- Search
| Neurospine > Volume 19(2); 2022 > Article |
|
|
Abstract
Objective
The study investigated our institutional learning curve for the ROSA ONE spine system (ROSA) based on ROSA usage time.
Methods
ROSA was designed to provide high accuracy for spinal pedicle screw placement through a built-in tracking technique. This study was conducted from November 2018 to January 2021. The time taken to complete each step of the robotic workflow was recorded. Patient demographics, comorbidities, surgical indications, and number of screw placements were examined in subgroup analysis. The Curve Fitting-General package (a part of NCSS 2021 software) was used to fit a mathematical model to the learning curve. Patient demographics, imaging data, and surgical time were reviewed retrospectively.
Results
A total of 167 patients who had undergone surgery were included. The mean total ROSA usage time was 107.1 ┬▒ 27.3 minutes. The estimated learning rate was 90.4%, and the largest slope change occurred close to the time of the 20th surgery. The observed overall learning trend in the 4-screw group could be attributed to screw planning. The presence of scoliosis (p = 0.73) or spondylolisthesis (p = 0.70) did not significantly influence the mean total time (TT) for all patients; however, the mean TT differed significantly (p < 0.01) among subgroups stratified by body mass index, screw number placement, and thoracic spine involvement.
In recent decades, substantial advances have been made in pedicle screw techniques for treating spinal diseases [1]. Threecolumn fixations, which provide the most rigid form of posterior stabilization, are commonly used to treat spinal degenerative disease, fractures, and deformities. Nevertheless, pedicle screw malposition, particularly pedicle violation (which often occurs when performing freehand techniques), is still an unavoidable problem. Multiple surgical techniques (e.g., imageguided or navigation devices and robot-assisted pedicle screw placement) have been developed to improve the precision of pedicle screw placement and reduce pedicle violation risk [2,3]. Robot-assisted techniques provide noninferior to superior benefits relative to freehand techniques in terms of pedicle screw placement accuracy, pain scores, Oswestry Disability Index scores, and intraoperative radiation exposure time (i.e., shorter exposure time) as well as equivalent postoperative stay [2]. New technologies are increasingly being applied to improve the capabilities of established systems. Three major robotic systems are currently used for spine surgery, namely the Renaissance Robotic Surgical System (Mazor Robotics, Caesarea, Israel), TiRobot Orthopaedic Robotic System (TINAVI Medical Technologies, Beijing, China), and the newly U.S. Food and Drug Administration (FDA)-approved ROSA robot (Medtech, Montpellier, France) [4].
Although robot-assisted spine surgery allows for efficient and accurate hardware placement, this technology is relatively new and is seldom used by spine surgeons. The newly FDA-approved ROSA robot was designed to aid spinal surgeons in performing minimally invasive spine procedures. The ROSA ONE spine system (ROSA) was designed to provide assisting spinal pedicle screw placement through a built-in tracking technique. However, few studies have examined the use of this technology and have developed corresponding guidelines. New surgical technologies such as the ROSA robotic system have a substantial learning curve [5]. In 2018, our institute became the first in Asia to utilize ROSA, which has been utilized in 171 spinal surgeries so far. Therefore, the present study investigated our institutional learning curve for ROSA based on ROSA usage time.
A retrospective chart review (the requirement for informed consent was waived by the relevant Institutional Review Board) of data pertaining to the period from November 2018 to January 2021 was conducted. The demographic, preoperative, and intraoperative data of 167 patients who underwent thoracic and lumbosacral pedicle screw placement with minimally invasive navigated robotic guidance using intraoperative computed tomography (CT, O-arm device) were analyzed. A total of 171 surgeries were included; 4 surgeries were excluded because they were not performed as per the current surgical workflow. The included surgeries were all elective surgeries for spinal diseases requiring spinal fixation, which included degenerative spondylolisthesis, scoliosis, spinal stenosis, vertebral compression fracture, vertebral body malignancies, cord compression, and disc disease. The decision to perform robot-assisted or conventional posterior instrumentation was made on the basis of clinical findings independent of the present study. The time taken for each step of the robotic workflow was recorded. Subgroup analysis of patient demographics, comorbidities, surgical indications, and number of screw placements were conducted. The spinal level of screw fixation (thoracic, lumbar, or sacral level) was recorded accordingly. Of the 167 patients, 158 patients had bone mineral density (BMD) results. Pedicle variation and deformities such as scoliosis and spondylolisthesis were graded as per the Nash and Moe grading method and Meyerding classification, respectively.
