 |
 |
- Search
| Neurospine > Volume 16(4); 2019 > Article |
|
|
Abstract
Objective
Pyogenic spinal infections account for 2%ŌĆō4% of orthopaedic infections. They are often difficult to diagnose, resulting in a delay in diagnosis. Risk factors for orthopaedic and spinal infection are well-documented in the literature, yet there is a paucity of studies examining risk factors specifically for multifocal spinal infections. The objective of this study was to identify predictors of multifocal spinal infections in comparison to unifocal spinal infections.
Methods
The medical records, imaging studies, and bacteriology data of 20 patients treated surgically for pyogenic spinal infection over 6 years at a tertiary referral center were reviewed and analyzed after receiving Institutional Review Board approval. Univariate and multivariate analyses were performed to identify factors associated with a multifocal spinal infection.
Results
Seven patients (35%) had multifocal infections. Three were bifocal, and 4 were trifocal. Patients with surgically treated cervical or thoracic spinal infections had a high rate of concomitant multifocal spinal infections (71% and 83%, respectively). Other potential predictors (e.g., patient age, body mass index, magnetic resonance image findings, etc.) did not reach statistical significance. Each of the multifocal infections involved the lumbar spine.
Conclusion
In this study, the spinal region was the only statistically significant risk factor for multifocal infection. Patients who are diagnosed with a spinal infection that requires operative treatment should have their entire spine evaluated with magnetic resonance imaging to detect multifocal involvement promptly.
Pyogenic spinal infections account for 2%ŌĆō4% of all bony infections and are a significant cause of orthopaedic morbidity [1-3]. They can be difficult to diagnose and may mimic or be masked by other problems [4,5]. Delay in treatment or untreated disease can lead to paraparesis or even death [4-7]. The clinical picture can be further complicated by the presence of a multifocal infection, defined as an infection located in more than one region of the spine (i.e., cervical, thoracic, and lumbar) [4,7-9]. Patients with multifocal spinal infections are at an increased risk of morbidity and mortality compared to unifocal infections, due to the greater extent of tissue involved. Therefore, it is important to promptly diagnose all affected regions of the spine so that proper treatment can be administered.
Pyogenic spinal infections may affect the disc, vertebra, facet joint, and dura [6]. The most frequent location of spondylodiscitis is the lumbar spine, followed by the thoracic and cervical spine [7]. The most frequent location of the epidural abscess is the cervical spine, followed by the thoracic and lumbar spine [6]. Numerous patient-related factors have been shown to increase the risk of spinal infection including an immunocompromised state, advanced age, chronic steroid usage, diabetes mellitus, human immunodeficiency virus (HIV), intravascular devices, intravenous drug abuse, malignancy, malnutrition, recent spinal surgery, renal failure, and septicemia [10-12]. While conservative treatment with parenteral antibiotics may be sufficient, surgical intervention becomes necessary when the infection is complicated by abscess formation, neurologic impairment, segmental instability, severe pain, and/or an absence of clinical response [7]. Magnetic resonance imaging (MRI) has the highest sensitivity and specificity for detecting spinal infections in comparison to plain radiographs, bone scans, and computed tomography (CT) imaging [4,13]. MRI is often the most reliable method of assessing the presence or absence of a spinal infection [14]. According to the American College of Radiology guidelines, emergent MRI of the spine is indicated in patients with new or worsening back pain, neurologic deficits, and signs or symptoms of spine infection [15].
