which was only 66.9% in the traditional group, suggesting that TiRobot-assisted PKP has many advantages, such as high puncture accuracy, shorter time of working channel establishment, less radiation exposure, lower bone cement leakage rate, uniform bone cement distribution in the vertebral body. In recent years, the incidence of Kummell disease (KD) has increased gradually, vertebroplasty remains the mainstream treatment for KD, but this technique may cause a catastrophic complication of postoperative bone cement displacement. A comparison clinical study of RA versus conventional fluoroscopy-guided pediculoplasty combined with vertebroplasty in the treatment of patients with KD without neurological symptoms led by Wang et al. [19] showed that the one-time success rate of RA puncture was up to 95.4%, RA pediculoplasty can effectively avoid bone cement displacement, lessen radiation exposure, achieve satisfactory vertebral body height restoration and kyphotic deformity improvement. With robot assistance, the first step of PVP, i.e., puncture can be performed precisely. In terms of the second step of PVP, i.e., the infusion of bone cement, Neumann et al. [70] developed a robotic system that can be remotely controlled to inject bone cement, along with a cold passive exchanger that slows down the cement curing and an active exchanger that controls the injected cement temperature. With the use of RA puncture combined with bone cement infusion controlled by the above-mentioned robotic system, vertebroplasty is expected to be performed automatically.
PTED is a common minimally invasive surgical treatment for lumbar disc herniation [71]. The percutaneous targeted puncture is the first step for PTED, which is also a difficulty. Precise puncture to the target lesion can not only reduce radiation exposure, avoid injury caused by repeated puncture, but also guide the placement of endoscope, thereby providing a better surgical field and facilitating adequate surgical decompression [13]. Yang et al. [72] performed 2-dimensional (2D) fluoroscopy-guided RA PTED and C-arm fluoroscopy-guided PTED in 102 patients, respectively, and found that 2D fluoroscopy-guided RA PTED had several advantages compared with C-arm fluoroscopy-guided PTED, including fewer puncture times (1.20 ± 0.42 vs. 4.84 ± 1.94), fewer fluoroscopy times (10.49 ± 2.16 vs. 17.41 ± 3.23), and shorter operation time (60.69 ± 5.63 minutes vs. 71.19 ± 5.11 minutes), suggesting that RA PTED allows the operator to plan the puncture path accurately and precisely, makes the targeted puncture easier to perform, reduces the number of punctures and fluoroscopies, shortens the operation time, optimizes the operation process, and reduces complications. The Kambin triangle has been considered an “anatomical corridor” and common entry point for lumbar interbody fusion, accessing Kambin’s triangle in a less invasive manner indicate that the surgical procedure is more minimally invasive [73]. Dalton et al. [74] designed a single-center clinical study involving 10 patients to explore the clinical application of RA access into Kambin triangle during lumbar interbody fusion, they found that the Kambin triangle were successfully accessed, interbody cage was placed successfully and safely with minimal invasiveness.
3. RA Complete Tumor Resection
Since spinal tumors are adjacent to blood vessels and nerves, and severely obscured by the surrounding bony structures, so it is difficult for surgeons to precisely separate the relevant tissues and ligate the blood vessels within the limited surgical field, this may easily lead to more bleeding and severe damage to the surrounding tissues. The DaVinci surgical robot can offer highly magnified views (up to 15 times) of the surgical area, with greater freedom of movement, which provides a guarantee for complete tumor resection, enables the operator to precisely distinguish the surrounding tissues in a narrow space and operate with more precision, thus greatly enhancing the safety of the operation [20]. A clinical study by Pu et al. [75] used DaVinci surgical robot to assist in resection of tumor tissues in 12 patients with presacral nerve sheath tumors, they found that the magnified view and flexible arm of the DaVinci surgical robot allowed tumor resection to be performed with high surgical safety and accuracy, the tumors were completely removed in all 12 patients, with less intraoperative blood loss and short length of hospital stay. Additionally, the DaVinci robot has been used for the resection of thoracolumbar neurofibroma, paravertebral schwannoma, and transoral odontoidectomy [14,76]. But these studies are case reports, large-sample, multicenter studies on clinical application of DaVinci robotic system in tumor resection are needed to confirm its efficacy.