In the operating room, after the general anesthesia, the patient is put on the radiolucent surgical table with prone position and 2 bolsters are positioned longitudinally beneath both sides of trunk. The surgical region is sterilized and draped, and the O-arm device and ROSA are positioned. After the percutaneous reference pin is fixed in the right iliac wing, the ROSA registration is started. The fiducial box which is held by ROSA arm is placed above surgical region, and the 3-dimensional (3D) image acquisition is performed by O-arm device afterwards. The 3D transformation is conducted through the ROSA workstation. The surgeon plans the 3D trajectory of screw by the ROSA workstation. The ROSA workstation provides several choices of the screw simulation with distinguished diameter and length, as well as different color for the operative side. After the screw planning, the surgeon scrubs again for the guide pin placement under the ROSA guide. Subsequently, the 3D image confirmation of guide pin placement by the O-arm device is performed (Fig. 1). When necessary, decompression, fusion, or other surgical procedures are subsequently performed using minimally invasive techniques [6] before the pedicle screws are placed along the pin. Eventually, the surgical arm is removed, and the wounds were closed.
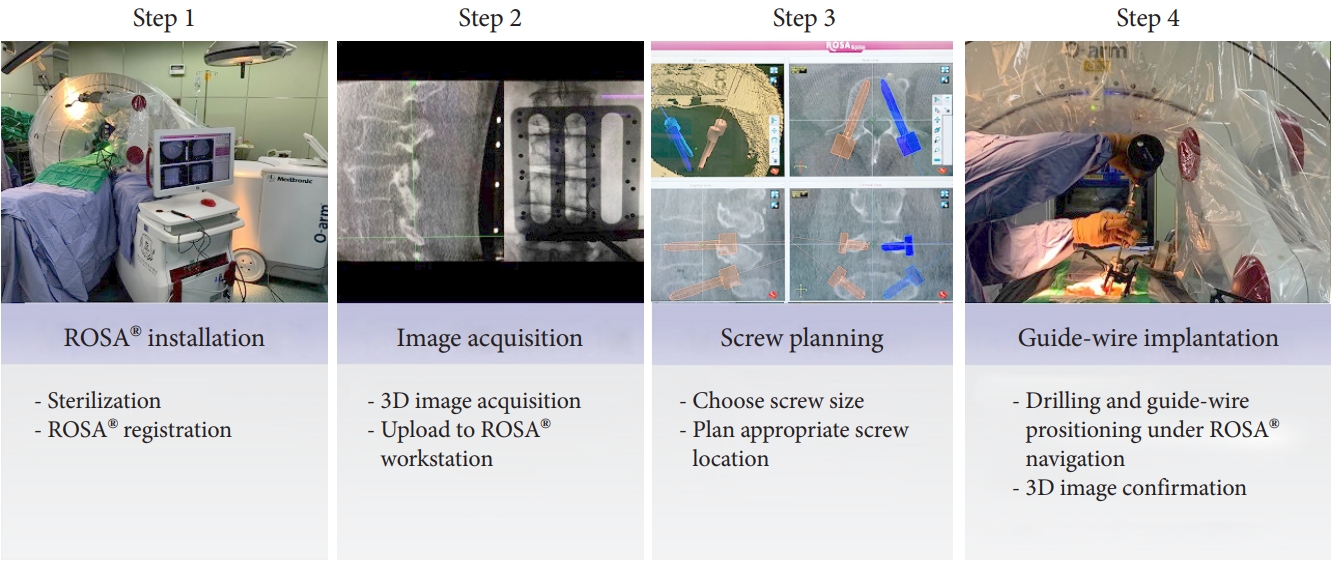
In order to analyze the learning curve of the ROSA ONE Spine System, we segmented the ROSA usage time into the 4 periods as follows (Fig. 2) [4]:
This step includes the sterile draping of the surgical region and devices, percutaneous insertion of the reference pin on the right iliac wing or spine process, and the performance of ROSA boot-up registration.
