- Search
|
|
||
Abstract
Objective
Methods
Results
Supplementary Material
Supplementary Table 2.
Supplementary Table 3.
NOTES
Funding/Support
This present study was supported by grants awarded by the Chinese Academy of Medical Sciences (CAMS) Innovation Fund for Medical Sciences (2021-I2M-C&T-B-016) and National High Level Hospital Clinical Research Funding (2022-PUMCH-B-112) for Jun Gao.
Author Contribution
Conceptualization: SH, FF, YL, JG; Data curation: SH, BH, FF; Formal analysis: SH, BH, ZL, FF, YL, JG; Funding acquisition: JG; Methodology: SH, BH, FF, YL, JG; Project administration: SH, BH, FF, YL, JG; Visualization: SH, BH, ZL; Writing - original draft: SH, BH, ZL, FF, YL, JG; Writing - review & editing: SH, BH, ZL, FF, YL, JG.
Fig. 1.

Fig. 2.

Fig. 3.
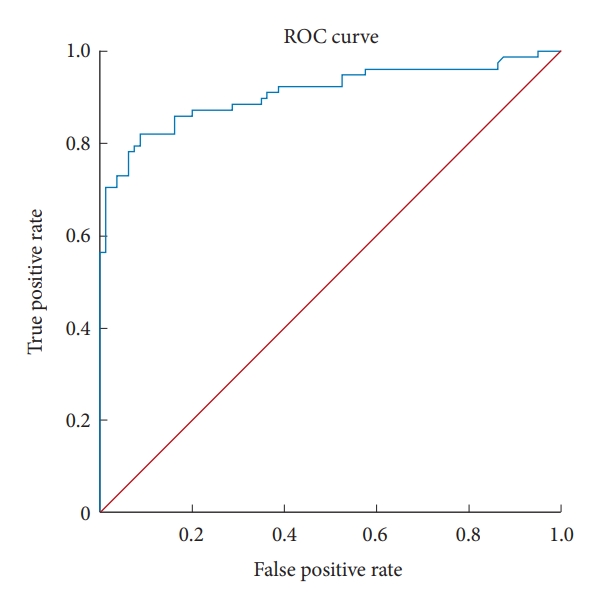
Table 1.
| Variable | Total (n = 78) | PFD (n = 22) | PFDD (n = 20) | PFDDO (n = 36) | p-value† |
|---|---|---|---|---|---|
| Sex | |||||
| Female | 60 (76.9) | 18 (81.8) | 17 (85.0) | 25 (69.4) | 0.335 |
| Male | 18 (23.1) | 4 (18.2) | 3 (15.0) | 11 (30.6) | |
| Age at surgery (yr) | 43.50 ± 16.25 | 43.68 ± 13.83 | 41.15 ± 12.58 | 42.28 ± 11.29 | 0.802 |
| Symptoms related to CSF obstruction | 0.320 | ||||
| Yes | 39 (50.0) | 14 (63.6) | 9 (45.0) | 16 (44.4) | |
| No | 39 (50.0) | 8 (36.4) | 11 (55.0) | 20 (55.6) | |
| Symptoms related to brainstem, cerebellar, or cranial nerve compression | 0.100 | ||||
| Yes | 26 (33.3) | 10 (45.5) | 3 (15.0) | 13 (36.1) | |
| No | 52 (66.7) | 12 (54.5) | 17 (85.0) | 23 (63.9) | |
| Symptoms related to spinal cord dysfunction | 0.695 | ||||
| Yes | 64 (82.1) | 17 (77.3) | 16 (80.0) | 31 (86.1) | |
| No | 14 (17.9) | 5 (22.7) | 4 (20.0) | 5 (13.9) | |
| Preop syrinx origin level | 0.912 | ||||
| Upper than C3 | 68 (87.2) | 19 (86.4) | 17 (85.0) | 32 (88.9) | |
| Lower than C4 | 10 (12.8) | 3 (13.6) | 3 (15.0) | 4 (11.1) | |
| Preop syrinx levels | 0.453 | ||||
| Involving C level alone | 24 (30.8) | 7 (31.8) | 4 (20.0) | 13 (36.7) | |
| Extending to T or lower levels | 54 (69.2) | 15 (68.2) | 16 (80.0) | 23 (63.9) | |
| Preop tonsillar herniation (mm) | 7.39 (11.01–5.63) | 7.72 (11.34–5.83) | 6.42 (10.92–5.31) | 6.86 (11.44–5.70) | 0.609‡ |
| Preop tonsillar descent (mm) | 41.01 ± 5.48 | 41.29 ± 6.23 | 41.10 ± 4.85 | 40.78 ± 5.47 | 0.941 |
| Preop cervical syrinx volume (mm3) | 4,147 (7,840.50–1,525.25) | 4,347 (8,282.50–1,649.50) | 2,191 (7,509.50–577.05) | 4,450 (8,046.00–2,695.88) | 0.243† |
| Preop aqueduct diameter (mm) | 1.75 (2.51–1.37) | 1.87 (2.44–1.50) | 1.57 (2.28–1.32) | 1.76 (2.80–1.38) | 0.342‡ |
| Preop aqueduct diameter scale | 0.273 | ||||
| ≥ 1.8 mm | 33 (42.3) | 12 (54.5) | 6 (30.0) | 15 (41.7) | |
| < 1.8 mm | 45 (57.7) | 10 (45.5) | 14 (70.0) | 21 (58.3) | |
| Preop pB–C2 (mm) | 8.73 ± 1.83 | 8.91 ± 1.95 | 8.60 (9.12–7.36) | 8.66 ± 1.70 | 0.538‡ |
| Preop pB–C2 scale | 0.440 | ||||
| ≥ 9 mm | 36 (46.2) | 10 (45.2) | 7 (35.0) | 19 (52.8) | |
| < 9 mm | 42 (53.8) | 12 (54.5) | 13 (65.0) | 17 (47.2) | |
Values are presented as number (%), mean±standard deviation, or median (interquartile range).