To date, relatively few studies have investigated the predictors of multifocal spinal infection. Previous studies have cited risk factors for spinal infection [4-7,16] and multifocal infections [17-19], but none have identified specific risk factors for multifocal versus unifocal infections. The purpose of this study is to describe significant risk factors for multifocal spinal infection. Our results may promote timely recognition and diagnosis of multifocal involvement, which is correlated with improved patient outcomes [20,21]. A similar approach has been applied to noncontiguous fractures in the setting of spine trauma [22]. In the lumbar spine, the BatsonŌĆÖs paravertebral venous plexus may act as a potential route of infection, particularly in cases of sepsis originating from the pelvic organs [23]. The prevertebral pharyngeal venous plexus may allow bacteria to spread from the head and neck region to the cervical spine [24]. These differences in vascular anatomy led us to hypothesize that the initial level of involvement in the spine (either cervical, thoracic, or lumbar) is a significant predictor of multifocality.
We conducted a retrospective review of all pyogenic non-tuberculous spinal infections treated surgically by 1 of the 3 fellowship-trained spine surgeons at St. MaryŌĆÖs Medical Center from April 1, 2006 to March 31, 2012. Preliminary screening was performed using ICD-9 codes to search the electronic medical record for all potential patients. Different types of pyogenic spinal infections were considered, such as vertebral osteomyelitis, discitis, epidural abscess, and septic facet arthritis. The diagnostic criteria included characteristic MRI findings (e.g., hypointense signal on T1-weighted images, hyper-intense signal on T2-weighted images) and suggestive laboratory results (e.g., elevated white blood cell count [WBC], erythrocyte sedimentation rate [ESR], C-reactive protein [CRP], and lactate). The final diagnosis was based on clinical, radiological, and serologic evidence. Exclusion criteria were adolescents aged under 18 years and early postoperative infections (within 6 months of a previous spine operation). The human subjects review committee at St. MaryŌĆÖs Medical Center provided approval for this study.
Data were collected regarding demographics (age, sex, race), patient characteristics (weight, height, body mass index), predisposing factors (past medical history, social history), preoperative laboratory data (WBC, ESR, CRP, lactate), the indications for surgery, level of spinal involvement, bacteriology results (blood cultures, spinal tissue cultures), MRI/CT imaging, therapeutic management, and duration of hospitalization. Albumin <3.5 was used to define a state of malnutrition. A fellowship-trained musculoskeletal or neuro-radiologist reviewed each of the imaging studies and determined the presence of prevertebral soft tissue component involvement, epidural soft tissue component involvement, and T1 abnormality.
Patients with pyogenic infections involving multiple regions of the spinal column (e.g., cervical, thoracic, lumbar) were allocated to the ŌĆ£multifocal infectionŌĆØ group, including infections that were contiguous across regions (e.g., an epidural abscess spanning across the thoracolumbar junction). Patients in the ŌĆ£unifocal infectionŌĆØ group had infections involving one spinal region only, although multiple anatomical areas may have been involved (e.g., noncontiguous L2ŌĆō3 epidural abscess plus L4ŌĆō5 discitis).
