- Search
|
|
||
Abstract
Objective
Interlaminar endoscopic spine surgery has been introduced and utilized for lumbar lateral recess decompression. We modified this technique and utilized it for bilateral lateral recess stenoses without significant central stenosis. Here we present the surgical details and clinical outcome of ligamentum flavum sparing unilateral laminotomy for bilateral recess decompression (ULBRD).
Methods
Prospectively collected registry for full-endoscopic surgeries was reviewed retrospectively. One hundred eighty-two consecutive cases from a single center between September 2015 and March 2021 were reviewed and 57 of them whom underwent ULBRD were enrolled for analysis. Basic patient demographic data, perioperative details, surgeryrelated complications, and clinical outcome were reviewed. The detailed surgical technique is presented as well.
Results
Among the 57 patients enrolled, 37 were males while the other 20 were females. The mean age was 58.53 ± 14.51 years, and a bimodal age distribution at the age of mid-fifties and mid-sixties or older was noted. The later age-peak was related to coexistence of degenerative scoliosis. The average operative time per lamina was 70.34 ± 20.51 minutes and mean length of stay was 0.56 ± 0.85 days. Four perioperative complications were reported (7.0%) and the overall reoperation rate at the index level within 1 year was 8.8%. The preoperative back/leg visual analogue scale scores and functional outcome scales including EuroQol-5 dimension questionnaire, Oswestry Disability Index presented significant improvement immediately after surgery and were maintained until final follow-up.
Radiculopathy of the lumbar spine is one of the most common pathologic conditions that spine physicians encounter in their daily practice. Lumbar spine diseases presenting with radiculopathy exert an enormous socioeconomic burden with increasing numbers of patients and costs associated with this disease [1]. Radiculopathy can be a consequence of various pathologic changes of the lumbar spine such as herniated intervertebral discs, lateral recess stenoses (LRSs), spondylolistheses, and rarely may be due to tumors or infections.
Diagnosis of symptomatic LRS is occasionally controversial, given that radicular symptoms often correlate poorly with imaging findings on magnetic resonance imaging (MRI) [2]. The lateral recess is a distinct anatomic area located ventral to the superior articular process (SAP) of the lower lumbar vertebrae, and is delineated by the intervertebral disc (IVD) ventrally and the facet joint dorsally. Since it was first reported in the early 1980s [3,4], LRS defines a degenerative condition resulting in subsequent neural compression characterized by significant compression of the traversing root at the entry zone of the foramen of lumbar roots. While the majority of lumbar central canal stenoses are seen in older patients due to the degenerative nature of the disease [5,6], LRS without central canal stenosis can be seen in the early stage of degeneration. Unlike the central stenoses which clinically present with neurogenic claudication, LRS usually present clinically very similar to herniated IVDs, making LRS a distinct pathology. For LRS patients not responding to thorough conservative treatment, surgical decompression of the lateral recess is recommended [7-9], and open laminectomy is widely accepted as a safe and cost-effective surgical approach for these intractable LRSs [10,11]. However there are significant drawbacks to traditional open laminectomies, including possible surgical injuries to the paraspinal structures. In addition, a certain amount of bony resection is necessary to create a path to the lateral recess and to achieve optimal bony decompression. Since the lateral recess is adjacent to the facet joint, there is a risk of iatrogenic segmental instability, which in some cases require arthrodesis surgeries [12-14]. Thus, various minimally invasive spine surgery techniques, including tubular retractor surgeries and endoscopic surgeries, which minimize approach-related injuries to the adjacent structure have been introduced [15-19]. Among these, full-endoscopic decompression for various lumbar degenerative lesions has gained popularity because of several advantages over open surgery, including lesser paraspinal injury, lesser blood loss, and rapid return to work and daily activities [7,17,19-21]. The use of full-endoscopic technique in the field of spine surgery has undergone considerable