The intraoperative 3D image acquisition of the surgical region is conducted using the O-arm device with installed reference devices, and 3D transformation is conducted through the ROSA workstation.
The surgeon plans the 3D trajectory (including the screw entry point, direction, and paramedian longitudinal skin incision) for the minimally invasive surgical approach by using the ROSA workstation.
This step includes robotic auto-tracking movement to the planned skin surface, soft tissue deepening, bone drilling, and placing the guide-wire needle into the vertebrae. The steps are repeated to complete the implantation of all guiding pins, after which CT image confirmation of guide pin placement (through the O-arm device) is performed.
Total time (TT) of ROSA usage measures the TT taken to complete all of the aforementioned steps.
All categorical variables are expressed as numbers (percentages), and all continuous variables are expressed as means ┬▒ standard deviations. All data analyses were conducted using NCSS 2021 software (NCSS, East Kaysville, UT, USA) with 2-sided tests and a type 1 error rate of 0.05. The learning curve was plotted according to TT and the number of procedures. The Curve Fitting-General package (which is a part of NCSS 2021) was used to fit the following mathematical model to the learning curve: Y= A├Ś XB
where Y = TT of each procedure, A = the TT taken for the first procedure (i.e., the first case), X= the procedure index, and B= the index of learning. The estimated B value (and its 95% confidence interval) was then used to calculate the estimated learning rate (LR) as per the following mathematical formula:
A general linear mixed-effect model was used to determine the effect of the fixed factors on each ROSA time interval. The fixed factors included in the model were stratified categories of clinical experience, sex, number of screws (4, 6, or 8), presence of scoliosis (yes or no), and T spine involvement (yes or no). Two interaction terms (number of screws├Ś presence of scoliosis, T spine involvement ├Ś presence of scoliosis) were also included in the model. The age, body mass index (BMI), and BMI of each patient were measured as covariates, and the procedure index was measured as the random factor.
In our cohort (Table 1), the mean age of patients was 64.7┬▒10.3 years (range, 27ŌĆō88 years). Moreover, 104 (62.3%) and 63 (37.7%) were women and men, respectively. Their BMI was 25.9┬▒ 4.3 kg/m2 (range, 16.2ŌĆō41.1 kg/m2), and their mean BMD T-score was -0.9┬▒ 1.7 (range, -4.4 to 5.4). Comorbidities comprised cancer (n=4), osteoporosis (n=31), obesity (n=56), diabetes (n=31), cardiovascular disease (n= 21), and hypertension (n= 83). Surgical indications comprised spondylolisthesis (n= 156), scoliosis (n= 46), and fracture (n= 8) or malignancies (n= 4) of the vertebral body resulting in spinal stenosis and myelopathy. Robotic surgery was most often performed with 4 screws (n= 106), followed by 6 (n= 51) and 8 (n= 10) screws. The spinal level of screw fixation was from T2 to S1, and the lumbar spine was involved for most patients (95.2%; the thoracic spine was involved in only 8 patients).