Descriptive statistics, including the mean, variance, and standard deviation were calculated for continuous variables. Categorical variables were summarized with frequencies and proportions. Logistical regression and t-tests were performed using JMP ver. 11.0.0 (SAS Institute Inc., Cary, NC, USA). Regression coefficients were obtained for each independent variable when all other variables were held constant. Chi-square tests and t-tests with Tukey-Kramer correction were used to identify statistically significant differences between the multifocal and unifocal spinal infection groups. First, variables trending toward significance or that were significantly associated with multifocal infection were identified in a univariate logistical regression analysis (p<0.10). These variables were then entered into a multivariate logistical model to identify individual risk factors. A p-value of 0.05 was used as the significance level when evaluating multivariate significance tests.
Twenty subjects meeting the selection criteria were treated surgically for spinal infection by 1 of 3 fellowship-trained orthopaedic spine surgeons during the 6-year study period. The majority of patients were male (n=18, 90%), and 35% of our population had multifocal infections. Of the 7 subjects with multifocal infections, 3 were bifocal, and 4 were trifocal. Summary data of categorical and continuous variables are displayed in Tables 1 and 2, respectively.
All patients were started on empiric parenteral antibiotic therapy at the time of admission to the hospital, and therapy was modified according to culture results and sensitivities. Infectious disease consultation was obtained for all patients. Length of hospitalization ranged from 8ŌĆō92 days. The most common surgical procedures performed were incision & drainage of abscess with decompression (n=6), incision & drainage of abscess, decompression, interbody fusion with instrumentation (n=3), incision & drainage of abscess, decompression, and fusion with bone graft and instrumentation (n=3). The most common surgical procedures used to treat multifocal infections were incision & drainage of abscess, decompression, interbody fusion with instrumentation (n=3), and incision & drainage of abscess with decompression (n=2).
There were no statistically significant differences in mean age, weight, height, body mass index, WBC, ESR, CRP, or lactate between the unifocal and multifocal infection groups (p>0.05 for all comparisons). We also did not observe a statistically significant association between any of these continuous variables and multifocal infection on univariate analysis (p>0.05). Comparison of categorical data elements between unifocal and multifocal infection patients also revealed very few differences between the 2 populations. There were no statistically significant differences in sex, immunocompromised state, antibiotic use, history of spine surgery, smoking, intravenous drug use, malnourished state, diabetes, alcohol use, hepatitis C infection, HIV, hypercholesterolemia, or coronary artery disease between the unifocal and multifocal infection groups (p>0.05 for all comparisons). Rates of abnormal epidural findings on MRI, abnormal prevertebral findings, and abnormal T1 findings were also not significantly different between the 2 groups (p>0.05 for all comparisons). Among the 20 patients in our study, 35% had cervical infections, 30% had thoracic infections, and 85% had lumbar infections (Table 3). The presence of cervical and thoracic infections was significantly associated with multifocal infection on univariate analysis, as shown in Table 3 (p=0.022 and p=0.007, respectively). Cervical and thoracic involvement remained statistically significant in multivariate analysis (p=0.006 and p=0.001, respectively). No statistically significant difference was observed with lumbar involvement (p=0.52). Detailed clinical characteristics of patients with multifocal spine infections are shown in Fig. 1.