evolution [22], and scientific evidence is mounting regarding favorable clinical and radiologic outcomes, specifically for lateral recess decompressions [7,17,18,23-26]. However, previous research has focused primarily on unilateral decompressions, or lateral recess decompressions accompanied by central stenosis decompression for cases of bilateral decompression. These reports do not address the subset of LRS cases, often associated with a younger population, which require bilateral decompression of the lateral recess, but do not require decompression of the central canal. The objective of our research was to use our prospectively collected endoscopic spine surgery database to determine complication and clinical outcomes for patients at our institution who underwent unilateral laminotomy for bilateral recess decompression (ULBRD).
A prospectively collected database of consecutive endoscopic interlaminar lumbar surgeries by a single surgeon was retrospectively screened and queried for ULBRD between September 2015 and March 2021. The University of Washington Human Subjects Division reviewed and approved this study and all patients provided informed consent for participation. Patients enrolled included those with history of more than 6 weeks of conservative nonsurgical treatment including systemic medication (analgesics, nonsteroidal anti-inflammatory drugs). Patients with significant instability, grade C and D central stenosis [27], or significant disc herniation at the index level were excluded from analysis. Demographic information and clinical variables were obtained from the medical record, including preoperative imaging study results, clinical outcomes, intraoperative estimated blood loss (EBL), length of stay (LOS), underlying comorbidities, American Society of Anesthesiologists (ASA) physical status (PS) classification grade, perioperative complications, preoperative preparation time for anesthesia, positioning and drape, and incision to closure operative time. Clinical outcomes were measured by visual analogue scale (VAS) for both back and leg pains, Oswestry Disability Index (ODI) scores, and EuroQol-5 dimension (EQ-5D) self-reported questionnaires at preoperative, 2-week postoperative, 3-month postoperative, chronic (6-month to 1-year postoperative) and final postoperative follow-up. Follow-up clinical data was available for 52 of 57 patients (91.2%); 5 patients were lost to follow-up immediately after surgery and an additional 4 patients were lost to follow-up after the 2-week follow-up. Mean follow-up duration for the remaining 48 patients (84.2%) was 26.5± 18.7 months (range, 3–54 months).
All patients underwent routine imaging evaluation including radiographs and MRI. Preoperative radiographs consisted of plane anteroposterior and lateral and flexion/extension images. Instability at the index level was defined as excessive motion more than 3 mm on flexion/extension images [28]. MRI included multiple sequences of T1 and T2 weighted images. The presence of central canal stenosis and LRS was graded as previously described [17,27].
Under monitored general endotracheal anesthesia, the patient is positioned on surgical table with a Wilson frame. Similar to most endoscopic spine surgery techniques, ULBRD is facilitated with maximum flexion of the lumbar spine, resulting in maximized size of the interlaminar window for approach. Once the patient is positioned, an intraoperative C-arm fluoroscopic image is taken to check the level for surgery and to determine an optimal entry point for the endoscopic procedure. The endplate view of the lower vertebral body is found, and then by tilting the C-arm caudally, the maximized view of the interlaminar window can be achieved. At this fluoroscopic view, the point for skin incision is marked at the point where the caudal margin of the target lamina meets the upper endplate line of the caudal vertebral body. This point is the exact point of the lateral recess which is the target for decompression. This step can be done either before or after the patient is prepped and draped.
A vertical 0.7-cm-sized incision is made at the marked point, penetrating the skin, subcutaneous layer, and the lumbar fascia simultaneously. Step-by-step serial dilators are advanced through the incision and the inferomedial margin of the rostral index level lamina is palpated. Intraoperative fluoroscopy is taken to confirm the level and to assure that the dilators are located on the target area. Then a tubular retractor is placed with the bevel initially facing medially to avoid creep of the paraspinal muscles. Radiofrequency cautery and endoscopic surgical equipment are utilized to control any bleeding and remove any remnant soft tissue or debris, optimizing the endoscopic surgical view at the caudal margin of the index lamina and the medial aspect of the facet joint.
