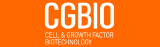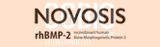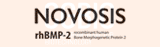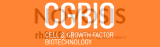Thoracolumbar OVCF is common in older patients, and its complications sometimes affect the quality of life. PVP has become a representative surgical treatment for OVCF with several advantages, and unilateral PVP has recently been widely performed because of its advantages, such as short operation time and reduced radiation exposure [
1,
2,
11-
13]. Clinical efficacy and incidence of SVCF, which are the most problematic complication following PVP, are strongly correlated with the surgery-related structural changes induced by BCV and BCD in and out of the fractured vertebral body [
10,
11,
14,
17,
18]. Nevertheless, these previous studies concluded that unilateral PVP is a better surgical method than bilateral PVP according to results comparing the postoperative pain relief, operation time, and radiation exposure alone rather than based on those comparing the surgery-related structural change and SVCF [
1,
2,
11-
13]. These previous studies drew exaggerated conclusions based on insufficient results because accurate quantitative measurements of the surgery-related structural changes of fractured vertebral bodies have been impossible in clinical studies thus far [
1,
2,
10-
14,
17]. To the authors’ knowledge, no study has compared and analyzed SVCF incidence between UEV and BTV using reasonable and accurate methods, nor has any study compared the surgery-related structural change caused by injected BCV and BCD between UEV and BTV. Moreover, BCV and BCD after PVP, which are critically related to efficiency and complication rate, remain clinical controversies unresolved in many previous studies because of methodological limitations. Therefore, to solve these clinical pending questions, collaborative research using VBM, which is the only technique that can overcome these methodological limitations, was performed in this study as it can precisely measure and quantitatively calculate the structural changes induced by BCV injected in and out of the fractured vertebral body after PVP [
10]. VBM has been introduced in brain research and applied for the first time in the clinical spinal research field in previous collaborative studies [
10,
23,
24,
28]. The BCV/VBV injected into the vertebral body has been closely associated with the clinical efficacy and SVCF incidence of PVP, mainly by previous
in vitro cadaveric studies [
10,
29,
30]. Although many biomechanical
in vitro cadaveric studies have been conducted on this topic, optimal BCV with the ideal clinical efficacy of PVP has not been established [
10,
29,
30]. A biomechanical
in vitro cadaveric study by Martinčič et al. [
31] indicated that 15% of the BCV/VBV can effectively strengthen the fractured vertebral body stiffness without increasing adjacent intradiscal pressure and 15%–20% of the BCV/VBV has a similar effect, and that relative BCV> 20% increases the adjacent intradiscal pressure resulting in increased pressure to the endplate of the adjacent vertebra, rather than stiffness restoration. Therefore, the study recommends 15% of the BCV/VBV as optimal BCV (4–6 mL of BCV depending on the vertebral level) [
31]. Several major biomechanical studies indicated that a larger BCV/VBV in the augmented vertebral body is positively correlated with AVCF, which may be caused by increased intradiscal pressure and pressure on the adjacent vertebral bodies [
29]. Notably, this biomechanical
in vitro cadaveric study’s results were consistent with our previously published clinical research findings using VBM [
10]. As VBM can calculate bone cement and vertebral body voxels, it enables the BCV/VBV measurement, which more efficiently reflects its biomechanical effect than BCV. In this study, the BCV/VBV was 0.22±0.03 and 0.29±0.03 in the UEV and BTV groups, respectively. Thermal injury to the pathologic nerve endings induced by injected bone cement alters and stabilizes the pain pathways in the surrounding tissue around the fractured vertebra [
32,
33]. This thermal effect causes immediate pain relief after PVP, and clinically, BCV for this process is not significantly correlated. Additionally, in this study, postoperative pain relief was not significantly different between the UEV and BTV groups (5.44±1.11 vs. 5.59±1.12, p=0.308, t-test). Specifically, even a small BCV/VBV of ≥ 10% injected into the vertebral body can induce immediate pain relief [
31-
33]. UEV was performed as previously reported [
21,