Bacteriology results were determined from blood cultures (17 of 20) and spinal tissue cultures (20 of 20). Bacteriological studies were positive for growth in 16 of 20 cases. Staphylococcus aureus was the most prevalent organism (9 isolates), of which 5 were methicillin-resistant Staphylococcus aureus (MRSA) and 4 were methicillin-sensitive Staphylococcus aureus (MSSA). The other organisms isolated were group A Streptococcus (n=1), Streptococcus viridans (n=1), Enterococcus (n=1), Escherichia coli (n=1), Streptococcus pneumoniae (n=1), and polymicrobial (n=2). Of the 7 multifocal infections, culture results included MRSA (n=1), MSSA (n=1), group A Streptococcus (n=1), Enterococcus (n=1), polymicrobial (n=1), and 2 were negative for growth (Table 4). There were no statistically significant differences in bacteriology between patients with unifocal versus multifocal infection.
An 80-year-old male with a history of smoking and intravenous drug use presented to our Emergency Department (ED) with shoulder pain and weakness 10 days after a shoulder biopsy (patient #3 in Table 4). In the ED, the patient was tachycardic and afebrile. Laboratory testing revealed a WBC count of 17.8 K/╬╝L. The patient was admitted to our hospital, but failed to improve despite treatment for suspected septic shoulder arthritis. He developed urinary retention, which prompted an MRI of the spine on hospital day 4. Imaging revealed extensive abscesses in the cervical, thoracic, and lumbar regions (Fig. 1-4, Table 4). He received urgent multistage surgical treatment. After a prolonged hospitalization of 75 days, he was medically stabilized for discharge to a skilled nursing facility, but remained quite disabled with significant weakness in all 4 extremities. This case demonstrates the difficulty in diagnosing and treating multifocal spinal infections, especially when the initial presentation is atypical.
In this study, patients with cervical or thoracic spinal infections were at a significantly increased risk of having a multifocal spine infection (71% and 83%, respectively). While 100% of the patients with multifocal infections had lumbar involvement, 59% of the lumbar spinal infections were unifocal. Although there are relatively few studies on multifocal spinal infection [17-19], previous authors have suggested that multifocal spinal infections are likely underreported and increasing in frequency [12].
In our study, the clinical characteristics of the multifocal and unifocal infection groups were not significantly different except for cervical and thoracic involvement (spinal region). Since the rate of multifocality is high in our study (35%), we recommend an MRI of the entire spine when surgical treatment of spinal infection is indicated. Complete spine imaging ensures that other sites of infection are not missed, especially since presenting signs and symptoms may not always lead to a clear diagnosis. Physical examination findings of multifocal spinal infection may primarily localize to the region where it is most severe. Therefore, concomitant infections at other regions of the spine (e.g., cervical, thoracic, or lumbar) can be frequently missed. Signs and symptoms of spine infection can also be vague and nonspecific. Back or neck pain may be absent in up to 15% of spine infection cases [25]. Delayed diagnosis of spinal infections can have devastating consequences [4,10]. Previous studies suggest that patients with spondylodiscitis have substantially increased mortality and high rates of concurrent meningitis, endocarditis, or sepsis [26]. Reported rates of mortality in patients with epidural abscess ranges from 10% to 20%, and the incidence of permanent neurologic deficit may be as high as 30%ŌĆō50% [27]. According to Hadjipavlou et al. [28], the risk of severe neurological deficit (e.g., paraplegia) resulting from an epidural abscess is considerably higher in the thoracic spine and cervical spine, compared to the lumbar spine [6]. As a screening tool, we recommend obtaining sagittal T1- and T2-weighted sequences of the entire spine and axial images as needed. In some countries, it is routine practice to obtain pan-spine sagittal images (most often T2-weighted). In other countries with aggressive medico-legal systems (such as the United States), only a limited part of the spine is typically imaged during a given examination. Anecdotally, this is felt to protect radiologists legally, in cases where significant pathology is missed in incompletely visualized regions of the spine.