Surgical decompression is performed following the yellow ligament attachment along the rostral edge of the caudal index level lamina (Fig. 1A). First, minimal the resection or the inferomedial inferior articular process is carried out with the high-speed burr until the SAP is exposed (Fig. 1B). The medial aspect of the SAP is resected to achieve decompression of the lateral recess. After adequate bone work, the traversing nerve root is decompressed spanning from the tip of the SAP to the midportion of the caudal index level pedicle (Fig. 1C). The lateral aspect of the yellow ligament overlying the nerve root can be removed as necessary. The nerve root is then mobilized, any adhesions are resected with microscissors and decompression of the nerve root beyond the rostral and caudal aspect of the lateral recess is confirmed. After the ipsilateral decompression is completed, the working channel and endoscope is gradually tilted towards the contralateral side. Using the diamond burr the yellow ligament attachment on the rostral edge of the caudal index level lamina is followed to the contralateral side (Fig. 1D). Once the contralateral facet joint and lateral recess are visualized, the medial and ventral part of the SAP are undercut using a combination of high-speed drill and Kerrison rongeurs (Fig. 1E). The amount of ligamentum flavum removal at the bilateral lateral recesses depend on the severity of traversing root compression, but generally only the ligament overlying and compressing the nerve roots are removed and the central ligamentum flavum overlying central thecal sac is preserved. Finally, the contralateral traversing nerve root is mobilized with the blunt dissector and any adhesions are lysed (Fig. 1F). Typically, unless there is an accompanied significant disc herniation, there is no need for disc removal. The overall surgical procedure is also presented as a Supplementary videoclip 1.
Meticulous hemostasis should be performed prior to withdrawal of the endoscopic system and closure. Hemostasis can be assisted by radiofrequency cautery and/or use of hemostatic agents. Wound closure proceeds in layer-by-layer sutures. Steri-strips can be applied or skin sealing bonds may be used. Wound drains are not routinely placed unless required due to intraoperative issues. Patients are encouraged to ambulate immediately after surgery and discharged the same day if their medical status permits.
Demographic information, radiologic information, and clinical outcome measures were analyzed using descriptive statistics. Continuous variables are presented as mean± standard deviation, while categorical variables are shown by frequency and percentage equivalence. Statistical analyses comparing clinical results pre- and postoperatively were carried out by independent Student t-test; p< 0.05 was defined as statistical significance. IBM SPSS Statistics ver. 23.0 (IBM Co., Armonk, NY, USA) was used for analysis.
A total of 57 of the 182 patients in our database had ULBRD for bilateral LRS and met the inclusion criteria (Table 1). All patients had clinical and radiologic evidence of radiculopathy associated with bilateral LRS. The average age at time of surgery was 58.53± 14.51 years; 37 were male (64.9%) and 20 were female (35.1%). The age of patients who underwent surgery appear to cluster in a bimodal distribution, at the age of 40-mid 50 years and at > mid-60 years (Fig. 2). There were 5 patients with accompanying degenerative scoliosis of the lumbar spine (8.8%), and all were ≥ 65 years (20.9%, red circles in Fig. 2). A total of 26 patients had underlying hypertension (45.6%), 6 had diabetes mellitus (10.5%), and 23 had obesity with body mass index (BMI) higher than 30 (40.4%). The overall mean BMI was 29.85± 6.18 kg/m2. Preoperative back VAS was 7.04± 2.36, leg VAS was 6.31± 2.78, EQ-5D was 0.574± 0.182, and the ODI was 50.12± 15.01.