CMI, Chiari malformation type I; PFD, posterior fossa bony decompression alone; PFDD, PFD with duraplasty; PFDDO, PFDD with additional obex exploration; C, cervical spine; T, thoracic level; CSF, cerebrospinal fluid; preop, peoperative.
Table 2.
| Variable | Chiari (n = 78) | Control (n = 80) | p-value |
|---|---|---|---|
| Sex | 0.137 | ||
| Female | 60 (76.9) | 53 (66.3) | |
| Male | 18 (23.1) | 27 (33.8) | |
| Age (yr) | 43.50 (52.00–35.75) | 41.50 (49.00–35.25) | 0.546‡ |
| Preop tonsillar descent (mm) | 41.01 ± 5.48 | 32.17 ± 3.63 | < 0.001 |
| Preop aqueduct diameter (mm) | 1.75 (1.14) | 1.70 (0.48) | 0.542 |
| Preop pB–C2 (mm) | 8.73 ± 1.83 | 6.97 ± 1.52 | < 0.001 |
| Postop tonsillar descent (mm) | 38.71 ± 5.52 | - | < 0.001† |
| Postop aqueduct diameter (mm) | 1.54 (1.98–1.25) | - | < 0.001†,‡ |
| Postop pB–C2 (mm) | 8.34 (9.64–7.19) | - | 0.106†,‡ |
Table 3.
| Variable | Total (n = 78) | PFD (n = 22) | PFDD (n = 20) | PFDDO (n = 36) | p-value† |
|---|---|---|---|---|---|
| Postop tonsillar descent (mm) | 38.71 ± 5.52 | 39.15 ± 6.28 | 38.80 ± 4.25 | 38.38 ± 5.77 | 0.874 |
| Postop cervical syrinx volume (mm3) | 1,328 (3,627–553) | 1,106 (5,133–638) | 916 (2,090–244) | 1,964 (3,663–762) | 0.179‡ |
| Postop aqueduct diameter | 1.54 (1.98–1.25) | 1.54 (1.81–1.33) | 1.39 (1.80–1.19) | 1.65 (1.49–1.27) | 0.327‡ |
| Postop pB–C2 (mm) | 8.34 (9.64–7.19) | 9.08 (10.43–8.08) | 7.94 (9.19–6.88) | 8.20 (9.60–7.11) | 0.139‡ |
| Tonsillar descent resolution (mm) | 2.12 (3.09–0.80) | 1.31 (2.81–0.47) | 2.12 (4.47–0.45) | 2.27 (3.09–1.49) | 0.349‡ |
| Tonsillar descent resolution scale | 0.432 | ||||
| ≥ 2 mm | 40 (57.7) | 9 (40.9) | 10 (50.0) | 21 (58.3) | |
| < 2 mm | 38 (42.3) | 13 (59.1) | 10 (50.0) | 15 (41.7) | |
| Percentage change in cervical syrinx volume (%) | 42.94 (73.53–23.71) | 39.23 ± 28.56 | 51.66 ± 28.93 | 51.05 ± 28.96 | 0.258 |
| Cervical syrinx volume resolution scale | 0.550 | ||||
| ≥ 70 | 23 (29.5) | 4 (18.2) | 8 (40.0) | 11 (30.6) | |
| 30–70 | 28 (35.9) | 9 (40.9) | 5 (25.0) | 14 (38.9) | |
| < 30 | 27 (34.6) | 9 (40.9) | 7 (35.0) | 11 (30.6) | |
| CCOS scale | 0.506 | ||||
| 13–16 | 69 (88.5) | 20 (90.9) | 18 (90.0) | 31 (86.1) | |
| 9–12 | 8 (10.3) | 1 (4.5) | 2 (10.0) | 5 (13.9) | |
| 4–8 | 1 (1.3) | 1 (4.5) | 0 (0) | 0 (0) | |
| CCOS score | 15 (16–14) | 14.5 (15–14) | 14 (15-13.25) | 15 (16–14) | 0.091‡ |
| Pain | 4 (4–3) | 3 (4–3) | 3 (4–3) | 4 (4–3.25) | 0.072‡ |
| Nonpain | 3 (4–3) | 3 (4–3) | 3 (3.75–3) | 3 (4–3) | 0.692‡ |
| Functionality | 4 (4–4) | 4 (4–4) | 4 (4–3.25) | 4 (4–4) | 0.447‡ |
| Complications | 4 (4–4) | 4 (4–4) | 4 (4–4) | 4 (4–4) | 0.743‡ |
| Complications | 4 (5.1) | 1 (4.5) | 1 (5.0) | 2 (5.6) | > 0.999 |
| Meningitis | 3 (3.8) | 0 (0.0) | 1 (5.0) | 2 (5.6) | 0.610 |
| CSF leak | 0 (0) | 0 (0) | 0 (0) | 0 (0) | - |
| Wound poor healing | 1 (1.3) | 1 (4.5) | 0 (0) | 0 (0) | 0.538 |
| Follow-up (mo) | 73 (93–42.75) | 65 (72.25–55.75) | 82.50 (89.75–42.25) | 77 (95.75–25.75) | 0.246‡ |
| Radiological follow-up (mo) | 5 (10–2) | 7 (11–1) | 6 (11–2) | 4 (7–1) | 0.350‡ |
Values are presented as number (%), mean±standard deviation, or median (interquartile range).