22]. UEV proposed by the authors has been devised as a method of needle placement at the center of the fractured vertebral body, and complications that occur during the needling process and bone cement injection can be reduced. It is highly useful at the thoracic level but requires proficiency. When needling is not optimal at the center of the irregularly shaped fractured vertebral body during UEV, large BCV may have to be injected unintentionally in order to distribute the bone cement well, leading to intradiscal leakage. Otherwise, the procedure may have to be completed with the not well-distributed bone cement in order to prevent intradiscal leakage or leakage to the spinal canal during the procedure despite injecting excessive BCV. Therefore, surgeons may have intuitive concerns and some negative clinical experiences with UEV. In contrast to intuitive concerns, the UEV procedure injected smaller but more optimal BCV/VBV efficiently without an increased risk of SVCF based on data calculated using VBM. Although unilateral PVP has been increasingly performed, there are still intuitive concerns and some clinical experiences that unilateral PVP cannot evenly and symmetrically well-distribute bone cement to both sides compared with bilateral PVP. These concerns about BCD of unilateral PVP were raised more frequently than regarding injecting sufficient BCV and have become the main reason for performing bilateral PVP. Therefore, a new surgical technique that uses a bending angle injector, in addition to percutaneous unilateral PVP, has recently been introduced [
34,
35]. However, studies on BCD have been limited. Most of the previous BCD studies only evaluated the bone cement location by counting the numbers of occupied sectors induced by injected bone cement after sectorizing the fractured vertebral body [
8]. A study by Liu et al. [
36] revealed that BCD evaluation using x-ray images is effective and can reliably predict SVCF after PVP. In contrast, a retrospective study by Lin et al. [
37] showed that BCD using unilateral PVP was correlated with clinical efficacy after PVP by classifying the various BCDs into only 3 groups based on x-ray images. Several similar studies that categorize and compare BCD according to patterns have been published [
11,
12]. However, these studies were not based on accurate quantitative analysis of BCD; therefore, the evidence level was low. Despite many previous studies about BCD, the correlation between BCD and clinical efficacy, including the incidence of SVCF after PVP, remains a pending question because the quantitative calculation of BCD in and out of the fractured vertebral body was impossible in
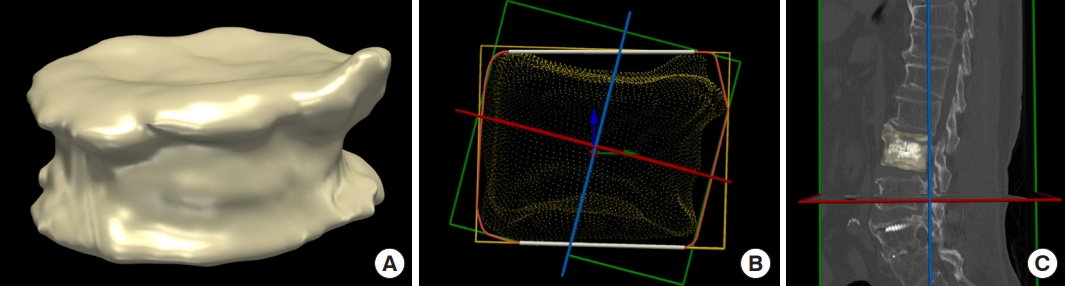
in vivo study using clinical data. Therefore, to solve the clinical issue of BCD, which is a more serious controversy than BCV, collaborative research using VBM was performed to quantitatively calculate the BCD in and out of the fractured vertebral body after PVP. Furthermore, the spatial, even, and symmetric distribution evaluation are required to evaluate the BCD, and clinical debate remains a pending question; therefore, the study was designed to meet these needs. Evaluation of spatial, even, and symmetric distribution was inevitable, particularly for comparative analysis between the UEV and BTV groups. VBM can automatically calculate the center of mass of the fractured vertebra and vectorize hundreds of thousands of bone cement unit voxels from this center [
10]. The vectorization of unit voxels enabled an accurate understanding of the location of the unit voxels. These calculated and vectorized bone cement unit voxels were plotted along the three axes, and a histogram plot chart of each axis was constructed (bottom-to-top, left-to-right, and AP axes) [
10]. Three-dimensional visualization using VBM and computed values based on these histograms along the 3 axes enabled the accurate quantitative comparison between the UEV and BTV groups for the first time. In the study, the UEV procedure spatially, evenly, and symmetrically well-distributed bone cement without increased risk of SVCF based on VBM in contrast to intuitive concerns and some clinical experiences, which indicate that bone cement cannot be evenly distributed to both sides in UEV compared with BTV (
Table 2,
Fig. 4C,
D). A 2D pixel corresponds to a 3D voxel. As the various unit voxels of the regions of interest, such as bone cement, vertebral body, adjacent intervertebral disc, and leaked intradiscal bone cement voxels, can be calculated using VBM, a small amount of intradiscal bone cement leakage can be confirmed, which was impossible in a previous x-ray-based study [
10].