A total of 46 cases were single level decompressions and 11 were 2-level decompressions; There were no 3 or more level surgeries in this cohort (Table 1). Among the 68 levels of surgeries in all 57 patients, the L4/5 level was the most prevalent level with 40 levels (58.8%) and the L5/S1 level was the 2nd most prevalent level with 12 cases (17.6%) (Table 1). Higher levels such as L1/2 or 2/3 were less than 10% of the overall number of levels. There were 23 patients with an ASA PS classification of III (40.4%) while the other 34 had ASA PS classification of I or II. All endoscopic procedures were completed without any anesthesia- or surgery-related intraoperative morbidities or deaths. As shown in Table 2, average preoperative preparation time was 68.02 ± 15.88 minutes per patient and the operative time per operated lamina was 70.34± 20.51 minutes. EBL was 8.34± 15.11 mL per level and the mean postoperative LOS was 0.56± 0.85 days. There were 2 intraoperative complications (3.5%) which were both iatrogenic dural tears and subsequent cerebrospinal fluid leakages. One case resolved spontaneously while the second case required lumbar drainage and a longer hospital stay. There were 4 perioperative complications during the immediate postoperative period (7.0%) which included transient urinary retention, recurrence within a month, deep vein thrombosis of the leg and a case of postoperative epidural hematoma which required hematoma removal. Representative pre- and postoperative T2-weighted MRI axial images are shown in Fig. 3.
The back VAS, leg VAS, EQ-5D, and ODI scores at final follow-up were 4.28± 2.87, 2.31± 2.89, 0.724± 0.170, and 32.44±22.81 respectively. The detailed clinical pain outcome and functional outcome scores by different follow-up time points after surgery (2 weeks, 3 months, chronic 6–12 months, > 1-year, and most recent follow-up) are presented in Table 3 and Fig. 4. The postoperative VAS, EQ-5D, and ODI scores at each time point were all statistically significantly improved compared to the preoperative data (p< 0.05). These clinical outcome measures demonstrated an overall trend toward improvement until the chronic follow-up time point. The final follow-up VAS, EQ-5D and ODI scores presented a slight worsening, however this was not statistically significant. The overall rate for reoperation at the index level within 1-year was 8.8%, and the detailed information of the causes for reoperation are presented in Table 2.
Increased utilization of endoscopy in spine surgery is evidenced by a recent global survey by Lewandrowski et al. [29]. Despite a steep learning curve for the acquisition of endoscopic spine surgery skills [30], the endoscope is emerging as an essential component of the armamentarium of the modern spine surgeon. Full-endoscopic spine surgery allows favorable functional outcomes, faster recovery, earlier rehabilitation, less utilization of opioid medication while minimizing approach-related morbidity compared with open and conventional minimally invasive techniques [7,30-32].
Favorable clinical outcomes of unilateral lateral recess decompression using full-endoscopic technique have been described by several groups [7,17,18,23-26]. For our study, we were interested in better understanding the demographics, and clinical outcomes for our patients who require bilateral recess decompression, in the absence of central stenosis.
About 3 quarters of the spinal levels treated in our cohort were located at the 2 most caudal lumbar levels, reflecting the anatomic nature of the lateral recess. The lateral recess becomes narrower at the caudal lumbar segments compared with rostral levels. A previous anatomic study revealed that nonpathologic anteriorposterior measurements for the lateral recess decrease in a caudal-direction from 9.1 mm at L1 to 6.0 mm at L4, and the height of the anteriorposterior lateral recess at L5 is approximately 6.1 mm [33]. While the physical space of the lateral recess becomes smaller in caudal levels, the nerve roots traverse more horizontally rather than vertically compared with rostral levels, rendering the lateral recess even more vulnerable to possible compressions by adjacent structures.