Imaging features suggestive of increased infection severity may also predict multifocality in spinal discitis/osteomyelitis. A decreased T1 signal intensity of the endplates strongly suggests the inoculation of the bone and is a more sensitive predictor of osteomyelitis than fluid-sensitive sequences alone [15]. The presence of a paravertebral abscess has also been used as a predictor of infection severity and hematogenous spread to other regions of the spine. However, there were no statistically significant differences between multifocal and unifocal spinal infections in terms of these MRI characteristics. A larger sample size may be sufficiently powered to detect differences in these imaging findings between unifocal and multifocal infections.
A strength of this study is the relatively high percentage of multifocal spinal infections in our cohort (35%). Prior studies have reported much lower rates of noncontiguous spine infection [29]. The cohort was derived from a single hospital and treated by providers within the same spine group; therefore, the evaluation, diagnosis, and treatment of these spinal infections were relatively uniform. Finally, our study investigated many different variablesŌĆöpatient factors, spinal diagnoses, levels of involvement, laboratory results, bacteriology, and MRI findingsŌĆöany of which could potentially be associated with multifocal spinal infections.
There were limitations to the study. The overall cohort size was small (n=20), and our study may have been underpowered to detect all possible predictors of multifocality versus unifocality. Increasing the cohort size would increase the power of the study and allow for more subtle differences to be found. In addition, our study was limited to patients with pyogenic spinal infections who required surgical treatment. Spine infection patients without neurological deficits, spinal instability, or intractable pain may be treated conservatively with antibiotic therapy, immobilization, or CT-guided percutaneous drainage [30]. Few studies have compared clinical outcomes of pyogenic spinal infections treated with surgical intervention versus conservative management [31]. However, 6 out of 7 patients with multifocal infections in our study failed a trial of nonoperative treatment. Since spine imaging is often unnecessary in patients who are undergoing conservative management, we limited our analysis to spine infection patients who required surgery [15]. Furthermore, the large percentage of multifocal spine infections in our study may be attributable to the patient population who seeks care at our hospital, which is a tertiary referral center and also receives a substantial number of indigent patients and injection drug users. The patient population presenting to our institution may not be representative of the population at other medical centers, which may limit the generalizability of our results. Another limitation is that we excluded patients with Mycobacterium tuberculosis, Brucella, and fungal species, as these granulomatous organisms can classically cause multilevel discitis [32]. Multiple prior studies have recommended the use of the whole spine MRIs for the diagnosis and evaluation of suspected vertebral tuberculosis [33,34].