Superior intradiscal bone cement leakage, which is a risk factor for AVCF, was frequent in the BTV group based on the data using VBM compared with that in the UEV group, without significant difference (UEV: 11%, BTV: 23%, p=0.056) (
Table 2). Although the BCV/VBV, which is known as a potential risk factor for AVCF, was significantly higher in the BTV group, no statistically significant difference was found in SVCF incidence between the 2 groups (SVCF, AVCF, and RVCF, p<0.303, p<0.227, and p<0.924, chi-square test) (
Table 3). Therefore, to evaluate the association between SVCF incidence and BCV and BCD using the UEV and BTV procedures, the effects of various risk factors for SVCF had to be reduced as much as possible [
9,
10,
38]. BMD is the single factor known to be the strongest risk factor for SVCF [
8-
10]. Only patients treated using bisphosphonates alone before and after PVP were enrolled in this study to reduce the BMD effect on SVCF, whereas those treated with receptor activator kappa B ligand (RANK-L inhibitor), SERM, and parathyroid hormone analogues (Teriparatide) were excluded [
10]. Additionally, multivariate logistic regression analysis was performed adjusting for age, sex, BMD, distribution along
z-axis, and upper intradiscal leakage to specifically reduce the BMD effect, which is the strongest risk factor for SVCF and other multifactorial risk factors [
8-
10]. The OR of SVCF incidence was 1.110 times higher in the BTV group than in the UEV group, and the 95% confidence interval (CI) at this time was 0.570–2.123, without significant difference (p=0.776,
Table 3). Models 2 and 3 showed an OR and 95% CI for AVCF and RVCF occurrence of 1.314 (0.564–3.066) and 0.951 (0.425–2.130), respectively, without significant difference (p=0.527 and p=0.904, respectively) (
Table 3). Furthermore, no statistically significant difference was found in SVCF incidence between the 2 groups based on this multivariate logistic regression analysis (
Table 3). The BCV/VBV of BTV was higher than that of UEV, with a significant difference (p<0.001, t-test). Consistent with these results, postoperative vertebral height restoration rate and KA correction in the BTP group were higher than that in the UEV group, without significant difference (p=0.151, Mann-Whitney U-test; p=0.844, chi-square test) (
Table 4) [
39,
40]. Although this was a retrospective study, the number of patients in each group was similar, and there were no criteria for choosing between UEV and BTV. However, the authors, as experienced surgeons in UEV, performed UEV at a higher rate with the rationale of performing minimally invasive procedures regardless of the spinal level than average experienced spine surgeons. Although it did not account for a large number, UEV was preferred for older patients who do not cooperate well during the procedures. This study had some limitations. First, we conducted a comparative analysis of clinical outcomes to evaluate the efficiency of 2 different PVPs.