Traditionally, decompression of the lateral recess for patients suffering from LRS utilized an open microsurgical approach. However, more recently, a broad range of surgical approaches for this type of decompression have been used, ranging from standard open laminectomies to endoscopic approaches using minimally invasive techniques [34-36]. Our group described a series of patients with symptomatic unilateral LRS who were treated successfully using interlaminar endoscopic decompression techniques [17]. Additional studies have demonstrated favorable clinical results comparing full-endoscopic with microsurgical techniques and highlighted the reduced approach-related morbidity afforded by a full-endoscopic approach [7,21,37]. Aforementioned studies and the current cohort underwent interlaminar full-endoscopic approach for decompression of the lateral recess. Another endoscopic approach, the trans-SAP approach for lateral recess decompression, provides an alternative option for exposing the exiting nerve root while avoiding inadvertent injury, particularly in cases where degenerative changes have distorted a patient’s anatomy [38]. While transforaminal or trans-SAP approaches cannot decompress the bilateral LRSs, our technique of ULBRD provides bilateral decompression via single approach, also providing a safe surgical corridor to access both lateral recesses in endoscopic decompression surgery. Considering the novelty of these techniques, additional research is warranted to further analyze the advantages provided by each approach for various patient populations.
The surgical approach for patients requiring bilateral recess decompression but lacking central stenosis, consists of a single approach for bilateral decompression preserving the central ligamentum flavum. In traditional open surgery or classical minimally invasive techniques, 2 different surgical approaches from each side are needed. Alternatively, with a unilateral approach, the midline structures, including the ligamentum flavum, must be sacrificed. There are consequences for this injury to the central structures, including the possibility of iatrogenic instability or post-laminectomy spondylolistheses [14], particularly in cases with no central stenosis, such as the cohort described here. The importance of preserving midline structures has led to development of less invasive normal structure preserving techniques. Minimizing medial facetectomies and preserving the midline ligamentous structures can cause a significant difference in lumbar stability compared to traditional decompressive laminectomies [39,40]. These results support leaving the medial border of the facet and preserving normal ligamentum flavum when a less destructive bilateral decompression is sufficient. Driven by this impetus, various minimally invasive techniques (open, tubular, and endoscopic) have been developed and evolved with the goal of maximally preserving midline structures from unnecessary removal and have shown promising results [15,16,41-43]. Our technique can provide another alternative by fully preserving the supraspinous and interspinous ligaments, and the ligamentum flavum, with minimal risks and excellent clinical outcomes.
Another potential advantage to preserving the ligamentum flavum during laminectomy is that epidural scarring can be minimized. Epidural scarring after a laminectomy surgery is known to serve as a possible factor inducing postoperative pain. Excessive production of fibrosis at the postoperative epidural bed can cause neural irritation or stretching, and sometimes even mass effects, resulting in radicular symptoms [44-46]. Therefore, avoiding or at least minimizing epidural scarring can improve postoperative clinical outcomes. Although it is impossible to completely prevent epidural scarring, efforts made to minimize this phenomenon in laminectomies [47,48] have demonstrated the clinical significance of ligamentum flavum preservation and support the use of endoscopic spine surgeries to minimize epidural scarring through minimizing the extent of laminectomies and ligamentum flavum resection. Further research is necessary to determine if surgical approaches preserving the ligamentum flavum benefit patients, including those in our cohort, by minimizing postoperative epidural fibrosis.
In our cohort of patients with bilateral LRS without central stenosis, age exhibited a bimodal distribution. The majority of patients were 40 to mid-50 years, with a second cluster at ≥ mid-60-years. Central lumbar stenosis is a very common condition; it is the most common indication for surgical decompression with or without fusion in the elderly [49]. The occurrence of central spinal stenosis is associated with advanced age, typically occurring in those > 65-years [50], consistent with our observation that the majority of our cohort was in the age range of 40’s–50’s. While patients in our cohort had not developed central stenoses, they may be experiencing the beginning stages of spine aging, resulting in their symptomatic LRSs. In our cohort, there were no cases of scoliosis in the younger subgroup, while > 20% of the older subgroup had accompanying scoliosis by diagnostic criteria, and additional patients exhibited subclinical (not meeting the diagnostic criteria) indications of scoliosis. Coronal imbalance of the lumbar spine can result in possible LRSs. It is possible that accompanying degenerative scoliosis-related coronal deformities are an explanation for the bimodal age distribution in this cohort; further research should aim at understanding asymptomatic LRS in older patients without central stenosis.