4. RA Decompressive Laminectomy
Decompressive laminectomy is the primary surgical procedure used to relieve mechanical compression of the spinal cord or the nerve root, and has become the gold standard for the treatment of these disorders [77,78]. However, clinical application of RA decompressive laminectomy has not yet been reported. The main technical difficulties involved in performing the procedure clinically include: (1) it is difficult to obtain accurate 3D models of the vertebral laminae from CT scan data; (2) steps involved in grinding trajectory and speed planning of robotic systems are complicated and cumbersome; (3) the physiological motion of the spine due to respiration occurred when the robot holds ultrasonic bone scalpel for lamina grinding, or abnormal motion of the spine caused by RA lamina grinding using a high-speed drill can both affect the accuracy and stability of the grinding process. Considering these 3 difficulties, RA laminectomy does not appear to provide obvious advantage over conventional laminectomy, its clinical application is limited. A previous study adopted a two-stage neural network for RA decompressive laminectomy [21], in the first stage, the intraoperative CT image was inputted to acquire the coarse segmentation of vertebrae, in the second stage, the target area was precisely segmented, and the other areas were segmented with low resolution. This 2-stage neural network can not only achieve precise laminae segmentation, but also result in less calculation amount, shorter image processing time, and improved accuracy of the reconstructed laminar models, which contributes to precise decompression of the laminae. Li et al. [79] proposed a novel lamina positioning neural network, which can reach a recognition accuracy of 95.7%, this neural network can also identify and precisely locate the surgical target area from CT images with a positioning error of only 1.12 mm. They also proposed a grinding trajectory generator algorithm that allows the computer to complete grinding trajectory planning automatically. And expected results were achieved after using the grinding trajectory generator algorithm for RA laminectomy in a sheep model, with more smoother and stable grinding trajectory. The above-mentioned studies are experimental researches, further studies are needed to confirm their clinical utility.
ADVANTAGES AND DISADVANTAGES OF RA SPINE SURGERY
1. Advantages of RA Spine Surgery
(1) High screw placement accuracy: Studies on comparison of the accuracy between RA and conventional fluoroscopy-guided pedicle screw placement showed that RA pedicle screw placement at all spinal segments had higher accuracy compared with conventional fluoroscopy-guided method (Table 2) [12,15,18,27,28,53,55,80]. However, some scholars believe that the screw placement accuracy of RA group was comparable with that of FH group [23]. Li et al. [12] also compared the accuracy of RA and conventional fluoroscopy-guided pedicle screw placement in 81 patients with degenerative lumbar spine disease, and found a screw placement accuracy of 93% in the RA group, which was higher than the conventional fluoroscopy group (83%). In terms of accuracy between RA versus navigation-assisted pedicle screw placement, a meta-analysis study consisting of 6 articles with 529 patients and 4,081 thoracolumbar pedicle screws conducted by Zhou et al. [81] demonstrated a higher accuracy with Mazor robotic system-assisted pedicle screw placement compared to computer-assisted navigation. Du et al. [82] conducted a comparative clinical study to compare the accuracy of pedicle screw placement using the TiRobot system (n = 136) versus O-arm navigation system (Medtronic PLC, Medtronic Inc., Dublin, Ireland) (n = 166), they found that a total of 760 and 908 screws were inserted, respectively, in the TiRobot and navigation groups. The accuracy of pedicle screw placement was significantly higher in TiRobot group (96.2%) than the navigation group (90.5%). And the TiRobot group showed significantly less sagittal and transversal deviations, and lower rate of FJV compared with the navigation group. These aforementioned clinical studies confirm that the use of spine surgical robots is more accurate than conventional fluoroscopy and navigation in assisting screw placement.