Recent publications have reported contradictory recommendations for spine imaging when the infection is suspected. In a retrospective study of 91 patients with tuberculous (TBS) and pyogenic spondylodiscitis (PS), Abbara et al. [18] found that 33% of TBS cases had multiple region involvement, versus none in the PS group. The authors concluded that pan-spine imaging is necessary in cases of TBS, but not in cases of PS. However, it is unclear whether the subjects received pan-spinal scans or imaging of symptomatic regions only. Another study of 1,138 consecutive cases of spondylitis over 16 years reported that nearly 7% of cases were noncontiguous multifocal infections in otherwise clinically silent disease [17,19]. The authors recommended pan-spine MRIs to detect multifocal infection regardless of patient symptomatology. This study demonstrated the importance of identifying clinically silent levels, especially before surgical intervention. The most common pattern of involvement was thoracic to lumbar seen in 43% of cases, followed by lumbar to lumbar in 16%, and cervical to lumbar in 14%. Our findings were similar in that the majority of multifocal infections started in the cervical or thoracic regions and spread to the lumbar spine. In contrast to our study, patient comorbidities and laboratory results were not compared between unifocal and multifocal infection cases. Future studies with an increased sample size may identify other potential predictors of multifocal spine infection.
In our retrospective study of 20 patients requiring surgery for spinal infection, the rate of multifocality was 71% and 83% if the cervical and thoracic regions were involved. While the rate of multifocality was only 41% in the lumbar region, 100% of multifocal infections in this study involved the lumbar spine. Therefore, we recommend that the entire spine be evaluated with an MRI when a patient is diagnosed with any spinal infection that requires operative treatment. Pan-spine imaging may lead to earlier detection and treatment of multifocal infections.
Fig.┬Ā1.
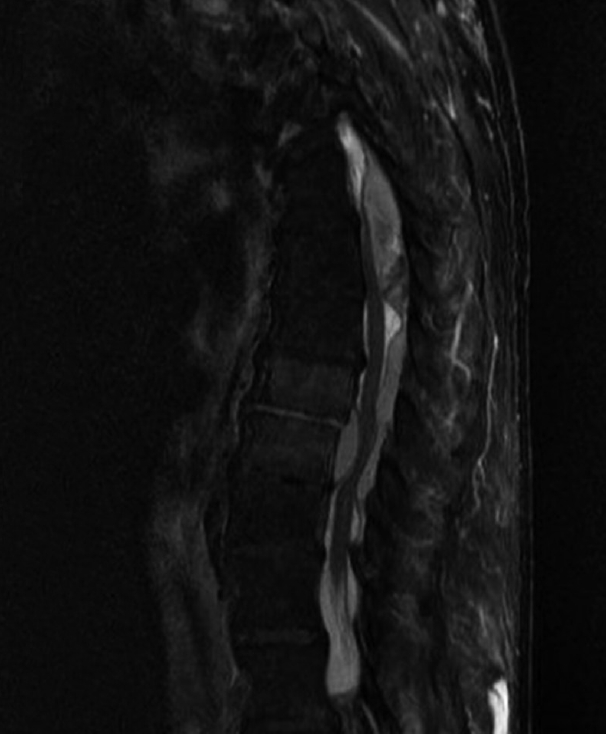
(A) T2-weighted sagittal magnetic resonance image of the lumbar spine demonstrating early spondylodiskitis at several nonadjacent levels, (B) sagittal Short tau inversion recovery or STIR sequence, (C) Axial T2-weighted image showing psoas musculature hyper-intensity (psoas sign).
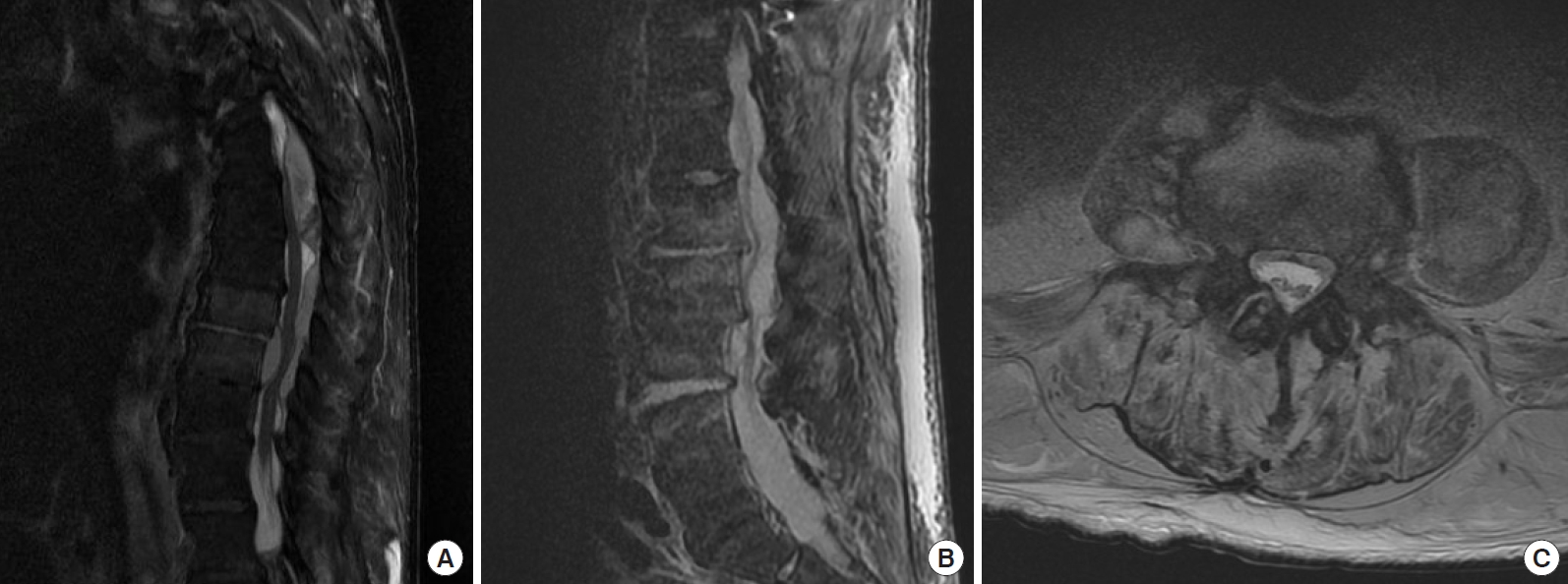
Fig.┬Ā2.
T2-weighted sagittal magnetic resonance image showing discontinuity of the thoracic and lumbar epidural abscesses.
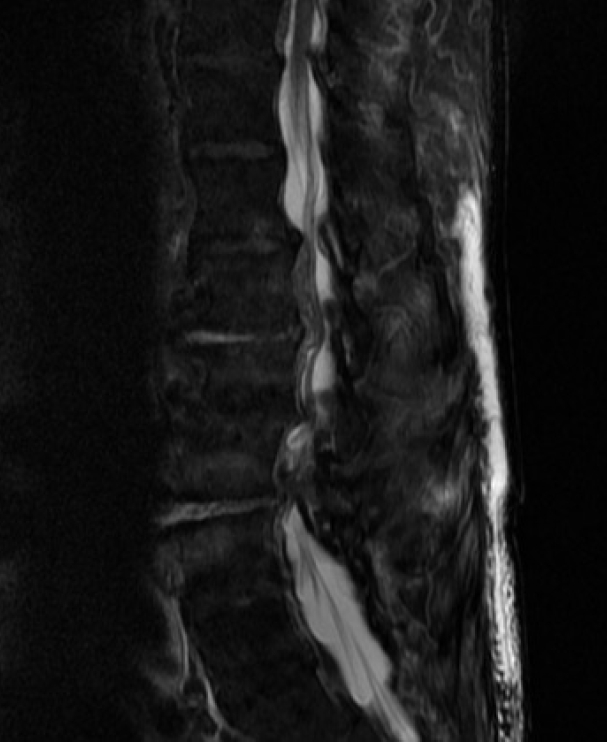
Fig.┬Ā3.
T2-weighted sagittal magnetic resonance image demonstrating extensive ventral epidural collection from C2 to T5, resulting in severe canal stenosis and spinal cord compression. Note the discontinuous dorsal epidural abscess in the upper thoracic spine as well.
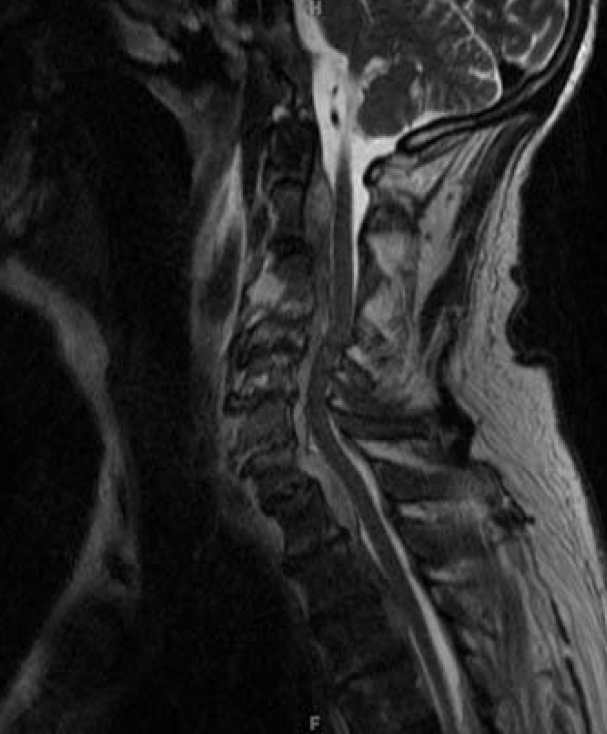
Fig.┬Ā4.
T2-weighted sagittal magnetic resonance image of the thoracic spine in the same patient, demonstrating massive ventral and dorsal epidural abscesses as well as early spondylodiscitis in the mid-to-lower thoracic spine. This results in severe canal stenosis and spinal cord compression.
Table┬Ā1.
Summary data of categorical variables collected during the study
Table┬Ā2.
Summary data of continuous variables collected during the study
Table┬Ā3.
Summary of unifocal and multifocal infections by region
| Region | Infections | Unifocal infections | Multifocal infections | p-value |
|---|---|---|---|---|
| Cervical | 7 (35) | 2 (29) | 5 (71) | 0.022 |
| Thoracic | 6 (30) | 1 (17) | 5 (83) | 0.007 |
| Lumbar | 17 (85) | 10 (59) | 7 (41) | 0.521 |
Table┬Ā4.
Detailed clinical characteristics of patients with pyogenic multifocal spine infections
REFERENCES
1. Danner RL, Hartman BJ. Update on spinal epidural abscess: 35 cases and review of the literature. Rev Infect Dis 1987 9:265-74.