The current study has several limitations. It is a single-center series and describes a small patient’s cohort. Moreover, given that this is the first description and analysis following ULBRD, we are planning to further study patient selection criteria in order to identify patients who require additional foraminal decompression or stabilization and might be better suited for alternative procedures. Further studies are needed to provide the scientific/clinical evidence of beneficial effect of ligamentum flavum sparing to counteract epidural fibrosis.
In this study cohort, patients receiving ULBRD for bilateral LRSs without significant central stenosis obtained good clinical outcomes with acceptable perioperative complications rates. ULBRD enabled sufficient decompression of bilateral lateral recesses via a single endoscopic incision without sacrificing central structures such as the midline ligamentum flavum. Further research is needed to understand long-term outcomes in large, diverse patient populations, with the ultimate goal of providing recommendations for the use of ULBRD under various clinical scenarios.
SUPPLEMENTARY MATERIALS
Supplementary videoclip 1 can be found via https://doi.org/10.14245/ns.2244344.172.
NOTES
Conflict of Interest
Dr. Hofstetter is a consultant for Johnson & Johnson, Globus, Innovasis, and Joimax. Other authors have nothing to disclose.
Fig. 1.
(A) For unilateral laminotomy for bilateral recess decompression, bony decompression is carried out along the bony insertion of the ligamentum flavum (LF) (principal anatomical landmark) from ipsilateral to contralateral superior articular process (SAP) (blue arrow). (B) Upon minimal resection of inferomedial aspect of the ipsilateral inferior articular process the SAP is seen. The green dotted line shows the border of the LF and SAP. (C) The medial aspect of the SAP is resected and the traversing nerve root is decompressed and mobilized. Using the diamond burr the LF attachment on the rostral edge of the caudal index level lamina is followed to the contralateral side. (D) The asterisks show bilateral opening of the LF for lateral recess decompression. (E) The contralateral recess is decompressed using the burr and Kerrison rongeur. (F) The contralateral traversing nerve root (white arrow) is decompressed and mobilized. SP, pinous process.
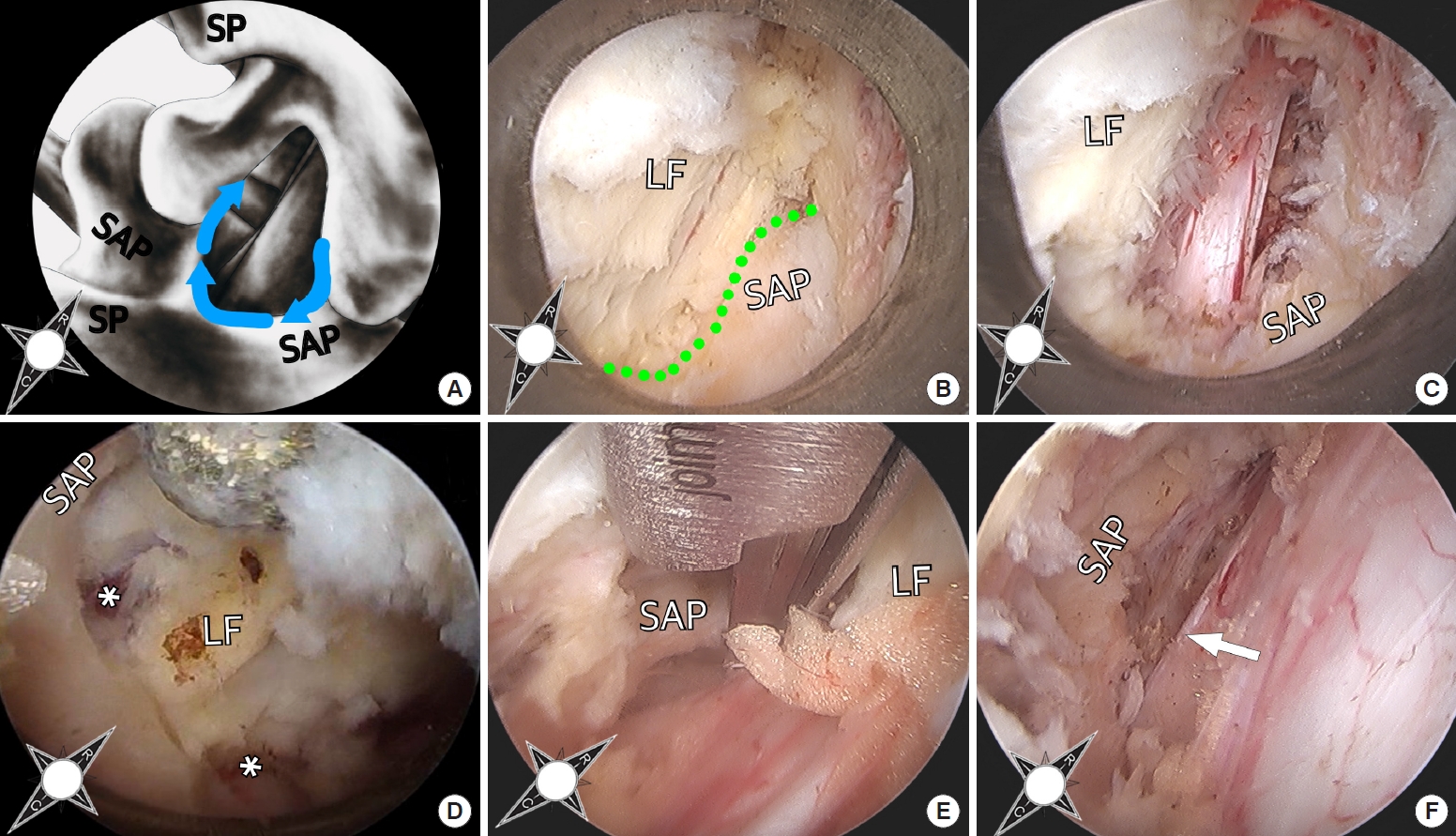
Fig. 2.
Graph depicting the age distribution of our patient cohort. A bimodal age distribution was observed with one patient cluster at the age of 40-mid 50 years and the other one at >mid-60 years. Red circles demarcate patients with accompanied degenerative lumbar scoliosis.
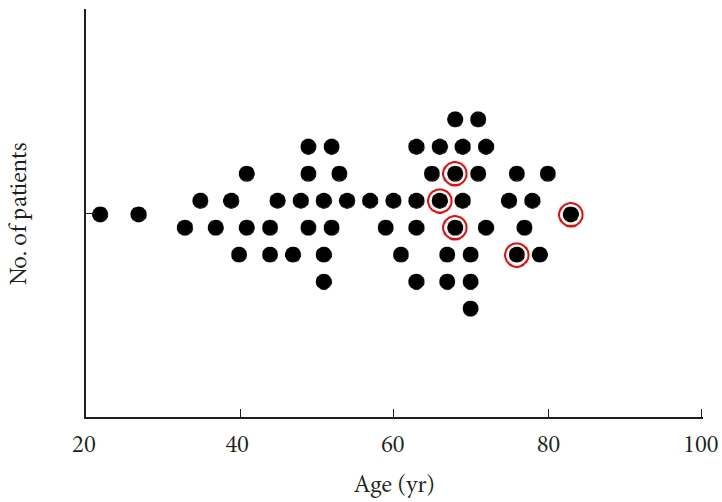
Fig. 3.
Pre- and posteroperative magnetic resonance imaging (MRI) following unilateral laminotomy for bilateral recess decompression. (A) Preoperative T2-weighted MRI depicts bilateral lateral recess stenosis. (B) Postoperative imaging confirms bilateral recess decompression while midline structures including paraspinal muscles, lamina, and yellow ligament are spared.
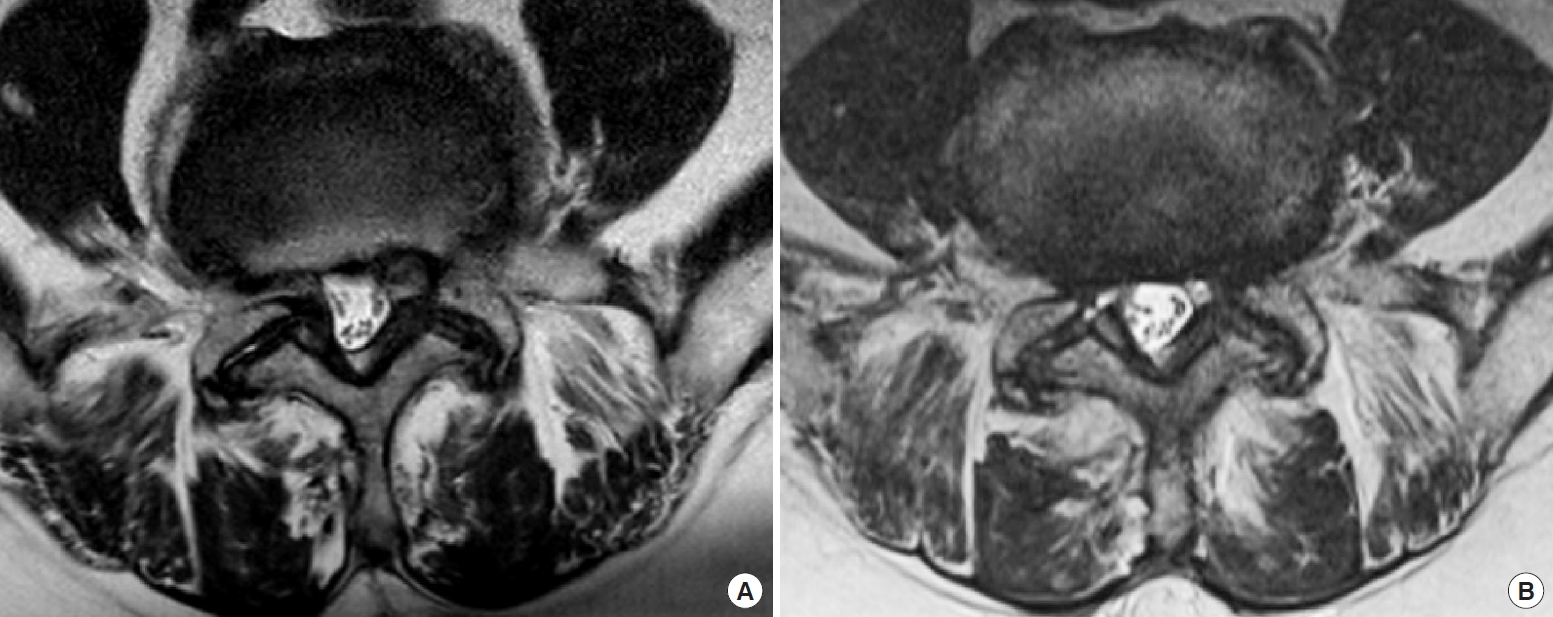
Fig. 4.
Postoperative (preop) back and leg pain following unilateral laminotomy for bilateral recess decompression (ULBRD). Both the visual analogue scale (VAS) for back pain (A) and the VAS for leg pain (B) improved significantly following ULBRD. Functional outcome scales including EuroQol-5 dimension (EQ-5D) self-reported questionnaires (C) and Oswestry Disability Index (ODI) (D) scores presented statistically significant improvement after ULBRD. *p<0.05, statistically significant differences.
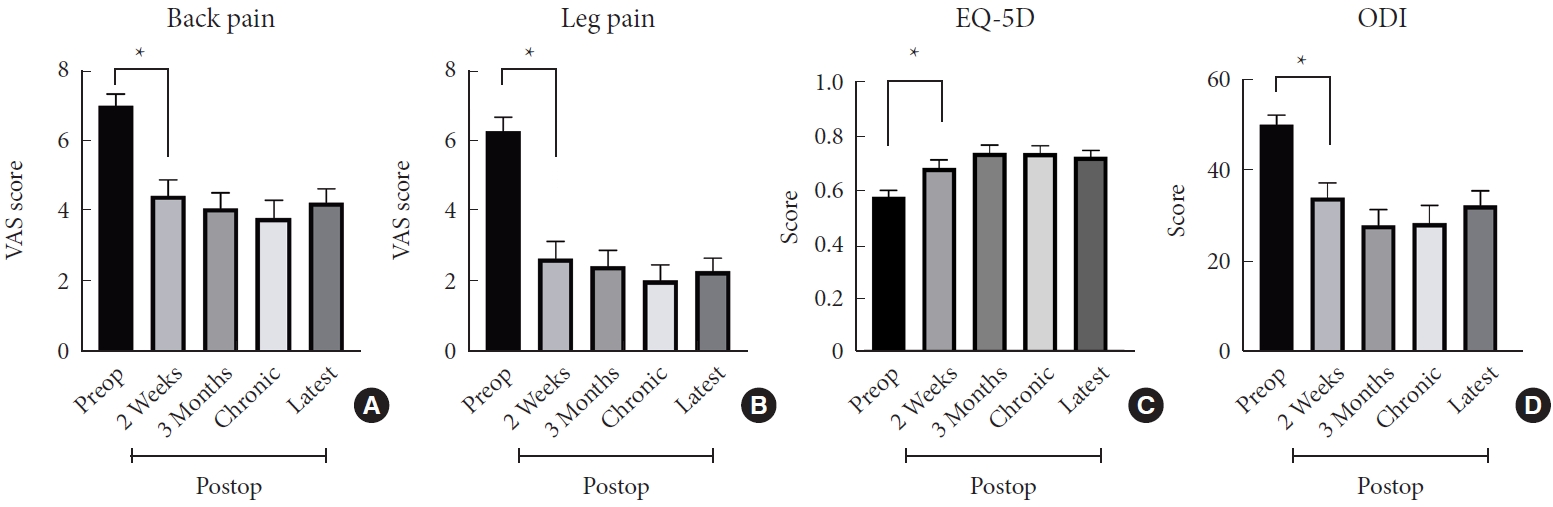
Table 1.
Patient demographic and clinical characteristics
Table 2.
Perioperative surgical details
Table 3.
Preoperative and postoperative functional evaluation
REFERENCES
1. Lee CH, Chung CK, Kim CH, et al. Health care burden of spinal diseases in the Republic of Korea: analysis of a nationwide database from 2012 through 2016. Neurospine 2018;15:66-76.