The mean total ROSA usage time was 107.1┬▒ 27.3 minutes, and the most time-consuming fractioned ROSA usage component was operating theater preparation and reference device installation (S1= 40.2┬▒ 16.1 minutes), followed by guide pin placement (S4= 29.5┬▒ 11.0 minutes), screw planning (S3= 15.5┬▒ 7.3 minutes), and image acquisition (S2= 12.0┬▒ 7.3 minutes). Mean TT differed significantly (p< 0.01) among the subgroups stratified by number of screws, with the shortest mean TT (98.70 minutes; 95% CI, 85.10ŌĆō112.30 minutes) achieved for 4-screw surgeries, followed by the mean TT for 6-screw (113.00 minutes; 95% CI, 100.90ŌĆō125.09 minutes) and 8-screw (140.62 minutes; 95% CI, 120.58ŌĆō160.67 minutes) surgeries. The subgroup analysis of TT was performed for patients who underwent ROSA spinal surgery, and the results revealed that thoracic spine involvement was associated with a longer mean TT ([131.11 minutes; 95% CI, 109.21ŌĆō153.01 minutes] vs. [103.77 minutes; 95% CI, 96.73ŌĆō110.82 minutes], p= 0.01). The presence of scoliosis (p = 0.73) or spondylolisthesis (p = 0.70) did not significantly influence mean TT for all patients. An analysis was further conducted to adjust for the interacting effect of number of screws, presence of spondylolisthesis, and presence of scoliosis; it did not reveal any significant difference among subgroups (Table 2).
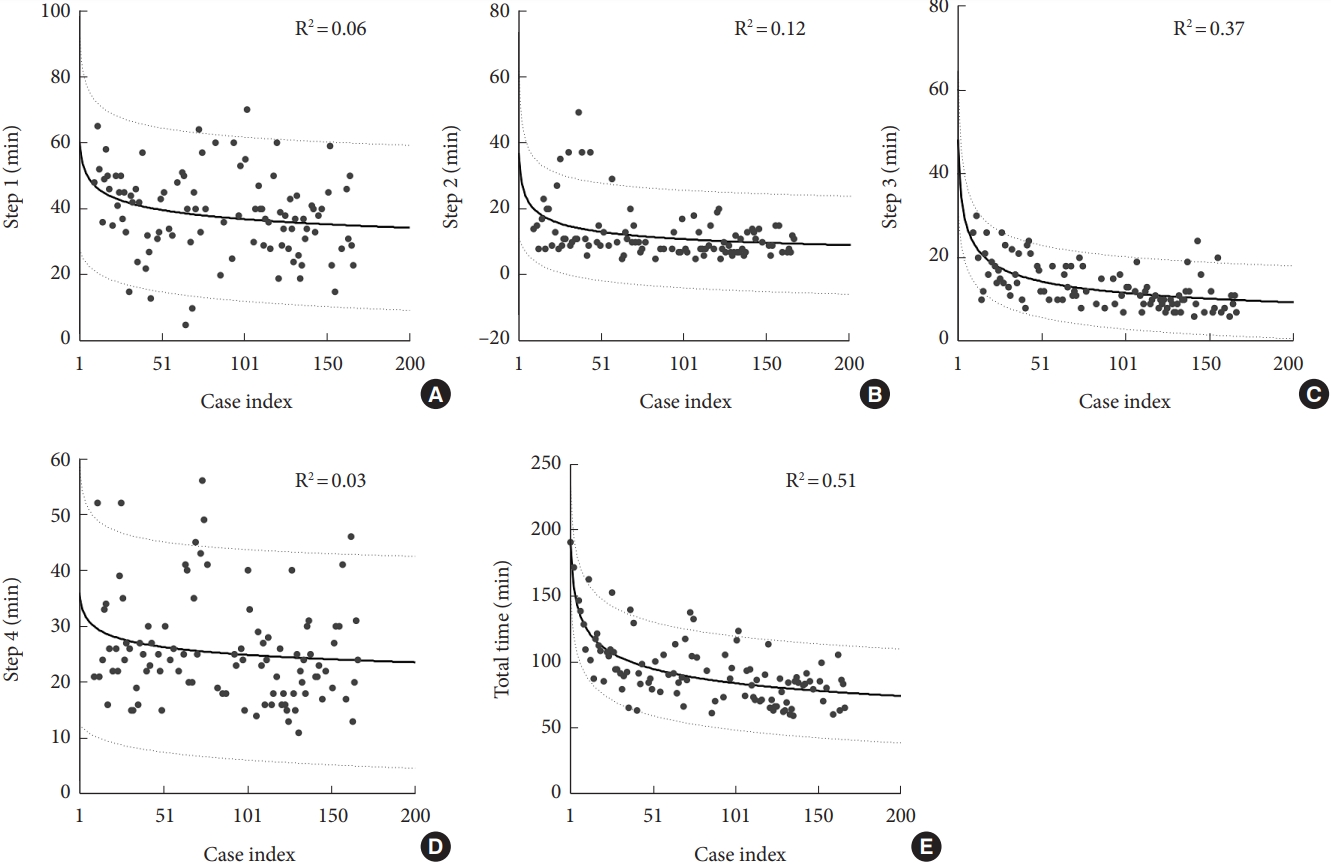
Through the application of a cumulative average model, we fitted a learning curve on the TT of all patients (Fig. 3), with an R2 value of 0.35. The estimated LR was revealed to be 90.1% (88.3%ŌĆō92.0%), and the largest slope change occurred close to time of the 20th surgery. Most enrolled patients received 4 screws; hence, we applied the same model to estimate the learning curve for each ROSA usage period in this specific group. Fig. 4 indicates that in the 4-screw group, the R2 values of the estimated learning curve for S1, S2, S3, S4, and TT were 0.06, 0.12, 0.37, 0.03, and 0.51 respectively. These findings indicated that the observed overall learning trend for the 4-screw group can primarily be attributed to S3.