2. Joughin E, McDougall C, Parfitt C, et al. Causes and clinical management of vertebral osteomyelitis in Saskatchewan. Spine (Phila Pa 1976) 1991 16:261-4.


3. Krogsgaard MR, Wagn P, Bengtsson J. Epidemiology of acute vertebral osteomyelitis in Denmark: 137 cases in Denmark 1978-1982, compared to cases reported to the National Patient Register 1991-1993. Acta Orthop Scand 1998 69:513-7.


4. Butler JS, Shelly MJ, Timlin M, et al. Nontuberculous pyogenic spinal infection in adults: a 12-year experience from a tertiary referral center. Spine (Phila Pa 1976) 2006 31:2695-700.


5. Malawski SK, Lukawski S. Pyogenic infection of the spine. Clin Orthop Relat Res 1991 (272):58-66.

6. Hadjipavlou AG, Mader JT, Necessary JT, et al. Hematogenous pyogenic spinal infections and their surgical management. Spine (Phila Pa 1976) 2000 25:1668-79.


7. Korovessis P, Repantis T, Hadjipavlou AG. Hematogenous pyogenic spinal infection: current perceptions. Orthopedics 2012 35:885-92.


8. Chow GH, Gebhard JS, Brown CW. Multifocal metachronous epidural abscesses of the spine. A case report. Spine (Phila Pa 1976) 1996 21:1094-7.


9. Deshmukh VR. Midline trough corpectomies for the evacuation of an extensive ventral cervical and upper thoracic spinal epidural abscess. J Neurosurg Spine 2010 13:229-33.