(2) Reduced radiation exposure: Reducing intraoperative radiation exposure is a major concern for operative room staff during surgical procedures. Currently, several studies have shown that RA spine surgery significantly reduced the radiation exposure for the surgical team [12,23,26,80,83-85] (Table 3). Torii et al. [85] included 46 patients who underwent a posterior lumbar interbody fusion, 29 patents received screw placement using Mazor robotic system, 17 patients received conventional FH screw placement, the results showed that the fluoroscopy time per segment in the RA group was 29.6% of the time of the conventional surgery group, indicating that the use of Mazor robotic system results in marked reduction of radiation exposure to the surgeon. The radiation exposure time was also related to the choice of different robotic systems. Li et al. [86] demonstrated significantly longer radiation exposure time with the use of TiRobot and ROSA robotic systems compared to Mazor robotic system. At present, minimally invasive surgery is being increasingly requested by patients, but this surgical method can result in more radiation exposure when compared to open surgery. Lee et al. [87] compared radiation exposure between percutaneous and open RA short lumbar fusion in 310 patients, and found that the fluoroscopy time per screw was 14.4 seconds for percutaneous surgery, 10.1 for the open surgery, indicating that percutaneous RA surgery had significantly longer radiation exposure time compared with the open RA surgery. Shahi et al. [88] conducted a comparison of radiation exposure between navigation-assisted and RA 1-level or 2-level MIS-TLIF, there were 133 and 111 patients, respectively, in the navigation- and RA groups, they found that the total fluoroscopy time (20 seconds vs. 25 seconds), total radiation dose (38 mGy vs. 42 mGy), and percentage of intraoperative radiation exposure (58% vs. 65%) were significantly less in the RA MIS-TLIF group compared to the navigation-assisted group.
(3) High stability: RA techniques can prevent operation errors caused by physiological factors, such as surgeon fatigue and tremor of human hands [89]. At the same time, the high stability of the robotic platforms could obviously reduce deviation of screw placement performed by less experienced young surgeons, and shorten the screw placement time, this may also be related to the fact that young surgeons can adapt quickly to new techniques [90,91]. In addition, the robot assistance allows for the placement of “optimal screws” with longer length and larger diameter to maximize structural stability and reduce screw loosening, thus creating conditions for successful fusion [44,92].
(4) RA spine surgery is more minimally invasive: RA spine surgery is associated with smaller surgical incision, less vascular and nerve damage due to improved visualization of the surgical field intraoperatively, faster postoperative recovery, shorter hospital stay, lower complication rates [29]. A multicenter study led by Lee et al. [93] explored the 5-year trends in outcomes and complications in 722 patients undergoing RA spine surgery using Mazor robot, and found that the rates of postoperative complication such as nerve injury and loss of motor/sensory function remains extremely low over time (1.7%–5.7%), and the length of hospital stay can be reduced by almost 1 day, as the surgeon’s experience with robots increased.
(5) RA spine surgery can improve the unequal distribution of medical resources: Surgical specialists can control the robotic systems remotely to perform complex spine surgeries on patients living in medically underserved areas, thereby benefiting more patients with spine disorders [5,11].
(6) Robot assistance can facilitate the training of young surgeons: interactive learning model can be established using robotic system software. Young surgeons can use such model to simulate any surgical procedures and formulate multiple surgical plans, which can be further checked and modified by a senior surgeon prior to execution [94,95].
(7) Diversified image guidance methods: With the continuous updating and iteration of spine surgical robotics, the image guidance methods it supports are more diversified and more suitable for the uneven distribution of global medical resources [8,10]. Among the mainstream robot systems, Mazor SpineAssist, Mazor Renaissanc and Orthbot only support the matching of uploaded preoperative CT data and intraoperative C-arm anteroposterior and oblique x-ray fluoroscopy to complete the image registration process [15,23,28], while TiRobot and ROSA only support using intraoperative O-arm or C-arm scanning to complete the image registration process [5,18]. Although the use of intraoperative O-arm guidance will undoubtedly provide better accuracy and safety, however, due to the high cost of O-arm, the application of intraoperative C-arm matching will be more popular in areas where medical resources are scarce [9]. It is worth mentioning that Mazor X, Mazor X Stealth Edition and Excelsius GPS, the 3 robotics, support the above 2 image registration approaches, but if conditions permit, intraoperative O-arm guidance should be preferred to improve accuracy and safety [96].