For the conventional freehand technique, surgeonŌĆÖs experience, spinal level, number of screw insertions, obesity, and presence of spinal deformity (e.g., spondylolisthesis and scoliosis) were assumed to affect time usage for transpedicular screw placement. Therefore, we examined whether these measures moderated the effect of phase on fractioned ROSA usage period. Table 3 summarizes the results of the F tests for each of the fixed effects in each model, and they revealed the significant main effect of surgeonŌĆÖs experience (per 20 surgeries) on S2 (F= 4.37, p< 0.01), S3 (F= 6.69, p< 0.01), S4 (F= 5.11, p< 0.01), and TT (F= 9.99, p< 0.01). No significant association between surgeonŌĆÖs experience and S1 (F = 1.18, p = 0.32) was observed, and age and sex did not significantly influence the fractioned ROSA usage periods. BMI significantly influenced and positively predicted S4 (F= 17.86, p< 0.01) and TT (F= 6.74, p= 0.01). BMD did not significantly influence any fractioned/total ROSA usage period. Number of screws significantly influenced and positively predicted S3 (F= 11.10, p< 0.01), S4 (F= 23.51, p< 0.01), and TT (F = 11.31, p < 0.01). Also, the thoracic spine involvement significantly influenced and positively predicted S4 (F=4.09, p= 0.05) and TT (F= 7.90, p= 0.01). When the presence of scoliosis or spondylolisthesis was added to the model, no significant main effect was observed (Table 3).
Similar to most contemporary surgical robotic devices, ROSA was designed as a shared-control system that combines the application of navigation with robotic technology [4]. Studies have demonstrated ROSAŌĆÖs benefits, particularly those derived from its real-time guidance feature [6]. Furthermore, the accuracy and reliability of 3D trajectory are helpful for minimally invasive surgery when exposure is limited [4]. However, surgeons who use ROSA are still affected by several major limitations, including a steep learning curve, inability to produce preoperative screw trajectories, and prolonged time taken to perform system installation and setup [5].