2. Splettstosser A, Khan MF, Zimmermann B, et al. Correlation of lumbar lateral recess stenosis in magnetic resonance imaging and clinical symptoms. World J Radiol 2017;9:223-9.



3. Ciric I, Mikhael MA, Tarkington JA, et al. The lateral recess syndrome. A variant of spinal stenosis. J Neurosurg 1980;53:433-43.


5. Suri P, Boyko EJ, Goldberg J, et al. Longitudinal associations between incident lumbar spine MRI findings and chronic low back pain or radicular symptoms: retrospective analysis of data from the longitudinal assessment of imaging and disability of the back (LAIDBACK). BMC Musculoskelet Disord 2014;15:152.




6. Jarvik JJ, Hollingworth W, Heagerty P, et al. The Longitudinal Assessment of Imaging and Disability of the Back (LAIDBack) study: baseline data. Spine 2001;26:1158-66.


7. Ruetten S, Komp M, Merk H, et al. Surgical treatment for lumbar lateral recess stenosis with the full-endoscopic interlaminar approach versus conventional microsurgical technique: a prospective, randomized, controlled study. J Neurosurg Spine 2009;10:476-85.


8. Atlas SJ, Keller RB, Wu YA, et al. Long-term outcomes of surgical and nonsurgical management of lumbar spinal stenosis: 8 to 10 year results from the maine lumbar spine study. Spine 2005;30:936-43.


9. Atlas SJ, Keller RB, Robson D, et al. Surgical and nonsurgical management of lumbar spinal stenosis: four-year outcomes from the maine lumbar spine study. Spine 2000;25:556-62.


10. Weinstein JN, Tosteson TD, Lurie JD, et al. Surgical versus nonsurgical therapy for lumbar spinal stenosis. N Engl J Med 2008;794-810.



11. Wilby MJ, Seeley H, Laing RJ. Laminectomy for lumbar canal stenosis: a safe and effective treatment. Br J Neurosurg 2006;20:391-5.


12. Fox MW, Onofrio BM, Onofrio BM, et al. Clinical outcomes and radiological instability following decompressive lumbar laminectomy for degenerative spinal stenosis: a comparison of patients undergoing concomitant arthrodesis versus decompression alone. J Neurosurg 1996;85:793-802.


13. Jonsson B, Annertz M, Sjoberg C, et al. A prospective and consecutive study of surgically treated lumbar spinal stenosis. Part II: five-year follow-up by an independent observer. Spine 1997;22:2938-44.


14. Guha D, Heary RF, Shamji MF. Iatrogenic spondylolisthesis following laminectomy for degenerative lumbar stenosis: systematic review and current concepts. Neurosurg Focus 2015;39:E9.



15. Alimi M, Hofstetter CP, Pyo SY, et al. Minimally invasive laminectomy for lumbar spinal stenosis in patients with and without preoperative spondylolisthesis: clinical outcome and reoperation rates. J Neurosurg Spine 2015;22:339-52.


16. Alimi M, Hofstetter CP, Torres-Campa JM, et al. Unilateral tubular approach for bilateral laminotomy: effect on ipsilateral and contralateral buttock and leg pain. Eur Spine J 2017;26:389-96.



17. Birjandian Z, Emerson S, Telfeian AE, et al. Interlaminar endoscopic lateral recess decompression-surgical technique and early clinical results. J Spine Surg 2017;3:123-32.



18. Lee CW, Yoon KJ, Kim SW. Percutaneous endoscopic decompression in lumbar canal and lateral recess stenosis - the surgical learning curve. Neurospine 2019;16:63-71.




19. Lee CH, Choi M, Ryu DS, et al. Efficacy and safety of fullendoscopic decompression via interlaminar approach for central or lateral recess spinal stenosis of the lumbar spine: a meta-analysis. Spine 2018;43:1756-64.


20. Ruetten S, Komp M, Godolias G. A New full-endoscopic technique for the interlaminar operation of lumbar disc herniations using 6-mm endoscopes: prospective 2-year results of 331 patients. Minim Invasive Neurosurg 2006;49:80-7.


21. Komp M, Hahn P, Oezdemir S, et al. Bilateral spinal decompression of lumbar central stenosis with the full-endoscopic interlaminar versus microsurgical laminotomy technique: a prospective, randomized, controlled study. Pain Physician 2015;18:61-70.


22. Hofstetter CP, Ahn Y, Choi G, et al. AOSpine Consensus paper on nomenclature for working-channel endoscopic spinal procedures. Global Spine J 2020;10(2 Suppl):111S-121S.




23. Lee CW, Yoon KJ, Ha SS. Comparative analysis between three different lumbar decompression techniques (microscopic, tubular, and endoscopic) in lumbar canal and lateral recess stenosis: preliminary report. Biomed Res Int 2019;2019:6078469.