2. Disadvantages of RA Spine Surgery
(1) High cost: The development, fabrication, and maintenance costs of the robotic systems are high. There is a necessity to purchase sterile materials in order to ensure a smooth and safe robot-guided procedure, and medical institutions need to provide additional training for surgeons and employ technicians for the use and maintenance of the robotic systems, resulting in increased surgical costs by approximately 30%. Small and mediumsized medical institutions and patients with financial difficulties are unable to afford these costs, thus limiting the widespread utilization of this technique [97].
(2) The occurrence of screw misplacement: Zhang et al. [98] showed that obesity, osteoporosis, severe vertebral rotation, and congenital scoliosis are risk factors for RA screw misplacement. Some scholars have proposed solutions to this problem. For patients with high body mass index and increased soft-tissue thickness of lower back, the incision should be lengthened appropriately, and assistants should pay attention to protect screw placement process with the use of a retractor. Additionally, the probability of “screw slippage” may be reduced after the screw entry point is trimmed and flattened using a rongeur forcep. However, more clinical evidences are needed to support the efficacy of the above-mentioned methods [10,96,99].
(3) Unexpected change in surgical methods: In some patients, there is a mismatch between preoperative CT image and the intraoperative fluoroscopic image, screws need to be placed via FH technique, this change can prolong the operation time [100].
(4) Limited clinical application: Currently, robot assistance is only used in targeted puncture, tumor resection and spinal implant placement [89,96]. The application of spine surgical robots in other spine surgeries still has many technical difficulties.
(5) Learning curve: Several studies have shown that the length of learning curve for RA spine surgery varies due to the use of different robotic systems [101-104]. The learning curve was determined by a variety of factors, including total time, screw insertion time, screw accuracy and number of cases required to complete the learning phase. Clinicians who first use robotic systems can pass the learning stage after completing approximately 20–30 surgeries, while clinically experienced clinicians only need 17–20 cases to pass this stage [101]. A comparative study by Vasan et al. [105] showed that compared with RA MIS-TLIF assisted by ROSA, the use of Mazor Renaissance for MIS-TLIF was easy to operate, with significant shorter operation time and smooth learning curve. In addition to the difference in the length of learning curve using different robotic systems, surgeons’ personal factors may also affect the learning curve of this new technique, for example, older surgeons may be less adaptable to the new technology, and a heavy clinical workload does not leave sufficient time for surgeons to learn RA surgical procedures systematically. Recently, several studies showed that with effective teamwork, the learning curve of RA surgery will be smooth and shorter [102,106].
SUMMARY AND FUTURE PERSPECTIVES
Currently, a large number of clinical studies have documented the safety, accuracy, and stability of using spine surgical robots to assist in screw placement, targeted puncture, and tumor resection [23,107]. However, operation processes associated with the use of robots in spine surgery are complicated, hence the clinical application of spine surgical robots is relatively limited. Their use in laminectomy and osteotomies has not been reported. In the future, with the development of science and technology, operation processes of spine surgical robots will be simplified, high-precision RA laminectomy and osteotomy is expected to be applied clinically.
It is believed that with the rapid development of artificial intelligence and the deepening of medical-engineering integration, spine surgery will become more intelligent, minimally invasive, and precise, with improved visualization. Spine surgical robots may be more clinically applicable, thus greatly improving surgical safety, and ultimately benefiting numerous patients.
Notes
Conflict of Interest
The authors have nothing to disclose.
Funding/Support
Key Research and Development Program of Shaanxi Province (No. 2020GXLH-Y-003); Key Research and Development Program of Shaanxi Province (No. 2020SFY-095); Shaanxi Province Science and Technology Nova Project (No. 2022KJXX-64).
Author Contribution
Conceptualization: JW, JM, BW; Formal Analysis: JW, JM, YW; Investigation: JW, JM, YZ, YD, YW; Methodology: JW, BW, YZ, DH; Project Administration: BW, DH; Writing – Original Draft: JW; Writing – Review & Editing: BW, DH.