Our single-institution retrospective study revealed the experience derived from performing a high number of surgeries, a large case number, and the learning curves for each crucial step of ROSA-guided pedicle screw placement. Significant, but nonlinear, increases in TT of ROSA usage, screw planning time, and guide pin implantation time were observed when more inserted guide pins were used. Notably, thoracic spine involvement and high BMI significantly prolonged TT and guide pin placement time. This finding indicates that the structural complexity and tension of the myocutaneous flap after its dissection along a screw trajectory (e.g., stiffer thoracodorsal fascia compared with lumbodorsal fascia or thicker subcutaneous fat tissue in obesity patients) can affect the difficulty of ROSA usage and, consequently, increase ROSA usage time. In addition, the presence of osteoporosis or spinal deformity (i.e., scoliosis and spondylolisthesis) did not significantly influence total/fractioned ROSA usage time, which indicates the benefits of ROSA usage relative to the freehand technique. Our data suggest that the learning curve for pedicle screw placement in terms of time taken can be shortened for an experienced surgeon. We noted a decrease in the learning curve gradient after the first 20 surgeries, with an LR of 90.1% (Fig. 3); this can primarily be attributed to the trajectory planning period (S3; R2= 0.37) of the 4-screw group (Fig. 4). This finding is comparable to that of Schatlo et al. [7] who reported higher misplacement rates between the 10th and 20th surgery, which limited the learning curve for robotic spine surgeries. Thus, for surgeons with no experience in using the technique, experienced supervision should be provided for the first 25 surgeries.
Due to familiar with the manipulation of the ROSA and Oarm device, and the diminishing the S3 time-consuming, the TT of ROSA usage can be reduced. In our experience, the more practice in patient positioning with the ROSA and O-arm device we did, the more S1 time-consuming we reduced. Besides, you also can put some labels on the floor to mark the position between. About the reducing the time of the S2, we believed the most importance is the intraoperative 3D image acquisition which is related to the positioning between the fiducial box and O-arm device. Moreover, there were the skills required for the pedicle drilling which might be influenced the surgical time of the S4, although the distribution of the statistic Fig. 4 in the S4 is too wide to be reliable.
Nine cases of technical error were reported for our cohort; they included delays in equipment sterilization (n= 2), repeat sterilization of equipment dropped by accident (n= 1), re-registration of ROSA (n= 1), system failure of ROSA (n= 3), system failure of O-arm system (n= 1), and hardware failure due to broken wheel of O-arm device (n= 1). Notably, major improvement was achieved after the rebuilding of the operating floor to eliminate the unnoticeable yet problematic tilting of ROSA, which hindered the successful and accurate registration of ROSA. Similar to the finding reported by BÄcker et al. [8] who investigated the use of the Renaissance robotic system, we observed that the surgical workflow may be influenced by variations in surgical teams comprising scrub nurses and residents. Therefore, to enhance the efficacy and reproducibility of the ROSA setup, several measures were implemented to correct the position of the surgical table, O-arm, and ROSA. This measure reduced the number of interpersonal errors made by scrub nurses and residents, especially with respect to the mounting of the device over the operating room table, which is crucial in the system setup step. All devices should be routinely checked and serviced to minimize the occurrence of software and hardware failure.
The present study was a retrospective study. We detected missing data points and outliers that were related to the technical errors that led to an increase in time taken and which might have had a negative effect on the results of the analysis. Nine missing data points (5.3%) and 4 cases of technical error (2.4%) were identified for S1, 9 missing data points (5.3%) were identified for S2, 9 missing data (5.3%) were identified for S3, 9 missing data points (5.3%) were identified for S4, and 1 missing data point was identified for TT (0.6%).
The present study addresses a single surgeonŌĆÖs learning curve and experience with respect to robot-assisted pedicle screw placement using ROSA. The indicative learning curve involved 20 surgeries, and the presence of scoliosis or spondylolisthesis did not significantly influence ROSA usage time. With the enhancement of system installation and teamwork, ROSA usage time can be reduced.