CMI, Chiari malformation type I; PFD, posterior fossa bony decompression alone; PFDD, PFD with duraplasty; PFDDO, PFDD with additional obex exploration; preop, preoperative; CCOS, Chicago Chiari Outcome Scale; CSF, cerebrospinal fluid.
Table 4.
| Variable | Beta† | 95% CI | p-value |
|---|---|---|---|
| Male sex | -0.851 | -1.614 to 0.008 | 0.059 |
| Younger age at surgery (yr) | 0.034 | 0.006–0.063 | 0.018 |
| Symptoms related to CSF obstruction | -1.150 | -1.797 to -0.503 | 0.001 |
| Symptoms related to brainstem, cerebellar, or cranial nerve compression | 0.355 | -0.375 to 1.086 | 0.335 |
| Symptoms related to spinal cord dysfunction | -0.097 | -1.035 to 0.841 | 0.837 |
| Longer pB–C2 (≥ 9 mm) | 0.158 | -0.505 to 0.821 | 0.635 |
| More invasive surgical procedure | 0.241 | -0.156 to 0.638 | 0.230 |
| Syrinx originated from lower cervical levels (lower than C4) | 1.182 | 0.074–2.291 | 0.037 |
| Syrinx extending to T or lower levels | -0.066 | -0.769 to 0.637 | 0.851 |
| Narrower preop aqueduct (diameter < 1.8 mm) | 1.117 | 0.358–1.876 | 0.005 |
| Better tonsillar descent resolution scale (shrinkage ≥ 2 mm) | -0.176 | -0.899 to 0.547 | 0.629 |
| Better syrinx volume resolution scale | 0.701 | 0.237–1.165 | 0.004 |
| Longer follow-up (mo) | -0.004 | -0.017 to 0.008 | 0.471 |
Table 5.
| Variable | OR | 95% CI | p-value |
|---|---|---|---|
| Younger age at surgery | 1.019 | 0.978–1.062 | 0.373 |
| Higher CCOS score | 1.555 | 1.083–2.233 | 0.017 |
| Longer radiological follow-up | 1.060 | 0.995–1.129 | 0.073 |
| Sex | 0.252 | ||
| Male vs. female† | 1.935 | 0.625–5.992 | |
| pB–C2 (mm) | 0.885 | ||
| ≥ 9 vs. < 9† | 1.075 | 0.405–2.848 | |
| Surgical procedure | |||
| PFDD vs. PFDDO† | 0.817 | 0.249–2.685 | 0.739 |
| PFDD vs. PFD† | 1.257 | 0.399–3.953 | 0.696 |
| Syrinx origin | 0.688 | ||
| Lower than C4 vs. upper than C3† | 1.369 | 0.295–6.359 | |
| Preop syrinx levels | 0.817 | ||
| Involving C levels alone vs. extending to T or lower levels† | 1.128 | 3.133–0.406 | |
| Preop cerebral aqueduct diameter (mm) | 0.035 | ||
| < 1.8 vs. ≥ 1.8† | 3.341 | 10.265–1.087 | |
| Tonsillar descent resolution (mm) | 0.007 | ||
| ≥ 2 vs. < 2† | 4.052 | 1.47–11.17 |
REFERENCES

- TOOLS
-
METRICS

-
- 0 Crossref
- Scopus
- 1,268 View
- 127 Download
-
Journal Impact Factor 3.8
SURGERY: Q1
CLINICAL NEUROLOGY: Q1






