10. Gasbarrini AL, Bertoldi E, Mazzetti M, et al. Clinical features, diagnostic and therapeutic approaches to haematogenous vertebral osteomyelitis. Eur Rev Med Pharmacol Sci 2005 9:53-66.

11. Reihsaus E, Waldbaur H, Seeling W. Spinal epidural abscess: a meta-analysis of 915 patients. Neurosurg Rev 2000 23:175-204.



12. Sampath P, Rigamonti D. Spinal epidural abscess: a review of epidemiology, diagnosis, and treatment. J Spinal Disord 1999 12:89-93.


13. Dagirmanjian A, Schils J, McHenry MC. MR imaging of spinal infections. Magn Reson Imaging Clin N Am 1999 7:525-38.


14. Carragee EJ. The clinical use of magnetic resonance imaging in pyogenic vertebral osteomyelitis. Spine (Phila Pa 1976) 1997 22:780-5.


15. Lavi ES, Pal A, Bleicher D, et al. MR imaging of the spine: urgent and emergent indications. Semin Ultrasound CT MR 2018 39:551-69.


16. Mann S, Sch├╝tze M, Sola S, et al. Nonspecific pyogenic spondylodiscitis: clinical manifestations, surgical treatment, and outcome in 24 patients. Neurosurg Focus 2004 17:E3.

17. Ahmed Ezzat Siam, Hamdan Abdelrahman, Hassan Allouch, et al. Multi-level non-contiguous spinal infections. series of 77 cases in a single institution. Eur Spine J 2013 22:2582-669.



18. Abbara A, Tivey A, John L, et al. Whole spine imaging is justified in tuberculous spondylodiscitis but not pyogenic spondylodiscitis. J Infect 2016 72:125-6.


19. Siam AE. Whole spine MRI should be recommended for pyogenic spondylodiscitis; response to Abbara et al. J Infect 2016 72:631.


20. Redekop GJ, Del Maestro RF. Diagnosis and management of spinal epidural abscess. Can J Neurol Sci 1992 19:180-7.


22. Vaccaro AR, An HS, Lin S, et al. Noncontiguous injuries of the spine. J Spinal Disord 1992 5:320-9.


23. Batson OV. The vertebral system of veins as a means for cancer dissemination. Prog Clin Cancer 1967 3:1-18.

24. Wiley AM, Trueta J. The vascular anatomy of the spine and its relationship to pyogenic vertebral osteomyelitis. J Bone Joint Surg Br 1959 41-B:796-809.


25. Roblot F, Besnier JM, Juhel L, et al. Optimal duration of antibiotic therapy in vertebral osteomyelitis. Semin Arthritis Rheum 2007 36:269-77.


26. Aagaard T, Roed C, Larsen AR, et al. Long-term mortality after Staphylococcus aureus spondylodiscitis: a Danish nationwide population-based cohort study. J Infect 2014 69:252-8.


27. Schoenfeld AJ, Wahlquist TC. Mortality, complication risk, and total charges after the treatment of epidural abscess. Spine J 2015 15:249-55.


28. Hadjipavlou AG, Mader JT, Necessary JT, et al. Hematogenous pyogenic spinal infections and their surgical management. Spine (Phila Pa 1976) 2000 25(13):1668-1679.


29. Shiban E, Janssen I, Wostrack M, et al. A retrospective study of 113 consecutive cases of surgically treated spondylodiscitis patients. A single-center experience. Acta Neurochir (Wien) 2014 156:1189-96.



30. Duarte RM, Vaccaro AR. Spinal infection: state of the art and management algorithm. Eur Spine J 2013 22:2787-99.




31. Rutges JP, Kempen DH, van Dijk M, et al. Outcome of conservative and surgical treatment of pyogenic spondylodiscitis: a systematic literature review. Eur Spine J 2016 25:983-99.



32. Teo M, Trivedi R, Murphy M. Non-contiguous multifocal Staphylococcus aureus discitis: involvement of the cervical, thoracic and lumbar spine. Acta Neurochir (Wien) 2010 152:471-4.




- TOOLS
- Related articles in NS
-
Journal Impact Factor 3.2



