NOTES
ACKNOWLEDGEMENTS
The authors would like to acknowledge the Laboratory Animal Center at Taipei Medical University for technical support in manuscript preparation experiment.
Fig.┬Ā1.
(A) Sterile draping the surgical region. (B) Overview of operation room and ROSA registration. (C) Three-dimensional (3D) acquisition with fiducial box in position. (D) Screw planning by using the ROSA workstation. (E) Drilling and guide-wire placing under ROSA guide. (F) 3D image confirmation of the guide pin placement.
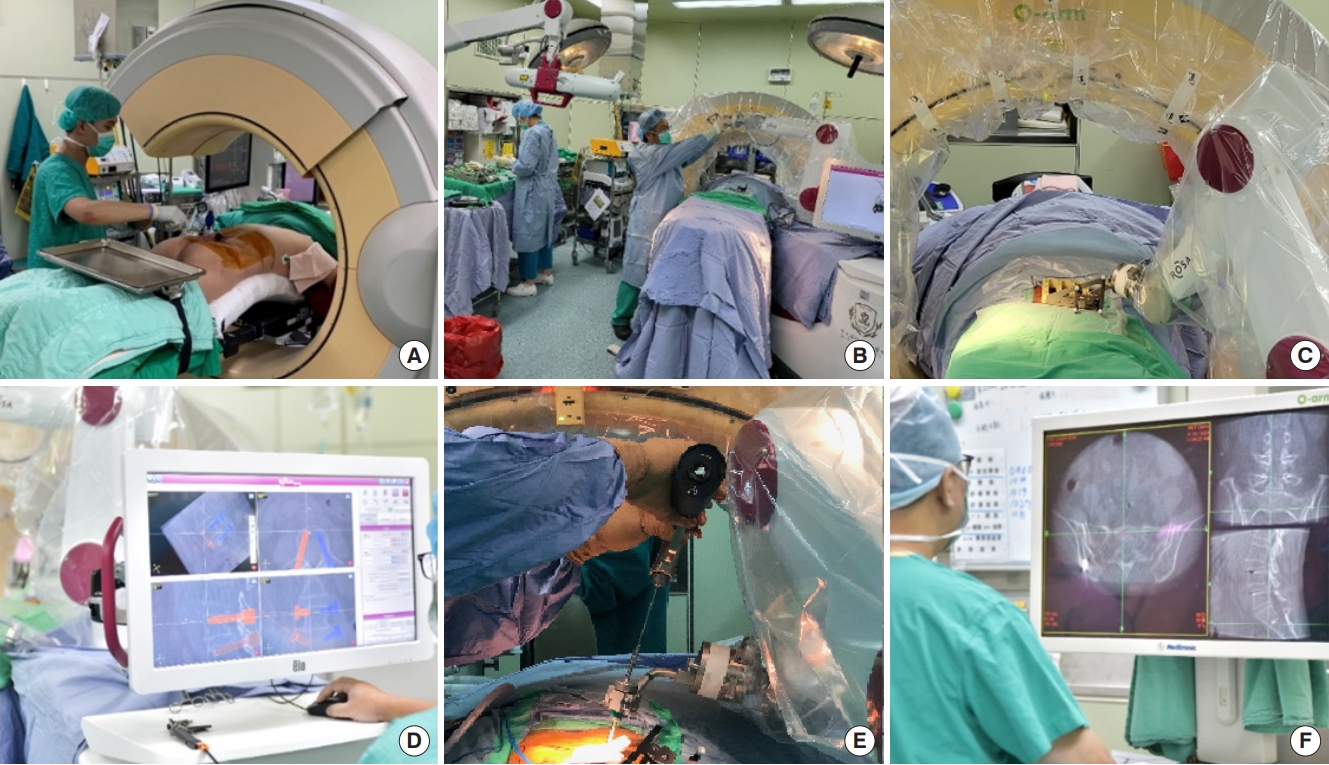
Fig.┬Ā2.