24. Krzok G, Telfeian AE, Wagner R, et al. Contralateral facetsparing sublaminar endoscopic foraminotomy for the treatment of lumbar lateral recess stenosis: technical note. J Spine Surg 2017;3:260-6.



25. Kim HS, Patel R, Paudel B, et al. Early outcomes of endoscopic contralateral foraminal and lateral recess decompression via an interlaminar approach in patients with unilateral radiculopathy from unilateral foraminal stenosis. World Neurosurg 2017;108:763-73.


26. Ahn Y. Percutaneous endoscopic decompression for lumbar spinal stenosis. Expert Rev Med Devices 2014;11:605-16.


27. Schizas C, Theumann N, Burn A, et al. Qualitative grading of severity of lumbar spinal stenosis based on the morphology of the dural sac on magnetic resonance images. Spine 2010;35:1919-24.


28. Leone A, Guglielmi G, Cassar-Pullicino VN, et al. Lumbar intervertebral instability: a review. Radiology 2007;245:62-77.


29. Lewandrowski KU, Soriano-Sanchez JA, Zhang X, et al. Surgeon training and clinical implementation of spinal endoscopy in routine practice: results of a global survey. J Spine Surg 2020;6(Suppl 1):S237-48.



30. Nomura K, Yoshida M. Assessment of the learning curve for microendoscopic decompression surgery for lumbar spinal canal stenosis through an analysis of 480 cases involving a single surgeon. Global Spine J 2017;7:54-8.




31. Qin R, Liu B, Hao J, et al. Percutaneous endoscopic lumbar discectomy versus posterior open lumbar microdiscectomy for the treatment of symptomatic lumbar disc herniation: a systemic review and meta-analysis. World Neurosurg 2018;120:352-62.


32. Ruetten S, Komp M, Merk H, et al. Full-endoscopic interlaminar and transforaminal lumbar discectomy versus conventional microsurgical technique: a prospective, randomized, controlled study. Spine 2008;33:931-9.


33. Arslan M, Comert A, Acar HI, et al. Lumbosacral intrathecal nerve roots: an anatomical study. Acta Neurochir (Wien) 2011;153:1435-42.



34. Wu PH, Kim HS, Jang IT. A narrative review of development of full-endoscopic lumbar spine surgery. Neurospine 2020;17(Suppl 1):S20-33.




35. Lu HG, Pan XK, Hu MJ, et al. Percutaneous transforaminal endoscopic decompression for lumbar lateral recess stenosis. Front Surg 2021;8:631419.



36. Pairuchvej S, Muljadi JA, Ho JC, et al. Full-endoscopic (biportal or uni-portal) versus microscopic lumbar decompression laminectomy in patients with spinal stenosis: systematic review and meta-analysis. Eur J Orthop Surg Traumatol 2020;30:595-611.



37. Komp M, Hahn P, Merk H, et al. Bilateral operation of lumbar degenerative central spinal stenosis in full-endoscopic interlaminar technique with unilateral approach: prospective 2-year results of 74 patients. J Spinal Disord Tech 2011;24:281-7.


38. Hasan S, Hartl R, Hofstetter CP. The benefit zone of full-endoscopic spine surgery. J Spine Surg 2019;5(Suppl 1):S41-56.



39. Abumi K, Panjabi MM, Kramer KM, et al. Biomechanical evaluation of lumbar spinal stability after graded facetectomies. Spine 1990;15:1142-7.


40. Hamasaki T, Tanaka N, Kim J, et al. Biomechanical assessment of minimally invasive decompression for lumbar spinal canal stenosis: a cadaver study. J Spinal Disord Tech 2009;22:486-91.


41. Evins AI, Banu MA, Njoku I Jr, et al. Endoscopic lumbar foraminotomy. J Clin Neurosci 2015;22:730-4.


42. Foley KT, Smith MM, Rampersaud YR. Microendoscopic approach to far-lateral lumbar disc herniation. Neurosurg Focus 1999;7:e5.


43. Dasenbrock HH, Juraschek SP, Schultz LR, et al. The efficacy of minimally invasive discectomy compared with open discectomy: a meta-analysis of prospective randomized controlled trials. J Neurosurg Spine 2012;16:452-62.



44. Cruccu G, Aziz TZ, Garcia-Larrea L, et al. EFNS guidelines on neurostimulation therapy for neuropathic pain. Eur J Neurol 2007;14:952-70.


45. Ozay R, Yavuz OY, Turkoglu ME, et al. The effects of ankaferd blood stopper and microporous polysaccharide hemospheres on epidural fibrosis in rat laminectomy model. Acta Cir Bras 2015;30:799-805.


46. Cemil B, Tun K, Kaptanoglu E, et al. Use of pimecrolimus to prevent epidural fibrosis in a postlaminectomy rat model. J Neurosurg Spine 2009;11:758-63.


47. Ozay R, Ogur T, Durmaz HA, et al. Revisiting ligament-sparing lumbar microdiscectomy: when to preserve ligamentum flavum and how to evaluate radiological results for epidural fibrosis. World Neurosurg 2018;114:e378-87.


48. Li S, Xia H, Han C. Retrospective analysis on correlation factors of preserving the ligamentum flavum in microendoscopic discectomy. Clin Neurol Neurosurg 2015;139:46-50.


49. McGrath LB, White-Dzuro GA, Hofstetter CP. Comparison of clinical outcomes following minimally invasive or lumbar endoscopic unilateral laminotomy for bilateral decompression. J Neurosurg Spine 2019 Jan 11:1-9. https://doi.org/10.3171/2018.9.SPINE18689. [Epub].

- TOOLS