Steps of surgical workflow. Step 1: ROSA installation step comprising sterilization and ROSA registration. Step 2: image acquisition step comprising 3-dimensional (3D) image acquisition and uploading of data to ROSA workstation. Step 3: screw planning step comprising selection of appropriate screw size and determination of appropriate screw location. Step 4: guide-wire implantation step comprising drilling and guide-wire positioning through ROSA navigation with 3D image confirmation.
Table┬Ā1.
Characteristics of 167 patients who received ROSAguided pedicle screw placement
Table┬Ā2.
Effect of number of screws, thoracic spine involvement, scoliosis, and spondylolisthesis on total time (TT)
| Variable | Mean (95% CI) | p-value |
|---|---|---|
| Screw numbers | < 0.01 | |
| ŌĆāGroup 1:4 | 98.70 (85.10ŌĆō112.30) | 0.02ŌĆĀ/ < 0.01ŌĆĪ |
| ŌĆāGroup 2:6 | 113.00 (100.90ŌĆō125.09) | < 0.01┬¦ |
| ŌĆāGroup 3:8 | 140.62 (120.58ŌĆō160.67) | |
| T spine involvement | 0.01 | |
| ŌĆāNo | 103.77 (96.73ŌĆō110.82) | |
| ŌĆāYes | 131.11 (109.21ŌĆō153.01) | |
| Presence of scoliosis | 0.73 | |
| ŌĆāNo | 118.63 (104.62ŌĆō132.64) | |
| ŌĆāYes | 116.25 (101.48ŌĆō131.01) | |
| Presence of spondylolisthesis | 0.70 | |
| ŌĆāNo | 116.19 (103.78ŌĆō128.61) | |
| ŌĆāYes | 118.69 (103.08ŌĆō134.29) | |
| Presence of scoliosis ├Ś screw numbers | - | 0.08 |
| Presence of spondylolisthesis ├Ś screw numbers | - | 0.08 |
| Presence of scoliosis ├Ś presence of spondylolisthesis | - | 0.30 |
Least square means of the TT for the levels of each fixed factor of interest when age=65.3, body mass index=25.8 kg/m2, and bone mineral density=-0.90.
CI, confidence interval.
All values are adjusted for sex, clinical experience (every 20 cases), and presence of scoliosis├ŚT spine involvement interaction.
Table┬Ā3.
Effect of clinical experience and each factor on fractioned ROSA usage period
REFERENCES
1. Kotani T, Akazawa T, Sakuma T, et al. Accuracy of pedicle screw placement in scoliosis surgery: a comparison between conventional computed tomography-based and O-arm-based navigation techniques. Asian Spine J 2014;8:331-8.



2. Li HM, Zhang RJ, Shen CL. Accuracy of pedicle screw placement and clinical outcomes of robot-assisted technique versus conventional freehand technique in spine surgery from nine randomized controlled trials: a meta-analysis. Spine (Phila Pa 1976) 2020;45:E111-9.

3. Mason A, Paulsen R, Babuska JM, et al. The accuracy of pedicle screw placement using intraoperative image guidance systems. J Neurosurg Spine 2014;20:196-203.


4. Lefranc M, Peltier J. Evaluation of the ROSATM Spine robot for minimally invasive surgical procedures. Expert Rev Med Devices 2016;13:899-906.

5. DŌĆÖSouza M, Gendreau J, Feng A, et al. Robotic-assisted spine surgery: history, efficacy, cost, and future trends. Robot Surg 2019;6:9-23.


6. Chenin L, Peltier J, Lefranc M. Minimally invasive transforaminal lumbar interbody fusion with the ROSA(TM) Spine robot and intraoperative flat-panel CT guidance. Acta Neurochir (Wien) 2016;158:1125-8.